
Inflammation in CKD
By Majd Isreb, MD, FACP, FASN, IFMCP
Causes of inflammation in chronic kidney disease
Inflammatory markers were found to be elevated in CKD. The levels of these markers increase with the decrease in kidney function. They also increase with increasing urine protein (albuminuria).
There are several reasons for immune imbalance and inflammation in CKD. Some of these factors are linked directly to the cause of CKD, while others are related to genetic predisposition and epigenetic factors.
Uremic environment
Furthermore, the accumulation of uremic toxins in CKD leads to oxidative stress. We discussed oxidative stress in a previous blog. When oxidative stress (or free radicals) builds up and aren’t handled properly, they can damage enzymes, cells, and tissues. Oxidative stress has been identified as a root cause of many diseases, including heart disease, Alzheimer’s, Parkinson’s, chronic kidney disease, and many more.
The build-up of harmful oxidative toxins in advanced CKD is associated with another problem which is described as “carbonyl stress.” Here, there are irreversible changes in many proteins leading to loss of their function. Higher oxidative and carbonyl stress is linked to inflammation in CKD. It is possible that carbonyl stress is just a biomarker of oxidative stress in CKD and not an independent phenomenon.
Download Your Copy!
Metabolic acidosis
Metabolic acidosis and high dietary acid load are very common in CKD. In metabolic acidosis, there is an increase in blood acidity. The foods that we eat may generate blood acidity (such as animal proteins) or alkalinity (such as fruits and vegetables). This is described as the dietary acid load We discussed these in a previous blog. They play an important role in the bone disease and protein-energy wasting that are common in CKD. In addition to that, a higher dietary acid load has been linked to faster progression of kidney disease. Metabolic acidosis has been linked to inflammation in CKD. In fact, higher inflammatory cytokines were seen in patients with metabolic acidosis.
Oral disease
Oral and periodontal disease (gum disease) are common in CKD. Over 700 species of microbes reside in the mouth. Together, they are called the oral microbiome. This microbiome produces metabolic by-products in the mouth. Some of these leaks into the bloodstream and lead to low-grade systemic inflammation.
Studies have shown that patients with severe gum disease have elevated levels of pro-inflammatory mediators and increased numbers of white blood cells known as neutrophils in the blood. This suggests that the inflammatory process in the mouth is affecting the whole body and probably injuring organs and tissues. In fact, successful treatment of gum disease was associated with improvement in inflammatory markers.
Intestinal dysbiosis
In CKD there is something called the dysbiosis cycle. We discussed this in a previous blog. Dysbiosis is a state of the intestinal microbiota where “bad” bacteria (or other organisms like yeast or parasites) outgrow the “good” bacteria. In dysbiosis, we see a rise in inflammatory markers that interact with the lining of the gut and result in damage and increased intestinal permeability (also known as “leaky gut”). “Leakiness” of the gut lining allows gut bacteria or bacterial particles from the gut into the bloodstream. It causes shifts in the breakdown of nutrients, including amino acids, which leads to the formation of many gut-derived uremic toxins. Intestinal permeability has been linked to autoimmune disease and systemic inflammation.
Vitamin D deficiency in CKD
Vitamin D deficiency in chronic kidney disease is quite common with 80% of CKD patients having been found to have low levels of 25-hydroxy vitamin D (25 (OH)D) when measured in the serum. Several factors have been implicated in the cause of this deficiency including inadequate outdoor physical activity, inadequate dietary intake, genetic variations, and loss of 25(OH)D in the urine. Vitamin D deficiency has been associated with an imbalanced immune system in CKD. This link between deficiency of vitamin D and kidney disease is important because it can lead to further inflammation in CKD.
Obesity
A higher body mass index is one of the strongest risk factors for CKD. In fact, the incidence of obesity-related kidney disease has increased 10-fold in recent years. Higher visceral fat is linked to increased protein in the urine (albuminuria). Visceral fat was also found to be associated with higher circulating inflammatory cytokines such as IL-6. This is because visceral fat cells produce these inflammatory cytokines.
Decreased clearance of cytokines
It has been well-documented that the levels of circulating inflammatory cytokines are elevated in kidney disease. Cytokines are chemical messengers that are produced by cells of the immune system. There are inflammatory cytokines and anti-inflammatory cytokines. When inflammatory cytokines are high in the blood, it is like a burning fire in the body, damaging and irritating tissues. In addition to the increased generation of cytokines in CKD, decreased renal clearance plays a significant role in the high level of cytokines in the blood.
Symptoms of inflammation in CKD
Higher levels of inflammatory markers such as IL-1beta, IL-6, TNF-alpha, C-reactive protein (CRP), and fibrinogen were found in patients with CKD, according to the Chronic Renal Insufficiency Cohort (CRIC) study. Inflammation is associated with many problems in patients with CKD. These problems include:
- Faster progression of kidney disease
- Increased protein loss in the urine
- Increased risk of heart disease and death
- Increased itching
- Worsening nutrition and protein-energy wasting
- Depression
- Worsening bone-mineral disorder
- Worsening anemia
Healing inflammation in CKD
When thinking about the best treatment for inflammation in CKD, it is crucial to address its root cause. For example, correcting the uremic environment may include strategies to decrease animal protein intake through a plant-dominant dietand a personalized gut restoration protocol. To address vitamin D deficiency, it is always recommended to check for it and correct it in all CKD patients.
Optimizing a person’s nutritional status is fundamental in healing inflammation in CKD and improving all the outcomes mentioned above. In addition, many supplements have been found to improve inflammation in CKD. These include omega-3 fatty acids, decaffeinated green tea extract, pomegranate, soy isoflavones, fiber, and probiotics.
The bottom line on inflammation and CKD
Low-grade inflammation is a common feature of CKD. Multiple root causes can be identified as a source of inflammation in kidney patients. These include decreased clearance of cytokines, oral and intestinal dysbiosis, vitamin D deficiency, uremic toxins, metabolic acidosis, and obesity. It is crucial to address the root causes of inflammation to improve kidney and heart outcomes in these patients.
The post Inflammation in CKD appeared first on Integrative Kidney.]]>Recently, there has been a growing interest in the gut-kidney axis, which refers to the overlapping relationship between gut integrity, microbiome diversity, resulting inflammatory process and kidney disease. The interaction between the gut and kidneys is very complex and can be divided into two major categories: the gut-derived uremic toxins that can worsen kidney disease, and the inflammatory autoimmune response that can trigger kidney disease. In this blog, we will focus on the gut-derived uremic toxins.
Uremic Retention Molecules (URMs)
Uremic toxins, accumulated waste products due to decreased renal clearance, have been linked to systemic inflammation. Recently, researchers have been successful at identifying many of these molecules thanks to a technique called metabolomic analysis. In general, URMs are divided into three categories:
- Small water-soluble molecules
- Middle size molecules
- Protein-bound uremic molecules
However, for our purpose, it is more helpful to divide these toxins according to their origin:
- Endogenous uremic toxins: toxins or waste that naturally and normally occur during metabolism or catabolism
- Exogenous uremic toxins: related to dietary intake
- Gut-derived uremic toxins: generated by pathogenic gut microbiota in the presence of dysbiosis
Block "fundamentals" not found
Gut-derived uremic toxins
The human gut contains trillions of microorganisms, collectively referred to as the gut microbiota or microbiome . The composition of the gut microbiome varies from person to person due to genetics, environmental factors, dietary, and disease state. These microorganisms are in constant communication with the body. They also produce multiple metabolites, nutrients, and cell signals for various physiological functions. [read more about the microbiome here]
Dysbiosis describes a state where “bad” bacteria (or other organisms like yeast or parasites) outgrow the “good” bacteria. In dysbiosis, we see a rise in inflammatory markers that interact with the lining of the gut and result in damage and increased intestinal permeability (“leaky gut”). This causes shifts in the breakdown of nutrients, including amino acids, which leads to the formation of many gut-derived uremic toxins.
Thanks to recent advances in metabolomic analysis, researchers have identified more than 60 uremic toxins that are derived from the gut. A number of studies have demonstrated that URMs are associated with high cardiovascular burden and mortality in CKD. There are four major gut-derived uremic toxins associated with CKD: Indoxyl sulfate (IS), para-Cresyl sulfate (pCS), Trimethylamine-N-oxide (TMAO), and Indole acetic acid (IAA).
Indoxyl sulfate (IS)
IS is a protein-bound uremic toxin. Gut microbiota produce indole from the breakdown of dietary tryptophan, which is then further metabolized to IS in the liver. The majority of IS circulates bound to albumin in the blood. It is excreted by the kidney tubules through organic anion transporters.
IS is one of the most studied uremic toxins. It has been associated with vascular calcifications and increased risk of death from heart disease in patients with advanced kidney disease. Higher levels of circulating IS have also been associated with faster progression of CKD.
para-Cresyl sulfate (pCS)
This is another protein-bound uremic toxin that is produced by the metabolism of amino acids tyrosine and phenylalanine by intestinal bacteria. This leads to the formation of p-cresol which is metabolized in the liver to pCS. pCS also circulates in the blood bound to albumin and is excreted by the kidneys in the same mechanism as IS.
Serum pCS levels increase with worsening kidney function. Similarly to IS, pCS was also found to predict the progression of kidney disease and is associated with an increased risk of death from heart disease in kidney disease patients.
Trimethylamine-N-oxide (TMAO)
TMAO is one of the small water-soluble URMs. It is produced by the metabolism of dietary L-carnitine and choline. The latter two compounds are metabolized into trimethylamine by gut bacteria, which is then absorbed by the gut and oxidized in the liver to form TMAO. The kidneys are the primary source for eliminating TMAO from the body. Dysbiosis and decreased kidney function have been documented to lead to elevated levels of TMAO.
TMAO levels increase with worsening kidney function and are associated with increased progression of kidney disease, increased risk of heart disease and all-cause mortality in kidney patients.
The L-carnitine controversy
L-carnitine is a relatively small water-soluble compound that is found abundantly in food sources including in red meat, dairy, poultry, and fish. Lysine, methionine, ascorbate, niacin, pyridoxine, and iron are among the major sources of endogenous carnitine production.
There is evidence that patients with kidney disease are deficient in L-carnitine and that supplementation leads to improved outcomes. However, there’s seemingly contradictory evidence that, the metabolism of L-carnitine by gut bacteria leads to the production of TMAO which, as described above, is associated with increased mortality in kidney patients.
One explanation for this discrepancy is that research on L-carnitine often fails to distinguish CKD patients who are not on dialysis with those on dialysis. In fact, one study showed L-carnitine levels are actually high in patients with CKD but are low in dialysis patients. Another reason for the confusion is that most research on L-carnitine supplementation uses intravenous L-carnitine which bypasses the gut microbiota and liver and, therefore, may not lead to the production of TMAO. Another important consideration is the composition of the microbiome and intestinal integrity of the gut. Those with certain dysbiotic markers might be at higher risk for producing TMAO and failing to break it down sufficiently, leading increased kidney injury.
The literature seems to support the need for restricting oral intake of dietary or supplement sources of L-carnitine in patients with kidney disease who are not on dialysis. This might also explain why CKD patients benefit from diets that are dairy-free and low in red meat.
Indole acetic acid (IAA)
IAA is yet another protein-bound uremic toxin that results from the metabolism of tryptophan by gut bacteria. Levels of IAA are proportional with CKD stage progression and normalize after kidney transplantation. IAA is linked to vascular inflammation and oxidative stress and has been associated with mortality and increased risk of death from heart disease in patients with CKD.
The role of diet
Dietary habits play an important role in gut microbiota composition. Diets high in animal protein are associated with increased dysbiosis and intestinal hyperpermeability and also contain a higher amount of amino acids precursors of uremic toxins. Meanwhile, whole-food and fiber-dense plant-based diets are associated with the growth of healthy gut microbiota, leading to decreased inflammation and better intestinal integrity.
Levels of protein intake have been studied in relation to CKD. Interestingly, it is not the amount but the type of protein that is associated with kidney disease. In fact, a large study of 12,000 adults with normal kidney function found no significant association between total protein intake and the incidence of CKD. The same study found that individuals consuming more red and processed meat were at higher risk for CKDcompared to those consuming more vegetable protein. Red meat intake has also been reported to increase the risk of worsening kidney function requiring dialysis.
There are various additional factors that should be considered as well, including consumption of processed carbohydrates, simple sugars, and reduced consumption of whole fruits, vegetables, and other fiber sources like whole grains. These nutrient-poor dietary habits can lead to dysbiosis which in turn produce inflammatory molecules that can lead to kidney disease. This can cause further accumulation of inflammatory uremic toxins.
Furthermore, the worsening of kidney function and accumulation of URMs can lead to further dysbiosis,and so on the cycle repeats. This is what we describe as the dysbiosis cycle in kidney disease.
To date, unfortunately, there are no studies that measure the effectiveness of dietary interventions on URM levels in kidney disease and subsequent prognosis. Studies focusing on the use of prebiotics and probiotics to improve gut health and reduce inflammation and CKD risk have had mixed results. However, a major weakness is that these studies only look at a single intervention that impacts kidney disease when in reality the process is much more complex. Future studies should take into consideration the complex interplay of the dietary and lifestyle factors on GI integrity, microbiome balance, genetics, and kidney outcomes.
The bottom line
There is a significant link between the microbiome, gut integrity, genetics, diet, lifestyle, and kidney disease. Most of this can be traced back to the accumulation of URMs in CKD causing a vicious cycle. The conventional approaches to improve gut health and slow the progression of CKD have yielded mixed results. Casting a wider net to include considerations for all factors that contribute to gut integrity, inflammation, and CKD risk must also be examined, including environmental exposures, genetic risk factors, metabolic factors and changes in body fluid volume, and overlapping factors which contribute to diabetes and hypertension, collectively contribute to increased CKD risk. Hence, we advocate for a comprehensive approach that starts with a gut restoration protocol and addresses the above factors in patients with CKD.
The post The Gut-Derived Uremic Toxins appeared first on Integrative Kidney.]]>Kidney stone formation (urolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. They are painful, inconvenient, and when left untreated, they may contribute to more serious conditions including obstruction and kidney damage.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Read more about the etiology and prevalence of kidney stones here.
In this series we’re building a case for a more integrative approach to preventing kidney stone formation.
Conventionally, the treatment approach does address kidney stones via a multi-pronged approach that may include medication, dietary and lifestyle, surgical removal, and using ultrasonic waves to break up stone.
However, these guidelines tend to focus too far downstream, on stone composition instead of on the underlying pathology upstream. Instead, we advocate for a more comprehensive approach that focuses on risk factors to prevent formation. Those factors include:
· Type of stone
· Socioeconomic factors
· Diet
· Hydration and electrolyte balance
· Microbiome and gut health
· Genetics
We covered individual dietary components in detail in a previous blog. Today we’ll look at the gut-kidney stone connection and the impact of the microbiome.
Gut Integrity and Kidney Stones: Leaky Gut
A normal and healthy GI tract has a natural barrier. This barrier serves to protect the GI and has three major jobs: 1. ensure proper digestion and absorption of nutrients and 2. ensure elimination of toxins and 3. protect the integrity of the microbiome – the “good” bacteria that lives in our GI tract and works with our body to maintain health.
Leaky gut describes a state when the cells that make up the lining of the GI tract separate enough to allow the contents of the gut to leak out. This is also sometimes called intestinal permeability or IP for short. This is a problem because it reduces absorption of nutrients, causes toxins to build up, alters the balance of the gut microbiome, and results in systemic inflammation.
One of the major contributors to leaky gut is the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
· Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
· Increased intake of processed/refined foods like cereals, crackers, baked goods, etc…
· Processed, fried, conventionally raised, high-nitrate animal protein
· Low intake of fiber and fresh produce in general
· A “beige” diet (low in phytonutrients and antioxidants) from consuming a variety of colorful fruits and vegetables
· Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
We have already established that eating more fresh produce, is protective from kidney stone formation, and we’ve done a deeper dive on specific nutrition impact on kidney stone risk in another blog if you’d like to learn more.
There are several factors that may contribute to development of leaky gut:
· “Proinflammatory” SAD: too much processed and high-sugar foods, not enough fiber and the wrong inflammatory fats
· Food sensitivities: consuming food that are cause reactivity
· Overconsumption of caffeine and alcohol – irritants to gut lining
· Use of certain medications, including NSIADs, steroids, antibiotics
· Stress and poor-quality sleep
We address risk factors for intestinal permeability in more detail in a previous blog here, as well as dive into a comprehensive gut restoration strategy here in this 5-part series.
The Microbiome and Kidney Stones
Balance of the gut bacteria also play an important role in causing or preventing kidney stones. The most studied organism is Oxalobacter formigenes, which has been found to be protective when present in adequate quantities as part of the GI microflora. This bacterium degrades oxalate in the gut decreasing its absorption and excretion in the urine.
When Oxalobacter was discovered, scientists thought they had pinpointed the key to curing kidney stones. They concluded that simply supplementing this missing species should reduce risk of stone formation in susceptible individuals. It would turn out that the connection wasn’t that simple.
More recent evidence points to a more complex picture in the connection between microbiome diversity and kidney stone pathology. The emerging research shows increased risk in kidney stone formation in certain susceptible individuals also presented with alterations in normal microbiome and metabolome (metabolic byproducts from microflora) – also termed dysbiosis.
In other words, it’s likely that genetic factors might be “turned on” by dysbiosis leading to increased risk of kidney stone formation in certain individuals. The good news is that means they should be “turned off” when the microbiome balance is restored.
Studies that looked at the use of targeted probiotics have failed to show enough significant improvement of risk of urolithiasis. Although there’s been some limited and temporary reduction in oxalate excretion and kidney stone formation with the use of a combination of Lactobacillus, Bifidobacterium, Enterococcus, it’s been shown to be temporary and limited in benefit. This is because dysbiosis cannot be addressed by simply applying a band aid of a probiotic.
We recommend instead a more comprehensive approach to gut restoration and microbiome balance. You can read more about the 5R protocol in our comprehensive 5-part series on gut restoration.
The Bottom Line
Although initial findings about the impact of the microflora that looked at Oxalobacter in isolation have not demonstrated significance in reducing incidence of kidney stone formation, more recent evidence pointing to an interplay of factors on microbiome diversity is promising. Furthermore, factors that impact kidney stone formation include dietary factors, including food quality, nutrient composition, and dehydration. Along with environmental factors, lifestyle, genetics, and gut integrity and microbiome balance should be addressed through a comprehensive and personalized approach. Practitioners working with individuals to prevent kidney stone formation should formulate a patient care plan that modifies all relevant components in their integrative approach to maximize effectiveness in preventing urolithiasis.
The post The Microbiome and Kidney Stone Formation appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
Recent studies have focused on the significance of a relationship between gastrointestinal (GI) integrity and microbiome diversity with various chronic diseases including kidney disease.
In previous blogs, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance leading to intestinal permeability (IP or “leaky gut”). The impact of leaky gut on kidney health and progression of chronic kidney disease (CKD) has been referred to as the gut-kidney connection is the result of complex biochemical and immune mechanisms.

A comprehensive approach to CKD includes addressing the health of the GI at the root of it. Furthermore, this means not simply supplementing with probiotics, but instead addressing all the mechanisms that underlie probiotic need. This includes modification of microbiota balance, integrity of the mucosa and epithelium of the GI tract, improving GI motility, absorption and digestion, and modulation of the immune system.
Below we will explore the fifth step, Rebalance. But first, let’s quickly review the first four steps of the comprehensive gut restoration protocol. A reminder that the 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD.
The 5 steps of the 5R protocol for healing leaky gut are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Rebalance
The goal of the fifth step is maintenance and prevention of recurrence of IP or leaky gut. There are various factors that lead to IP that involve dietary and lifestyle influences*, including:
· Standard American Diet (SAD) which is low in fiber, high in processed foods, and highly inflammatory
· Poor eating habits (for example, multitasking and not chewing adequately)
· Inadequate hydration and/or electrolyte imbalance
· Motility issues leading to constipation or unfavorable formation/frequency of stool
· Stress and poor sleep
· Not enough exercise
To reduce risk of CKD, we must work towards improving diet and lifestyle habits that support continued GI health.
*Read more about medications that impact gut health and ultimately increase risk for KD progression here.
Where to start?
Address lifestyle factors that impact gut health
Long term dietary goals focus on a plant-based diet that is high in fiber and a wide range of key nutrients. Organic sources of animal protein can beneficial when eaten in moderation. However, the key is to load up on naturally antiinflammatory, low carbohydrate vegetables to maximize vitamins, minerals, phytonutrients and, of course, fiber. Reduce intake of starchy vegetables and eat more**:
· Dark leafy greens (like spinach, arugula, and romaine)
· Cruciferous veggies (like broccoli, cauliflower, and kale)
· Fresh whole fruit (preferably lower sugar berries and avocados, and limit to two servings per day)
· Some colorful starchy veggies can be OK (for example, carrots, sweet potatoes, beets, and squashes)
Those veggies listed above promote a healthy microbiome and improve alkalinity associated with improved kidney health. In addition to those sources of prebiotic fiber, include probiotic sources like fermented vegetables and drinks like raw sauerkraut, kimchi, pickles, apple cider vinegar, and kombucha. It’s recommended these are eaten raw because pasteurization process will destroy the bacterial content of these foods.
In general, reduce processed carbs, like bread, cereals, and high sugar foods, desserts and pastries. These types of foods are usually low in fiber and nutrients and help contribute to starving of beneficial gut bacteria. Furthermore,high carb diets have been associated with increased risk of cardiovascular (CV) disease and diabetes. Instead, moderately consume whole grains like brown rice, quinoa, oatmeal, and legumes**.
Include antiinflammatory fats focusing primarily on omega 3 sources from fish, nuts, seeds, and mono- and polyunsaturated (MUFA and PUFA) like avocados and olive oil. Even though the topic of saturated fats is more controversial, recent evidence suggest that moderate intake of certain saturated fats like that found in beef, organ meat, or ghee (clarified butter) derived from grass-fed cows and virgin coconut oil might have health benefits. Everyone agrees, however, that trans fats(aka hydrogenated oils and artificial products like margarine) or excessive intake of processed and fried fats contributes to inflammation and increased risk of disease.
Ensuring adequate hydration, drinking at least half your body weight (pounds) in ounces of water, not only helps to maintain good kidney health, it also helps support daily regular bowel movements.
It’s well established that regular exercise can be beneficial for many reasons, including improved blood pressure, blood sugar, hypertension, stress relief, and even improved digestion and GI motility. In fact, exercise has been associated with improved microbiome balance as well as beneficial modulation of the immune system.
Poor sleep quality and stress are also deeply tied to many underlying factors impacting of GI health. Reduced sleep duration and quality has been associated with increased inflammatory markers (including TNF, IL-1, and IL-6) associated with GI disease like GERD and Irritable Bowel Disease/Syndrome (IBD/IBS) disrupting digestion and nutrient absorption. Furthermore, sleep has been shown to affect kidney health directly and indirectly, including associated risk of CV disease, diabetes, obesity, and hypertension(read more about the relationship between sleep and kidney health here).
**NOTE: Due to individual variations and progress of disease, work with a nutritionist to assess if you need to maintain any specific restrictions due to your unique case and needs.
The role of supplements
Because of the unique needs of kidney disease patients, many need to rely on supplements to help obtain adequate amounts of key nutrients to maintain GI and kidney health.
This may include GI and motility support including but not limited to digestive enzymes, bitters, probiotics, and magnesium citrate and triphala for motility). Furthermore certain individuals may benefit from supplementation of certain vitamins and important minerals, high potency antioxidants, and/or support of certain key underlying cellular mechanisms impacting mitochondrial health, detoxification and nitric oxide production.
That said, many factors must be taken into consideration when choosing appropriate supplements for each patient. Supplement quality and contamination are a common concern, as are potential interactions with medication or contraindications in certain commonly associated chronic disease. We suggest working under the care and guidance of a practitioner or team of providers who are trained in integrative and functional medicine and understand the unique needs of kidney patients.
Next steps
Unfortunately, addressing gut health is only the beginning. As mentioned above, kidney disease is often associated with multiple chronic diseases including CV disease, diabetes, high blood pressure, and obesity.
The comprehensive approach to kidney care means addressing the underlying causes of this constellation of diseases which is best accomplished by a Functional Medicine approach. The goal is to identify and then rebalance the biochemical and pathophysiological dysfunction at the root of chronic disease, we can stop the progression of kidney damage and preserve kidney function.
Bottom Line
In the final step in the 5R individualized gut restoration protocol, we Rebalance the foundational factors that impact the gut-kidney axis.
Although this might be the last step in the 5R protocol, within the broader context of kidney disease, it might signal the transition to a comprehensive therapeutic protocol that includes management of the underlying dysfunction associated with related conditions such as CV disease, diabetes, obesity, and hypertension.
Working with an integrative or functional medicine provider is essential to help you navigate the comprehensive program successfully, but can help you stabilize blood sugar, lower blood pressure, lose weight, and reduce the risk of CV and ultimately, KD.
More from InKidney on the gut-kidney connection:
· Comprehensive Gut Restoration Protocol https://old.inkidney.com/2019/07/05/comprehensive-gut-restoration-protocol-ckd/
· Feeding Gut Bacteria in Kidney Disease https://old.inkidney.com/2019/02/20/feeding-gut-bacteria-patients-kidney-disease/
· Kidney-Gut Axis: Nutrition can slow the progress of kidney disease https://old.inkidney.com/2018/09/20/kidney-gut-axis-nutrition-slow-kidney-disease/
· Inflammation, Leaky Gut And Kidney Disease https://old.inkidney.com/2018/10/05/inflammation-leaky-gut-kidney-disease/
· Leaky Gut And Kidney Disease: 6 Classes Of Medication That Might Be Contributing https://old.inkidney.com/2018/10/05/leaky-gut-kidney-disease-medications/
The post The 5R Protocol Part 5: Rebalance appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
The gut-kidney axis refers to the relationship between gut integrity and microbiome diversity with kidney disease. Excessive intestinal permeability, also known as hyperpermeability or more commonly as “leaky gut,” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
So far we looked at the first three steps of the 5R protocol, Remove , Replace, and Reinoculate. Applied sometimes sequentially and at times simultaneously, these steps are used to address the underlying factors associated with leaky gut. The idea is that comprehensive approach that reveres damage to the gut caused by exposure to food and environmental triggers, addresses the disruption of digestion and nutrient absorption, altered bowel motility, and dysbiosis, improves gut health and ultimately overall kidney health.
Read on below where we will explore step 4, Repair. But first, let’s first review the five steps of the comprehensive gut restoration protocol. A reminder that the 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD and in our upcoming blog on Rebalance, we will explore maintenance and next step in integrative kidney care.
The 5 steps of healing leaky gut are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Gut Repair
Up until this phase, we’ve focused on removing triggers that contribute to local and systemic inflammation. We’ve even taken steps to rebalance the microbiome. In the repair phase, we work to provide nutritional support that directly impacts the integrity of the gut mucosa and repairs hyperpermeability.
Recall from our previous blog, Part 1: Remove, that exposure to toxins, food sensitivity, and presence of pathogens leads to increased inflammation locally that triggers the immune system and leads to damage to the lining of the gut and mucosa.
The term Intestinal hyperpermeability (aka “leaky gut”) is the result of this inflammatory assault to the gut lining. These breaks in the integrity of the wall (imagine gaps in a fence on your lawn) let undigested food, bacteria, and metabolites “leak” through the holes.
The physiologic changes associated with this leaky state include a combination of factors that reduce your gut’s ability to absorb nutrients. These include hypochlorhydria (insufficient hydrochloric acid in the stomach to digest food), reduced production of digestive enzymes, altered bowel motility (often leading to constipation, but not always), and dysbiosis.
Now that we’ve addressed those aspects in steps 1-3, we can focus our energy on repair. This stage is often several weeks into the comprehensive gut restoration program because initiating it simultaneously may interfere with the efficacy of the prior steps. Working with an integrative or functional medicine practitioner can help guide you through your personalized program.
Where to start?
Address permeability with nutrients and herbs
To support intestinal mucosa regrowth and cell repair we focus on nutrients that have properties that promote rebuilding a healthy mucosal lining. They help improve the integrity of the intestinal wall by supporting building and formation of the intestinal epithelium, villi and cell connective tissue.
This includes a wide spectrum of micronutrients including vitamin A, D, E, and C. L-glutamine, butyric acid, and collagen supplements. These are useful to build collagen that forms the epithelium of the GI tract. Whey, colostrum or serum bovine immunoglobulins may be utilized to balance inflammatory mediators based in the gut. Zinc carnosine, melatonin, cabbage juice, aloe vera, and mucilaginous herbs like marshmallow root and slippery elm are also used therapeutically to support various aspects of the rebuilding process.
This is of course layered in on top of an anti-inflammatory, nutrient-dense, fiber-rich diet that includes healthy fats, moderate intake of animal protein, and a colorful variety of organic fruits and vegetables that provide added antioxidant and phytonutrient repair support.
Bottom Line
The fourth step in an individualized comprehensive gut restoration protocol involves leveraging food and herbs to promote the repair of the gut mucosa. This is often done after Remove, but might be simultaneous to Replace and Repair steps. However, every case is unique, and it’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this safely and successfully. Next, we will tackle the 5th “R” in the gut restoration protocol: Rebalance.
The post The 5R Protocol Part 4: Repair appeared first on Integrative Kidney.]]>
The Immune Response: The Firefighters and Sharpshooters
Before we dive into autoimmune kidney diseases, let’s review the basics of the immune system. The immune response can be generally be divided into two arms: innate and adaptive immunity.
Innate Immune response, sometimes referred to as the “primitive” arm, refers to a nonspecific defense mechanism. When a foreign invader, called an antigen, the innate system responds immediately.
The “first responders” of cells that make up the innate system tend to be concentrated around the physical barriers of the body including the skin, gastrointestinal tract, the airways of the lungs. The response is fast and fairly effective, but it’s not very specific so there tends to be some collateral damage. Think of this as the firefighters that work to extinguish a house fire without any regards to the water damage that might impact the house.
Adaptive immune response, on the other hand, is a more targeted immune response. In exchange, this response is more complex than the innate response and takes more time to mount. The antigen’s unique identifying markers is first processed, and the details are committed to memory. When the antigen matches a memory in the database, it’s recognized, and the adaptive immune response triggers a focused immune cells meant to attack and destroy it. This type of adaptive “memory” makes future responses that specific antigen more efficient. You can think of this as a sharpshooter, it takes time, training, and experience, but it’s effective and there’s typically little collateral damage as a result. That said, any problem with the cascade can result in misfiring that turns against the wrong target, as is the case of autoimmune disease.
The GALT
The mucosa of the GI tract has a built-in immune system. In fact, the gut is the largest immune organ in the whole body. This system is called gut-associated lymphoid tissue (GALT) and it is in constant interaction with the intestinal microbiome. The balance of the microbiome is an essential part of managing the GALT, and dysbiosis can trigger a complex immune response involving multiple pathways.
One particular pathway engages what is called the toll-like receptor (TLR) family. These receptors are basically proteins that span the cellular membrane of the many immune cells in the gut and act like scanners. In humans, there are 11 members of this receptor family. Of particular interest, TLR-4 and TLR-2 play important roles and are present on the mucosal epithelial cells of the gut.
These receptors are capable of detecting the unique molecular patterns present on the surface of microbes that are presented when there’s intestinal hyperpermeability (leaky gut). TLR-4, for example, can be activated by lipopolysaccharides (LPS) which are released by certain gram-negative bacteria such as Escherichia Coli (E. Coli). TLR-2 receptors are activated by lipoteichoic acid of gram-positive bacteria and yeast.
So, in essence, the gut has an intricate mechanism to detect pathogens and stimulate the necessary response. At the same time, friendly flora also communicates via the GALT and helps to regulate and improve immune response.
The Inflammatory Response
In the presence of LPS, TLR-4 is activated by binding with their receptors on the surface of the cell (CD14). This triggers a cascade of signals inside the cells (including NF-κB, MAPK, and others). This response leads to the formation of inflammatory molecules broadly called cytokines such as tumor necrosis factor-α (TNF-α) and interleukin among others.
In other words, the domino sequence is triggered when the immune system recognizes a pattern it associates with a pathogen and signals to the immune system to take action. Leaky gut increases the odds of immune response because it increases the exposure of the antigen to the immune receptors. This demonstrates an important concept, the overlap between the innate and adaptive immune system. Hyperpermeability of the gut barrier is often a result of “collateral damage” from non-specific innate response to antigens in the GI (food allergies/sensitivities, toxins, and pathogenic bacteria, yeast, or viruses) which then goes on to train and trigger the more sophisticated adaptive response.
Furthermore, genetics play a role in the likelihood of this complex system over or under performing. Minor genetic code alterations such as single nucleotide polymorphisms (SNPs) in protein presentation of various components (including CD14 and TLR-4) can be associated with an increased risk of immune misfiring and increasing incidence of systemic inflammation, insulin resistance, and autoimmune disorders. Basically, genetics load the gun, the environmental influences pull the trigger.
The Autoimmune Response & the Kidney
Keep in mind, TLR-4 are present not only in the gut but also in the systemic immune system, including the brain and the kidneys. In fact, the signaling pathways between the gut microbiome and the kidneys have been well-documented. The activation of this gut-kidney cascade has been critical in the development of many autoimmune disorders such as antineutrophil cytoplasmic antibody associated vasculitis such as Wegner’s granulomatosis. Furthermore, the activation of toll-like receptors has also been associated in systemic lupus erythematosus (SLE) and IgA nephropathy.
The Bottom Line on the Gut-Kidney Connection and Autoimmune Kidney Disease
The gut houses the GALT, an intricate mechanism necessary to identify and eradicate pathogens. In the presence of pathogenic invaders, an inflammatory response is triggered like a cascade of dominos. When triggered, this response in the presence of certain genetic predisposition can be the perfect storm that leads to various kidney-related immune diseases including vasculitis, lupus, and IgA nephropathy.
This is why when we consider the comprehensive approach to kidney health, it is important to address leaky gut and dysbiosis by employing a comprehensive gut restoration protocol in order to manage the progression of autoimmune kidney disorders and ultimately preserve kidney function.
The post The Gut-Kidney Connection and Autoimmune Kidney Diseases appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
In this series we examine the comprehensive gut restoration protocol used to as a foundational approach to heal gut integrity. Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
In our previous blogs, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance leading to intestinal permeability (aka “leaky gut”).
So far we have looked at how the first and second steps of the 5R protocol, Remove and Replace, help to address the underlying factors associated with leaky gut that include a combination of factors like exposure to food and environmental triggers causing local and systemic inflammation, disruption of digestion and nutrient absorption, altered bowel motility, and dysbiosis.
Below, we will explore step 3, Reinoculate. Let’s first review the five steps of the comprehensive gut restoration protocol. The 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD. The five areas of GI mucosal integrity are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Reinoculate
The basic premise of the third stage of gut restoration is to foster an environment that allows beneficial microflora (aka good bacteria) to thrive in the gastrointestinal (GI) tract. This is achieved by leveraging diet, supplementation, and lifestyle modification.
In previous blogs, we have explored the connection between dysbiosis and kidney health. An increasing number of studies have demonstrated a significant relationship between the health of the microbiome and the progression of kidney disease, a relationship referred to as gut-kidney axis.
Those with a healthy microbiome, abundance of good bacteria and no overgrowth of bad bacteria, are less likely to develop chronic kidney disease. In fact, the presence of certain strains of bacteria in the gut can actually slow the progression of chronic kidney disease and even reduce the need for dialysis.
Where to start?
Prebiotics (aka Fiber)
Most Americans fall significantly short of the recommended fiber intake of 30+ g/day. High fiber intake is associated with reducing the risk of heart disease, obesity, diabetes, and even certain kinds of cancers like colorectal cancer. These benefits are linked to to improved bowel movements, neutralizing and removing toxins, and “feeding” gut bacteria contributing to a favorable microbiome balance.
Research even suggests that dietary fiber-intake may be one of the most significant predictors not only of gut health, but overall health and risk of disease!
Remember to always choose whole, fresh, fiber-rich fruits and veggies whenever possible to maximize nutrients and prebiotics simultaneously. Whole grains and legumes make good fiber sources as well, but always avoid processed foods that claim to have “added fiber”. They are usually packed full of fillers, sugars, grains, cereals and artificial ingredients.
High-sugar diets can be a major disrupter of your gut microbiome, primarily because it feeds bad bacteria and yeast overgrowth. This is one of the proposed mechanisms contributing to metabolic diseases like diabetes and heart disease, two conditions associated with KD.
Prebiotic supplementation might also be necessary in some cases. Supplemental powders and capsules of resistant starch, arabinogalactan, and mastic gum, among others, might be useful in many situations. Your integrative or functional medicine provider can help you determine which, if any, are appropriate for your unique situation.
Probiotics
Supplementation with high intensity probiotics may be very useful in the Reinoculation phase of the 5R protocol. There are a variety of strains of bacteria and even beneficial yeast that are used to help to “seed” the gut (though technically more recent studies suggest the benefit is transient, it can still be helpful). Depending on your individual needs, your integrative and functional medicine provider may choose a particular strain or opt for a broad-spectrum formulation. Either way, the quality and potency of the probiotic is important to consider (not all brands are created equal).
That said, research seems to support that the best long-term strategy is to increase the intake of probiotics coupled with prebiotic fiber intake through diet. Traditionally fermented foods are a great source of naturally found probiotics, these include non-pasteurized traditionally made kefir, sauerkraut, kimchi, miso, and pickled vegetables.
Other factors impacting the microbiome
Interestingly, exercise impacts microbiome balance and promote changes that improve gut health. Aim for at least 20 minutes of exercise daily at a level appropriate to your physical fitness and make it priority to get up and move – you and your kidneys are worth it!
Furthermore, stress has been found to negatively alter the balance of the microbiome by reducing the presence of friendly flora and promote the growth of bad bacteria. Although we know that stress cannot always be avoided, everyone can find a stress management practice to reduce its negative impact. Breathing exercises, meditation, long walks, listening to music, adult coloring books – the options are endless. Find what works best for you!
Bottom Line
The third step in an individualized comprehensive gut restoration protocol involves promoting microbiome balance. This is often done after the Remove step but might be simultaneous to the Replace step. Sequencing the steps of the protocol is case-by-case dependent. It’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this successfully.
Next, we will tackle the fourth “R” in the gut restoration protocol: Repair.
The post The 5R Protocol Part 3: Reinoculate appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms. Here we discuss the Replace step of the 5R protocol for kidney health.
In our previous blog, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance. Intestinal permeability (aka “leaky gut”) is the result of inflammatory assault to the gut lining that results in breaks in the integrity of the wall. Imagine it like breaks in a “fence” that let undigested food, bacteria, and metabolites “leak” through the holes.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The physiologic changes associated with this leaky state include a combination of factors including hypochlorhydria (insufficient stomach acid to break down food adequately), reduced production of digestive enzymes, altered bowel motility (often leading to constipation, but not always), and dysbiosis.
As a result of these pathologies, leaky gut leads to increased local and systemic inflammation, poor digestion, reduced nutrient absorption, and reduced ability to remove toxins and digestive byproducts.
Though we address step two, Replace in more detail below, let’s review the five steps of the comprehensive gut restoration protocol. The 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD. The five areas of GI mucosal integrity:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate to provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Replace
Due to diet quality, food sensitivities, pathogenic microbes, epithelial inflammation leads to damage to the gut mucosa integrity. This causes damage to the structure of the villi and reduction in the intestinal surface area leading to reduced absorption of nutrients. This is referred to as malabsorption, or poor absorption of nutrients across the gut wall into circulation to be used by the body.
A second pathology is maldigestion, or ineffective breakdown of nutrients from food. This might be one or more of the major macronutrients, fat, protein, and/or carbohydrates. In these cases, you’ll notice GI distress after eating, bloating, distention, and or excessive flatulence. This not only reduces the effective absorption of essential nutrients necessary for healthy metabolism, but also creates a backup that contributes to increased inflammation, buildup of endotoxins, and further contributing to dysbiosis.
Furthermore, poor motility can contribute to chronic constipation. It’s been estimated that 20% of the adult population suffers from chronic constipation – that’s 1 in 5 adults! Ideally bowel movements should be daily, easy to pass and as characterized by the Bristol Stool Scale below, smooth soft and formed.
Constipation is not only uncomfortable, but leads to inefficient removal of toxins, recirculation of endogenous hormones, and alteration of the microbiome balance. Furthermore, stool consistency and frequency can be used as clues for clinicians signaling the need for a gut restoration protocol. From there, integrative and functionally trained practitioners can use advanced stool testing that employs a technique called PCR technology as discussed in Part 1 Remove blog.
Where to start?
Assess Nutrient status
Often, we begin by assessing the baseline nutrition status for any micro- or macronutrient deficiencies. These tend to be more significant with long-term maldigestion and malabsorption, depriving the body of necessary vitamins, minerals, amino acids, and essential fats needed for healthy function. Supplementation is often necessary, but dietary adjustments can be made to optimize absorption from food as long as digestion and motility are also addressed (see below).
Assessment can be done by physical exam to detect signs and symptoms of nutrient deficiency – for example dry hair and skin, brittle nails, bleeding gums, and difficulty digesting animal protein or fat can all be indications of various nutrient malabsorption/depletion or maldigestion.
Furthermore, laboratory assessment of nutrient status can confirm specific deficiency and help us target key nutrients strategically to customize the therapeutic approach.
Address Maldigestion and Malabsorption
To enhance digestion, use of broad-spectrum digestive enzymes such as lipase and bile salts, proteases, and cellulases that break down fats, proteins and carbohydrates, respectively is necessary because the digestive tract isn’t producing sufficient enzymes to breakdown and absorb nutrients form food. In addition, betaine HCL, a digestive aid that address low hydrochloric acid (hypochlorhydria) in the stomach may also be necessary to improve gastric pH.
Herbs and foods can also be useful for improving nutrient absorption and motility. For example, pineapple, papaya, and bitter foods like dandelion greens can be helpful for aiding digestion, stimulating motility and digestive enzyme production. Eating beets and staying hydrated promotes bile salt flow. Fermented and fiber-rich foods can also help promote motility and healthy microbial balance (though sometimes we don’t introduce these until step 3: Reinoculate). Last but not least, traditional concoctions of herbal bitters and teas can be very helpful in promoting this process.
Address Motility
We cannot emphasize enough how important it is to have at least one daily bowel movement for overall health. That is why integrative and functional practitioners will often prioritize resolving constipation. Bulk laxatives, stool softeners, and stimulants can be useful as a short-term strategy but are only a band-aid. It’s important to understand the underlying causes and address it: slow motility and dysbiosis.
Restoring stool frequency and consistency to ideal can be a challenging and time-consuming process with involves multiple aspects of the 5R protocol. Useful tools include supplements like magnesium citrate, triphala, Swedish bitters, electrolyte replacement and hydration, increasing fiber (dietary and supplemental) as well as stimulating the migrating motor complex and balancing the HPA axis (stress response) through lifestyle modifications including exercise, massage, gargling and stress management.
Bottom Line
The second step in an individualized comprehensive gut restoration protocol involves promoting improved nutrient status and motility by exogenously replacing the necessary enzymes and nutrients needed. This is often done simultaneously to the Remove step, but in some cases, it’s addressed in sequence. It’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this successfully.
Next, we will tackle the third “R” in the gut restoration protocol: Reinoculate.
The post The 5R Protocol Part 2: Replace appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
Past studies have attempted to explore the impact of dietary changes shifting the gut microbiome to help restore the lining of the gut and reduce the resulting inflammation. However, many of the studies in the literature looking at the use of probiotics to reduce uremic load show some limited benefit in reducing the chronic kidney disease (CKD) burden.
But these studies failed to present a comprehensive approach that reveres damage to the gut while simultaneously inoculating the necessary bacteria. This single dimensional approach does not acknowledge all the different factors involved in the gut-kidney Axis.
To assure that the patient is getting a comprehensive gut restoration protocol, all the mechanisms that underlie probiotic use should be addressed. These include modification of microbiota, competitive adherence to the mucosa and epithelium, strengthening of gut barrier and modulation of the immune system.
The 5R gut restoration program addresses these gaps and help reduce the risk of progression of CKD.
This program is designed to address five areas of GI mucosal integrity repair:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Remove
The first step of this protocol focuses on removing any exposures that may be contributing to increase inflammation in the gut. This includes food exposures, toxins, as well as screening for and treating pathogenic bacteria, fungi, parasites, or viruses that maybe disrupting the normal microbiota balance, which may require antimicrobial treatment to eradicate.
First, food known to cause sensitivities and allergies should be eliminated as part of an elimination diet. There are multiple categories of foods that contribute to inflammatory response that disrupts the lining of the gut. For example, gluten has been found to be associated with the development of leaky gut and IgA nephropathy. At the same time, we should be emphasizing the inclusion of key nutrient-dense foods that help to restore gut integrity and reduce inflammation, including antiinflammatory fats, organic fiber and phytonutrient-rich vegetables and fruit.
Typically, an elimination diet removes common food triggers like gluten, dairy, eggs, and soy. Depending on the root of the food reactivity, sometimes the removal or reduction of grains, legumes, FODMAPS, or night-shade vegetables is also necessary.
A nutritionist or clinician trained in implementing the elimination diet can help guide on which foods to eliminate or reduce. The decision may be done empirically, using symptom monitoring to guide the progress. Alternatively, the program can be personalized by using specialized testing for immune response against food can help guide this process.
Second, exposure to environmentally derived toxic substances should also be minimized, this includes mercury, arsenic as well as pesticides and other environmental pollutants whenever possible. We are exposed to toxic chemicals on a daily basis including pesticides in conventionally-farmed food, non-stick cookware, plastic use, and flame retardants and off-gassing released from furniture.
Addressing this aspect involves focusing on consuming organic produce and animal products and addressing environmental sources of toxicity. This topic is covered in more detail in this blog here.
Lastly, remove also entail identifying potential microbial triggers that might be contributing to inflammatory response. As mentioned above this may be “bad” bacteria, viruses, candida, and/or parasites. The presence of these offenders can be detected through symptoms as well as through advanced stool testing that employs a technique called PCR. Once identified, your practitioner can use antimicrobials to help eradicate the pathogenic organism, using prescriptive antibiotics or even herbal antimicrobials if appropriate.
Where to start?
Inflammatory foods
Some foods can be inflammatory or induce allergic reactivity. Furthermore, they may provide an environment that allows for the growth of pathogenic microbes, including low-fiber diets and high sugar diets. Furthermore, depending on your genetics, some foods can even lead to autoimmune conditions that may affect the kidneys. Eliminating food that is known to cause sensitivities varies dependent on the individual and their genetic predisposition.
An elimination diet can help with identifying what food an individual should avoid. This diet involves the removal of foods commonly associated with food sensitivities or immune reactivity. There are many variations of the elimination diet, and it’s very important to work with a nutritionist who can ensure you’re doing the protocol correctly and not missing any essential nutrients.
After a period of removal lasting at least 4 weeks, and assuming improvement in symptoms that suggests improved gut integrity, your integrative or functional medicine provider will work with you on gradual and careful reintroduction of foods to assess if tolerance has improved.
Decrease/Eliminate Exposure to Toxins
It is very hard to eliminate all sources of toxin exposure, but these steps can help minimize it:
1. Water filtration: we discussed options for water filtration in a previous blog.
2. Cookware: gradually eliminate all toxic non-stick cookware from your kitchen.
3. Avoid plastic containers or utensils: gradually switch from plastic to non-plastic containers and utensils.
4. Eat organic when possible: we know that choosing organic food can be expensive. If you are on a budget, there are certain fruits and vegetables that you should buy organic because of their high toxic burden. Visit the Environmental Working Group website to learn about their “Dirty dozen” T. This list gets updated yearly depending on their tests.
5. Be careful of processed drinks and juices. Not only do these contain excessive amounts of sugar, but they can also be a source of heavy metals and toxins because of poor regulations.
Eliminate Pathogens
As discussed above, laboratory analysis of the patient’s stool and serum can help give insight to the extent of intestinal permeability (leaky gut), as well as the status of microbiota. Establishment of baseline status is essential for measuring outcomes at designated milestones to give quantifiable outcomes. A comprehensive stool analysis can be a good start. In addition, a good dietary intake can help identify issues that could be related to food intake to guide this process.
Depending on the results of the individual’s comprehensive stool analysis, certain bacterial or yeast overgrowth can be detected. Also, parasites can sometimes be identified. Any of these should be treated by various medications, dietary changes, and/or herbal supplements as indicated and advised by an integrative or functional medicine provider.
Remove stress
Stress can have a bad influence on digestion and absorption. People who are stressed tend to eat too fast, make poor choices, and may eat too much at various intervals. This can lead to food choices that can lead to feeding the bad bacteria in the gut and poor digestion causing nutritional deficiencies. Furthermore, stress itself can influence epigenetic changes that impact dysbiosis as well as disease expression in general.
Bottom Line
The first step in an individualized comprehensive gut restoration protocol involves the removal of food sensitivities, environmental toxins, pathogenic organisms, stress. It’s important to work with an integrative or functional medicine provider trained to guide patients with elimination diets can help you navigate this successfully.
Next, we will tackle the second “R” in the gut restoration protocol: Replace.
The post The 5R Protocol Part 1: Remove appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Chronic kidney disease affects millions of American and is often associated with comorbid conditions such as cardiovascular disease, diabetes and hypertension. Dysbiosis and leaky gut are implicated in many systemic inflammatory and immune-related factors that lead to chronic disease. Dysbiosis is the term used to define alterations in the gut microbiota that includes overgrowth of bad bacteria, as well as underrepresentation of good bacteria. The normal integrity of the gut can be compromised resulting in systemic complaints, even in the absence of overt gut symptoms.
Research has established a relationship between gut microbes and different diseases, including kidney disease. This occurs through very complex biochemical and immune mechanisms. The presence of good bacteria has been associated with immune changes that inhibits inflammation. The use of probiotics and prebiotics that produce favorable changes in the microbiome have also been associated with gut changes that reduce advanced glycation end products (AGE), a uremic toxin associated with advancing kidney damage.
So, balancing the gut bacteria help slow kidney disease by directly inhibiting the immune response; and indirectly by reducing toxic burden known to progress CKD.
The Gut as a Potential Source of Inflammation
In the cases of dysbiosis, we see a rise in inflammatory markers that interact with the lining of the gut and result in damage and increased permeability or leaky gut. This causes shifts in the breakdown of nutrients, including amino acids. These shifts increase the levels of circulating substances involved in kidney disease and inflammation such as p-cresol, phenol, and indole. On top of that, these “pro-inflammatory” changes has also been associated with hypertension and increased risk of diabetes and cardiovascular disease; all of which are associated with CKD risk.
Yet, there have been inconsistencies in studies evaluating the benefits of targeting the gut-kidney axis with probiotics and prebiotics. Some suggest this may in part be due to the fact that research in this field is still in its infancy stage and conflicts in research may be due to statistical problems or inadequate interventions. But in an attempt to isolate the intervention, most studies utilized unilateral approach of inoculating the microbiome. In doing so, they usually fail to address other aspects of gut restoration and mucosal repair.
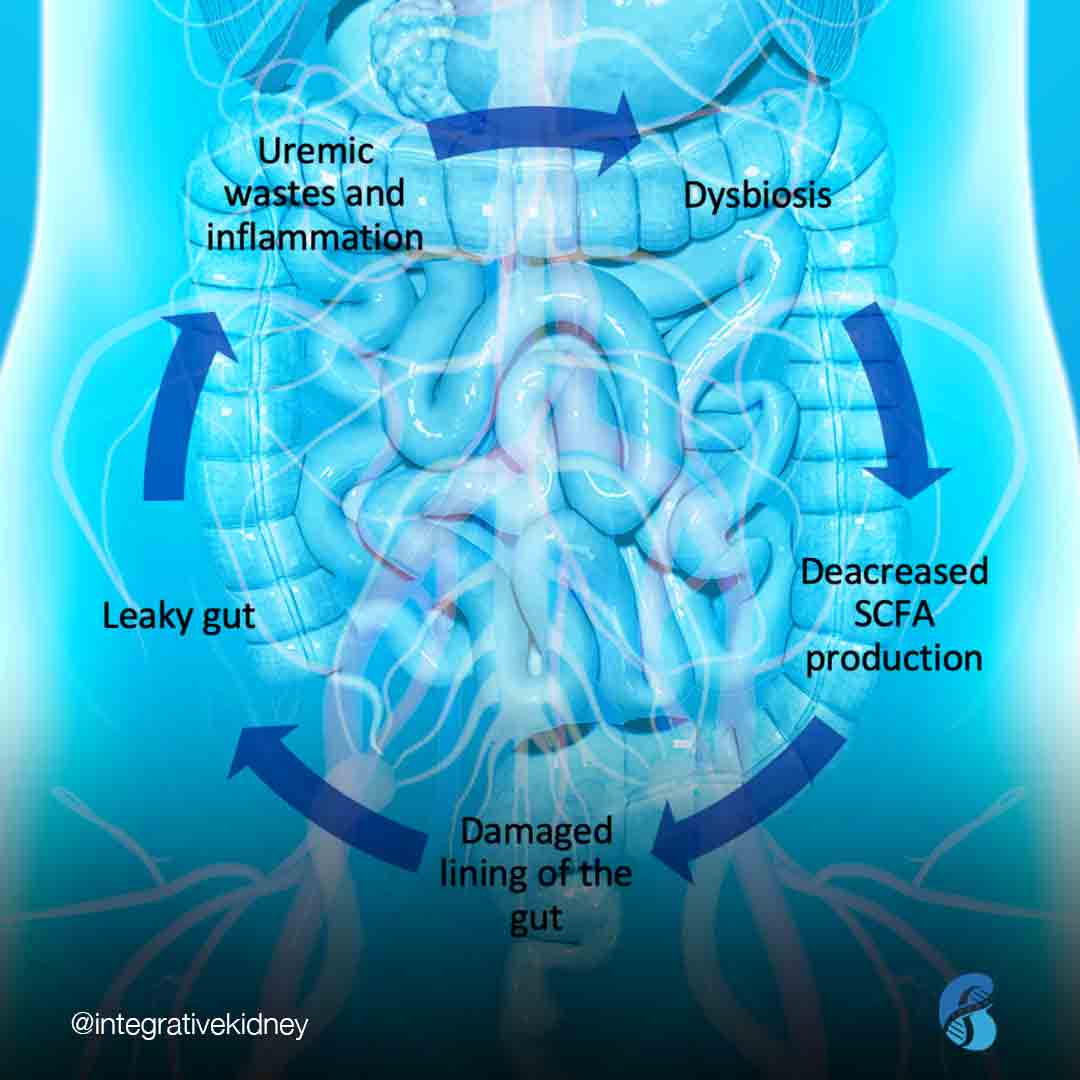
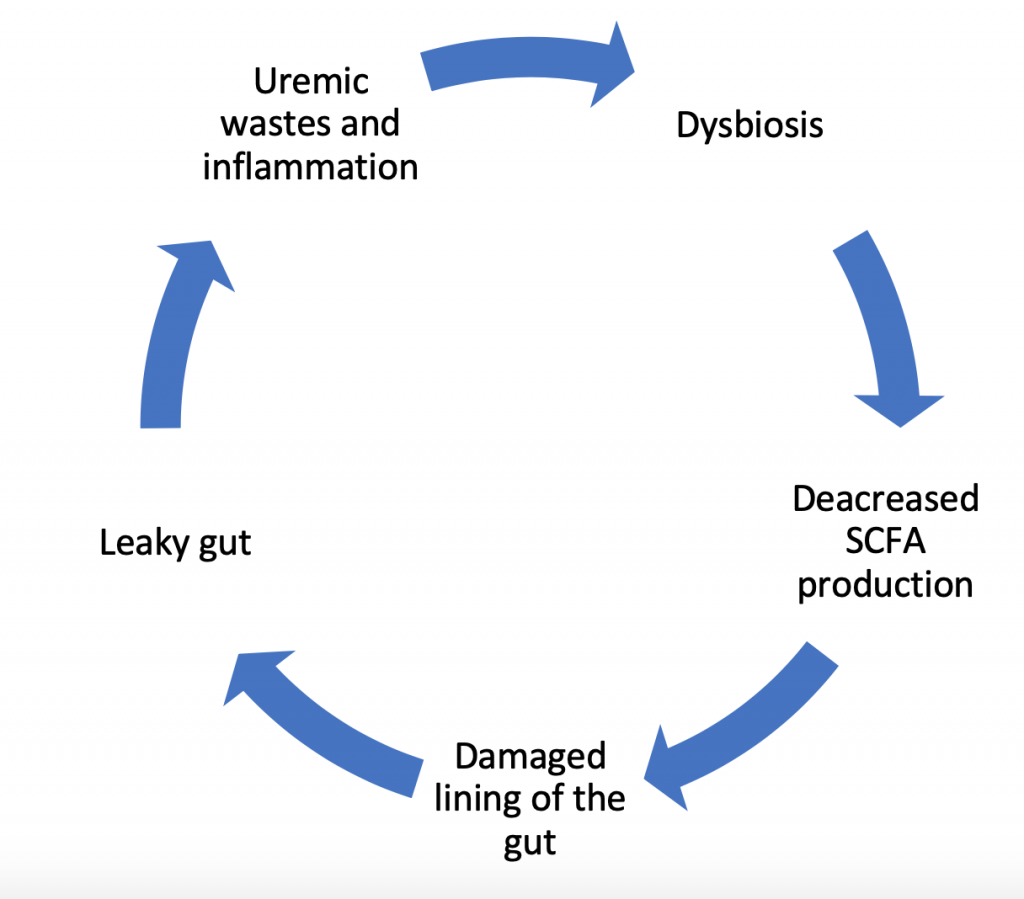
The Dysbiosis Cycle
In fact, changes in the gut bacteria do not operate as a one-way inflammatory force ending in damage to the lining of the gut. Instead it’s a dynamic process with a variety of contributing factors. Research confirms that it’s not only the dysbiosis that causes inflammatory damage, but also damage to the lining of the gut contributes to further dysbiosis and reduced production of short chain fatty acids (which feeds the lining of the gut). This, in-turn, increases the uremic waste products and further perpetuates growth of bad bacteria. This dysbiosis cycle damages the gut cell wall and promotes leakage of parts of the bad bacteria and toxins produced by these bacteria into the bloodstream. This leads to more inflammation, and the cycle repeats.
What is Missing in Current Research?
Individual Factors
As mentioned above, studies focusing on the use of probiotics to improve gut health and reduce inflammation and CKD risk have had mixed results. But these studies looked at a single change to see if it impacts kidney disease when in reality the process is more complex. Considerations for other factors that contribute to inflammation and CKD risk must also be accounted for. This includes environmental exposures, genetic risk factors, metabolic factors and changes in the body fluid volume that contribute to diabetes and hypertension, respectively, contributing to increased CKD risk.
Medications
On top of that, the effect of medication and polypharmacy on the gut and nutrient balance cannot be forgotten. Drugs commonly prescribed in kidney patients, like NSAIDs, Proton-pump inhibitors (PPI) or steroids, may also contribute to leaky gut. Changes in digestion due to some of these medications also contribute to altered nutrient absorption, including malabsorption of macronutrients, as well as micronutrients needed as cofactors in many biochemical reactions in the body.
Nutrients
The gut also can also serve as a source of increased oxidative stress, contributing to increased systemic inflammation that accelerates CKD and its comorbid conditions, including cardiovascular disease. Research suggests that one mechanism by which toxins generated by bacterial load speed up CKD progression is by altering metabolism and absorption of normally occurring nutrients. This may indicate that nutrient repletion maybe a necessary step for successful outcomes with probiotic re-inoculation.
This is why the use of an individualized comprehensive gut restoration protocol, that steps outside of the conventional linear model and takes into account the various layers described above can slow the progression of kidney disease. One such approach can be summarized by the 5R program which we will explain in details in the upcoming blogs.
The post Comprehensive Gut Restoration and Progression of CKD appeared first on Integrative Kidney.]]>





