
What is CoQ10?
CoQ10 is an essential part of the electron transport chain in the mitochondria. It is, therefore, crucial for energy production in the cells. It is abundant in the most metabolically active tissues, which include the heart, kidney, and liver. In fact, the concentration of CoQ10 in the kidneys is second only to the amount found in heart muscle. Deficiency in CoQ10 leads to insufficient electron transport, which leads to increased formation of reactive oxygen species (aka, oxidative stress). The body needs a certain amount of oxidative stress, but too much of it is harmful and is caused by an imbalance between free radicals and antioxidants. There are many other functions for CoQ10, but we will not have room to discuss them here.
CoQ10 and Kidneys: Why the Kidneys Need CoQ10
The kidneys filter waste products from the blood in two stages. The first involves the straining of a large amount of fluid through tiny blood vessels, called glomeruli. However, if the body does not reclaim most of that fluid (known as filtrate), the person will die of dehydration within a few hours. This is why the second stage is so important. In it, millions of tiny tubes in the kidney, called tubules, reclaim most of that filtrate.
About 90% of this retrieval process occurs in the first part of the kidney tubule, which is called the proximal tubule. The proximal tubules are also the site of excretion of many organic substances, medications, and uremic toxins. This process of retrieval and excretion by kidney cells requires a significant amount of energy, or ATP (adenosine triphosphate). This explains why the proximal tubules are so rich in mitochondria and CoQ10. Mitochondria are the powerhouses of the cells, and they manufacture the body’s energy currency, ATP.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
CoQ10 and Kidney Disease
A number of studies investigated CoQ10 and chronic kidney disease (CKD). In a small study of CoQ10 and kidney health from Comenius University Medical Center in Slovakia, plasma concentrations of CoQ10 were found to be decreased in patients with kidney disease. Despite that there was an obvious decrease in CoQ10 levels with decreasing kidney function in that study, it did not reach statistical significance due to the small number of patients tested. The author concluded that oxidative stress occurs very early in kidney disease. Decreased CoQ10 levels in CKD were also confirmed in another study. It also appears that the presence of hypertension worsens CoQ10 deficiency in CKD. In addition, our ability to produce CoQ10 internally decreases with aging.
Medications that deplete CoQ10
Several medications can lower the levels of CoQ10 in the body and these include:
- Statin drugs for cholesterol
- Fibric acid drugs for cholesterol
- Beta-blockers for high blood pressure
- Anti-depressant drugs such as amitriptyline
- Warfarin, a blood thinner
- Certain chemotherapy and HIV medications
Can CoQ10 Improve Kidney Function?
As mentioned above, the kidneys require a lot of energy production. The tubules are very sensitive to oxidative stress, which is common in kidney disease. Add to that the deficiency of CoQ10 that is noted in kidney disease. This makes CoQ10 a very attractive supplement to improve energy production and decrease oxidative stress in the kidneys.
There have been many studies evaluating the role of CoQ10 in CKD. CoQ10 was found to reduce total and LDL cholesterol in kidney patients. It was also found to decrease oxidative stress (by lowering malondialdehyde, a marker of oxidative stress).
Additionally, CoQ10 was found to improve blood pressure control. A CoQ10 dose of 200 mg daily was found to decrease systolic blood pressure by 11 mmHg and diastolic blood pressure by 7 mmHg. This was thought to be due to its improvement in nitric oxide balance leading to decreased constriction of blood vessels.
In addition, supplementation with CoQ10 has been found to improve endothelial function in patients with type 2 diabetes mellitus. Endothelial function refers to the function of cells lining the blood vessels. CoQ10 also improves cardiovascular outcomes in patients with or without kidney disease.
Finally, a CoQ10 supplement was even found to improve serum creatinine levels, which are a marker of kidney function. As kidney disease progresses, blood creatinine levels rise.
These studies show that the answer to the question “Is CoQ10 safe for kidneys?” is a resounding yes!
What is the Best CoQ10 Dosage for Kidney Health?
Studies that looked at CoQ10 dosage established its safety and tolerability in the normal population. Doses as high as 3,000 mg per day did not produce serious side effects in normal subjects. Gastrointestinal side effects were the most common. That means CoQ10 is generally safe for healthy people, even at unusually high doses.
In kidney disease, the doses that were studied ranged from 100-300 mg daily. It is, therefore, reasonable to target a dose of 200-300 mg daily split into three doses (100 mg three times a day). Since CoQ10 is fat-soluble, it is better absorbed along with the intake of fatty foods. As always, we recommend choosing high-quality supplements.
Serum CoQ10 levels are widely available and can be measured as part of routine bloodwork. A target blood level of CoQ10 of 4.1 micromol/L was found to be therapeutic. Studies also showed that blood mononuclear cells (a diverse mixture of immune cells) may be better for monitoring its levels.
The Bottom Line on CoQ10 Dosage and CKD
CoQ10 can be beneficial for patients with kidney disease. It may improve cholesterol levels and blood pressure control and decrease oxidative stress, as well as potentially improve renal function. A dose of 200-300 mg daily divided into three doses (100 mg three times a day) is recommended for patients with kidney disease. Consult with your integrative kidney specialist to customize your CoQ10 dosage, especially if you are on medications that deplete CoQ10.
Download Your Copy!

Inflammation in CKD
By Majd Isreb, MD, FACP, FASN, IFMCP
Causes of inflammation in chronic kidney disease
Inflammatory markers were found to be elevated in CKD. The levels of these markers increase with the decrease in kidney function. They also increase with increasing urine protein (albuminuria).
There are several reasons for immune imbalance and inflammation in CKD. Some of these factors are linked directly to the cause of CKD, while others are related to genetic predisposition and epigenetic factors.
Uremic environment
Furthermore, the accumulation of uremic toxins in CKD leads to oxidative stress. We discussed oxidative stress in a previous blog. When oxidative stress (or free radicals) builds up and aren’t handled properly, they can damage enzymes, cells, and tissues. Oxidative stress has been identified as a root cause of many diseases, including heart disease, Alzheimer’s, Parkinson’s, chronic kidney disease, and many more.
The build-up of harmful oxidative toxins in advanced CKD is associated with another problem which is described as “carbonyl stress.” Here, there are irreversible changes in many proteins leading to loss of their function. Higher oxidative and carbonyl stress is linked to inflammation in CKD. It is possible that carbonyl stress is just a biomarker of oxidative stress in CKD and not an independent phenomenon.
Download Your Copy!
Metabolic acidosis
Metabolic acidosis and high dietary acid load are very common in CKD. In metabolic acidosis, there is an increase in blood acidity. The foods that we eat may generate blood acidity (such as animal proteins) or alkalinity (such as fruits and vegetables). This is described as the dietary acid load We discussed these in a previous blog. They play an important role in the bone disease and protein-energy wasting that are common in CKD. In addition to that, a higher dietary acid load has been linked to faster progression of kidney disease. Metabolic acidosis has been linked to inflammation in CKD. In fact, higher inflammatory cytokines were seen in patients with metabolic acidosis.
Oral disease
Oral and periodontal disease (gum disease) are common in CKD. Over 700 species of microbes reside in the mouth. Together, they are called the oral microbiome. This microbiome produces metabolic by-products in the mouth. Some of these leaks into the bloodstream and lead to low-grade systemic inflammation.
Studies have shown that patients with severe gum disease have elevated levels of pro-inflammatory mediators and increased numbers of white blood cells known as neutrophils in the blood. This suggests that the inflammatory process in the mouth is affecting the whole body and probably injuring organs and tissues. In fact, successful treatment of gum disease was associated with improvement in inflammatory markers.
Intestinal dysbiosis
In CKD there is something called the dysbiosis cycle. We discussed this in a previous blog. Dysbiosis is a state of the intestinal microbiota where “bad” bacteria (or other organisms like yeast or parasites) outgrow the “good” bacteria. In dysbiosis, we see a rise in inflammatory markers that interact with the lining of the gut and result in damage and increased intestinal permeability (also known as “leaky gut”). “Leakiness” of the gut lining allows gut bacteria or bacterial particles from the gut into the bloodstream. It causes shifts in the breakdown of nutrients, including amino acids, which leads to the formation of many gut-derived uremic toxins. Intestinal permeability has been linked to autoimmune disease and systemic inflammation.
Vitamin D deficiency in CKD
Vitamin D deficiency in chronic kidney disease is quite common with 80% of CKD patients having been found to have low levels of 25-hydroxy vitamin D (25 (OH)D) when measured in the serum. Several factors have been implicated in the cause of this deficiency including inadequate outdoor physical activity, inadequate dietary intake, genetic variations, and loss of 25(OH)D in the urine. Vitamin D deficiency has been associated with an imbalanced immune system in CKD. This link between deficiency of vitamin D and kidney disease is important because it can lead to further inflammation in CKD.
Obesity
A higher body mass index is one of the strongest risk factors for CKD. In fact, the incidence of obesity-related kidney disease has increased 10-fold in recent years. Higher visceral fat is linked to increased protein in the urine (albuminuria). Visceral fat was also found to be associated with higher circulating inflammatory cytokines such as IL-6. This is because visceral fat cells produce these inflammatory cytokines.
Decreased clearance of cytokines
It has been well-documented that the levels of circulating inflammatory cytokines are elevated in kidney disease. Cytokines are chemical messengers that are produced by cells of the immune system. There are inflammatory cytokines and anti-inflammatory cytokines. When inflammatory cytokines are high in the blood, it is like a burning fire in the body, damaging and irritating tissues. In addition to the increased generation of cytokines in CKD, decreased renal clearance plays a significant role in the high level of cytokines in the blood.
Symptoms of inflammation in CKD
Higher levels of inflammatory markers such as IL-1beta, IL-6, TNF-alpha, C-reactive protein (CRP), and fibrinogen were found in patients with CKD, according to the Chronic Renal Insufficiency Cohort (CRIC) study. Inflammation is associated with many problems in patients with CKD. These problems include:
- Faster progression of kidney disease
- Increased protein loss in the urine
- Increased risk of heart disease and death
- Increased itching
- Worsening nutrition and protein-energy wasting
- Depression
- Worsening bone-mineral disorder
- Worsening anemia
Healing inflammation in CKD
When thinking about the best treatment for inflammation in CKD, it is crucial to address its root cause. For example, correcting the uremic environment may include strategies to decrease animal protein intake through a plant-dominant dietand a personalized gut restoration protocol. To address vitamin D deficiency, it is always recommended to check for it and correct it in all CKD patients.
Optimizing a person’s nutritional status is fundamental in healing inflammation in CKD and improving all the outcomes mentioned above. In addition, many supplements have been found to improve inflammation in CKD. These include omega-3 fatty acids, decaffeinated green tea extract, pomegranate, soy isoflavones, fiber, and probiotics.
The bottom line on inflammation and CKD
Low-grade inflammation is a common feature of CKD. Multiple root causes can be identified as a source of inflammation in kidney patients. These include decreased clearance of cytokines, oral and intestinal dysbiosis, vitamin D deficiency, uremic toxins, metabolic acidosis, and obesity. It is crucial to address the root causes of inflammation to improve kidney and heart outcomes in these patients.
The post Inflammation in CKD appeared first on Integrative Kidney.]]>By Majd Isreb, MD, FACP, FASN, IFMCP
What is oxalate?
Before we discuss the effects of oxalate on the kidneys, let’s answer the question: what is oxalate? Oxalic acid (or its ionized form, oxalate) is a simple chemical compound that humans cannot break down. It is essentially, therefore, a waste product that the body must eliminate. The kidneys are the major site of that elimination.
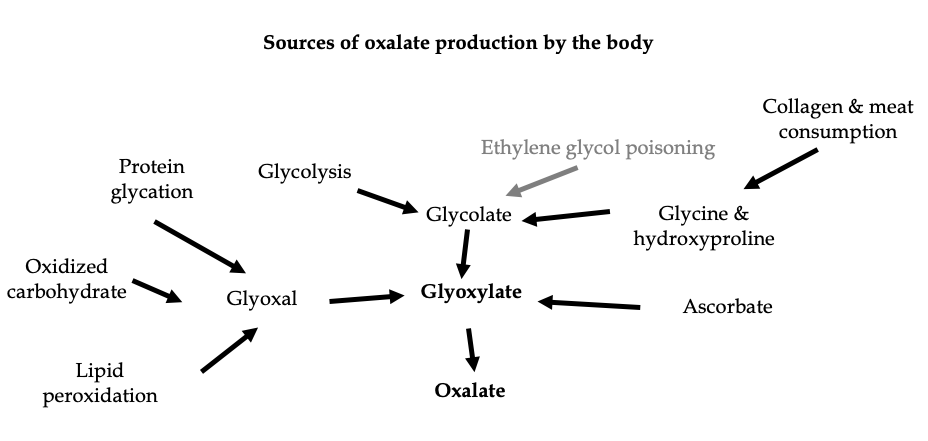
Despite its chemical simplicity, oxalate is actually very complex to study and understand. Most of the circulating oxalate is formed in the liver from the oxidation (the chemical combination with oxygen) of a substance called glyoxylate. Glyoxylate has two main sources. One is from carbohydrate metabolism and the other is from the degradation of collagen and protein.
While the human body cannot break down oxalate, it can use its precursor, glyoxylate, by converting it into the amino acid glycine. Vitamin B6 is a major cofactor in this conversion. This makes oxalate and glyoxylate a central link between the metabolic pathways of amino acids and carbohydrates.
Metabolism of ascorbic acid (vitamin C) can also lead to the formation of oxalate (we will discuss this in detail in a future blog). Finally, it has been noted recently that oxalate can be generated from the breakdown of proteins, carbohydrates, or lipids that have been damaged by oxidative stress or elevated blood sugars (see figure). Oxidative stress is an imbalance between free radicals and antioxidants. The body needs some oxidative stress, but too much of it can cause problems.
All of the above pathways are responsible for 80% of oxalate produced by the body and excreted in the urine.
Download Your Copy!
Dietary oxalate sources
Dietary sources of oxalate include green leafy vegetables, different nuts, seeds, and roots, cocoa, and tea. Healthy individuals usually consume 100-200 mg of oxalate per day. The small intestine is the primary site for oxalate absorption. But it is important to realize that not all consumed oxalate ends up getting absorbed. This is because of three factors:
- Oxalate tends to bind with calcium and magnesium in the diet to form insoluble complexes that end up excreted in the stool.
- The gut microbiome contains bacteria that break down and use oxalate for energy. Probiotics containing Oxalobacter formigenes and others decreased serum oxalate levels. Diets that are high in fiber are also associated with a decrease in intestinal oxalate absorption.
- Finally, the gut can excrete oxalate.
In fact, studies have shown that in healthy individuals, only 8% of the average daily intake of oxalate is absorbed.
Elevated oxalate in the urine – hyperoxaluria
When there is excessive oxalate in the blood, the excretion of oxalate in the urine increases. This is called hyperoxaluria. Hyperoxaluria is usually defined by the excretion of more than 40-45 mg of oxalate per day in the urine.
There are two major causes of hyperoxaluria:
- Genetic (primary hyperoxaluria). Here, there are mutations in enzymes that increase the production of oxalate by the liver.
- Secondary or enteric hyperoxaluria. This occurs in patients with inflammatory bowel disorders, malabsorption, or certain bariatric surgeries. In this type of high urine oxalate, the fat that is not absorbed by the gut binds dietary calcium. This leaves soluble oxalate more available for absorption into the systemic circulation, which increases levels of oxalate.
Oxalate and Kidney disease
Oxalate effects on the kidneys
Unquestionable data shows that patients with decreased kidney function tend to have higher levels of oxalate. In fact, oxalate excretion decreases with decreasing kidney function (as measured by glomerular filtration rate, GFR).
Oxalate has been found to be inflammatory to the proximal tubules of the kidneys and it leads to interstitial fibrosis (scarring). Proximal tubules are the part of the kidney cell that recycles (reabsorbs) the molecules that the kidneys are filtering. Oxalate also binds to calcium systemically and deposits in tissues causing fibrosis in the heart and hardening of blood vessels.
What causes kidney stones?
We discussed the different types of kidney stones in a previous blog. Calcium oxalate stones are the most common types of kidney stones. Excessive oxalate in the urine can also bind calcium, forming kidney stones.
In short, excessive urinary oxalates can cause kidney stones, and oxalate and kidney stones are something that kidney disease patients need to be cautious about.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Is dietary oxalate toxic to the kidneys?
Proponents of restricting dietary oxalate tend to restrict many of the plant sources of oxalate mentioned above. It is true that there are animal models of dietary oxalate-induced kidney disease. However, these animals were fed a massive amount of oxalate and a calcium-free diet. On the other hand, it is critical to emphasize here that there are no real outcome studies that demonstrated the efficacy of a low-oxalate diet on the progression of kidney disease or even kidney stones. In fact, limiting dietary oxalate may lead to adverse outcomes such as these:
- It may be harmful to the diversity of the gut microbiome and lead to dysbiosis. The gut microbiome is the population of microorganisms that reside in the gut. An imbalance, where the bad microbes outnumber the good, is called dysbiosis. Dysbiosis can promote kidney disease.
- A low-oxalate diet limits foods that have proven benefits in kidney disease, cancer, and cardiovascular disease prevention. Every day, there are new studies confirming the benefit of a plant-based or Mediterranean diet for kidney health.
- It decreases the intake of nutritional antioxidants. Oxidative damage is a contributing factor for people with CKD.
- It reduces the quality of consumed nutrients that may contain magnesium, fiber, and alkali, which are beneficial for health.
- It does not address the root cause of kidney disease or calcium oxalate stones.
- It does not address the bigger portion of oxalate produced from animal protein and collagen by the liver, which may not be as healthy as plant-based sources.
The bottom line on oxalate and kidney disease
Oxalate levels increase with advancing kidney disease and can lead to inflammation and its faster progression. Yet, going on a low-oxalate diet is not an evidence-based recommendation for chronic kidney disease patients and could cause more harm than good. Consuming a well-balanced, whole-food, plant-dominant diet that is rich in calcium, magnesium, probiotics, and prebiotics (fibers) is more important to slow the progression of kidney disease and improve its outcomes. These dietary changes should be done under the care of an integrative team comprised of a nephrologist and dietitian/nutritionist and a coach. Join us for free to learn about the integrative medicine approach to kidney health.
The post Dietary Oxalate and Kidney Disease: Is Dietary Oxalate Toxic to the Kidneys? appeared first on Integrative Kidney.]]>

Selenium, the micromineral
Selenium is classified as a trace mineral because it is typically needed in small doses to exert its biological effects. Unlike other trace minerals, though, selenium does not act as a cofactor of various enzymes or proteins. Instead, it is incorporated in the essential structure of various proteins called selenoproteins. There are about 20 known selenium-containing proteins in humans. Glutathione peroxidases (GPX) are some of the most important selenoproteins.
As mentioned earlier, selenium helps protect the body from oxidative stress. But what is oxidative stress? Free radicals, which cause oxidative stress, are a normal byproduct of daily living that happens in our cells. You might think of these as waste products. They need to be neutralized (or “reduced”) by antioxidant enzymes so that the cell stays healthy. When free radicals build up and aren’t handled properly, they can damage enzymes, cells, and tissues. Oxidative stress has been identified as a root cause of many diseases, including heart disease, Alzheimer’s, Parkinson’s, chronic kidney disease, and many more. A nutrient like selenium that prevents free radical buildup can help to repair cells, tissues, and improve diseases of oxidative stress.
Glutathione, the master antioxidant
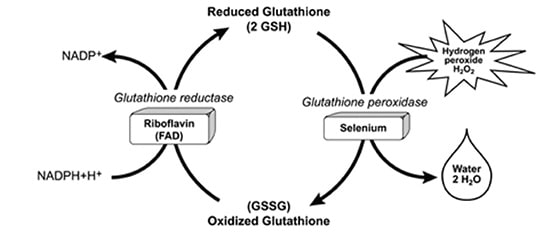
Selenium is crucial to the functioning of the antioxidant glutathione. Glutathione is a substance made of three amino acids (cysteine, glycine, and glutamic acid). Glutathione helps to recycle other antioxidants. On top of that, GSH has other functions such as metabolism, cell signaling, and protein interactions that can also mediate defense against oxidants. It helps the body get rid of heavy metals and a long list of toxins.
Glutathione is present in the cells in two forms: reduced (GSH) and oxidized (GSSG). The cycling between GSH and GSSG is what makes glutathione a major antioxidant. Simply put, when GSH is exposed to an oxidant it gets oxidized to GSSG by an enzyme called glutathione peroxidase. GSSG is, then, recycled back into GSH by an enzyme called glutathione reductase (see figure.)
The enzyme glutathione peroxidase is a selenoprotein that mediates the oxidation of GSH to GSSG. Selenium is an integral component of this enzyme. Selenium is, therefore, crucial for the body’s antioxidant ability.
Selenium deficiency and absorption
Selenium is primarily absorbed in the small intestines by active transport via a sodium pump, the means by which sodium ions are transferred across a cell membrane. Selenium in food exists in two forms: inorganic and organic (bound to methionine, called selenomethionine, or cysteine, called selenocysteine). The human body absorbs more than 90% of selenomethionine but less than 50% of the inorganic forms.
Food sources of selenium include Brazil nuts, seafood, and organ meats. In fact, two Brazil nuts have more than the daily requirements of selenium.
Selenium intakes and serum concentrations in the US vary by region because of differences in the amounts of selenium in the soil. Aging, smoking, and alcohol consumption affect selenium absorption. Men tend to consume more selenium and have slightly higher serum selenium concentrations than women.
Selenium deficiency is not common. However, insufficient selenium levels may predispose people with additional stressors to be at higher risk of certain illnesses. Selenium status can be measured in serum. Normal levels are 70-150 ng/mL. Studies have shown that increasing serum selenium levels were associated with decreased mortality up to a level of 130 ng/mL.
Download Your Copy!
Selenium and kidney disease
The kidneys and the thyroid gland have the highest concentration of selenium in the body. It turns out that the kidney proximal tubule cells have glutathione peroxidase near their basement membranes. This indicates that glutathione activity is necessary in that region due to high oxidative stress. This is probably because these cells are actively engaged in handling various toxins and in the reabsorption of amino acids, glucose, mineral salts, and other things the body wants to hold onto and recycle.
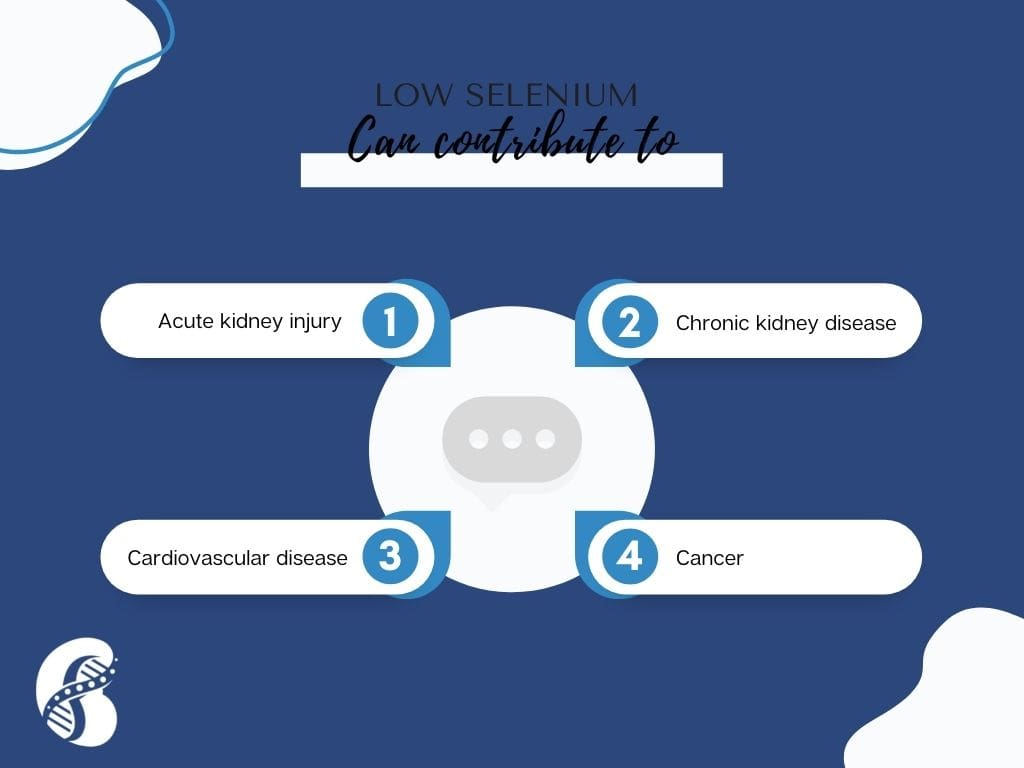
In addition, selenium levels have been found to be decreased in patients with chronic kidney disease (CKD). Selenium deficiency was also found to cause acute kidney injury in animal models. This was believed to be due to oxidative stress and mitochondrial dysfunction.
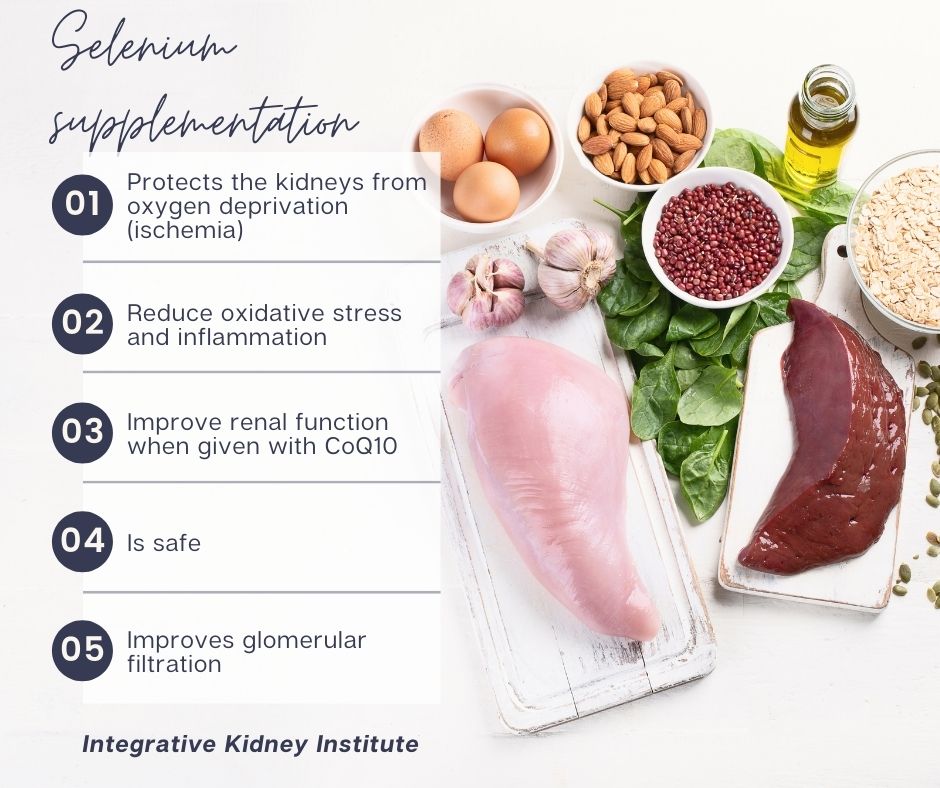
Studies have shown that using selenium as a kidney support supplement protects the kidneys from ischemic injuries, which occur when the kidneys are deprived of sufficient oxygen. Selenium supplementation also leads to reduced oxidative stress and inflammation in the kidneys. Supplementing selenium and CoQ10 in elderly patients with CKD and selenium deficiency was found to improve renal function. The dose of selenium that was used was 200 mcg per day.
On top of that, a small human study found that supplementing selenium in an amount as high as 700 mcg daily was not only safe, but it also improved glomerular filtration rate over a period of six weeks. It was thought that selenium plays a role in regulating the kidney blood vessels. Supplementation at these high levels should be done under the supervision of a healthcare provider.
Finally, selenium deficiency is associated with cardiovascular disease and cancer, leading to increased burden of comorbidities in patients with kidney disease.
Selenium and heavy metal toxicity
As we saw, selenium is incorporated in many enzymes and proteins that have antioxidant properties. That is why it plays an important role in protecting against the toxicity of heavy metals. In fact, selenium has been found to decrease the toxicity of arsenic and cadmium on the kidneys. This is thought to be due to selenium’s role in transforming these toxins into non-toxic forms. It is also related to the antioxidant activityof selenium-containing enzymes. Selenium was also found to protect the kidneys from the toxicity of mercury.
Selenium toxicity
Chronic high intake of selenium can be associated with health problems. The first sign of excessive seleniumis garlic breath odor and a metallic taste in the mouth. Selenosis (selenium poisoning) is characterized by hair and nail loss, mottled teeth, GI symptoms, fatigue, and irritability.
Are You Selenium Deficient?
Measuring oxidative stress in patients with CKD can be accomplished by various methods. One of our favorites is measuring the GSH:GSSG ratio. The ratio of GSH to GSSG gives an idea about the exposure to oxidative stress. In healthy cells this ratio is more than 100 but it drops to less than 10 in cells that are exposed to oxidative stress. However, this test is not widely available.
An alternative method to assessing oxidative stress is by measuring the damaged caused by free radicals. This damage can be measured in protein, lipid or DNA. Urinary 8-hydrox-2-deoxyguanosine (8-OHdG) is a test that measures oxidation of DNA. High levels are associated with increased oxidative stress. This test is commercially available.
Patients who have a low GSH:GSSG ratio or high urinary 8-OHdG can have their selenium serum level measured. We aim for a level of 100-130 ng/mL. Those who have low selenium can benefit from supplementation and follow up measurement in three-six months. Once again, we emphasize the need to take high-quality supplements to improve kidney function. Acute selenium toxicity has been reported due to the ingestion of an over-the-counter liquid supplement containing 200 times the labeled amount of selenium.
Ideally, once target serum levels are reached, the patient can maintain adequate selenium intake through a plant-dominant diet rich in phytonutrients.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
The bottom line
Selenium deficiency is linked to oxidative stress and inflammation, two processes that damage the kidneys. This is why adequate selenium status is essential for kidney health. Patients with kidney disease should aim for 200 mcg of selenium daily either through high-quality supplements or diet. This can be easily accomplished by eating only two Brazil nuts a day.
The post Is Selenium Good for Kidney Disease? appeared first on Integrative Kidney.]]>

Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>Low magnesium levels have been associated with a number of adverse events, such as high risk for heart disease. However, little is understood about magnesium and kidney health. Here, we will discuss the potential benefits of magnesium on the kidneys. This is one of two articles on magnesium and kidneys. For more on how to test and treat kidney patients with magnesium deficiency, see part two, “Magnesium Deficiency: Assessment and Management for Better Kidney Health.”

Dietary sources of magnesium
A daily intake of 3.6 mg/kg is necessary to maintain magnesium balance in humans under normal conditions. This is estimated to be between 320 to 420 mg/day (13–17 mmol/day) for adults. Sadly, there has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is due to depletion of magnesium in soil over time. This, along with the rise of ultra-processed food, sodas, and taking medications such as proton pump inhibitors and diuretics that deplete magnesium levels (polypharmacy), has led to rising prevalence of magnesium deficiency.
Traditionally, the highest food sources of magnesium are:
- Leafy greens (78 mg/serving on average)
- Nuts (80 mg/serving on average)
- Pumpkin seeds have the highest level of magnesium per serving (156 mg).
- Whole grains (46 mg/serving on average)
A complete list of foods high in magnesium can be found here.
Can Magnesium Help Kidney Function?
There are many potential benefits of magnesium for kidney health including improving blood pressure control, insulin sensitivity, bone health, vascular health, and preventing kidney stones. Let’s explore the data.
Magnesium and blood pressure control
Magnesium supplementation may help reduce blood pressure (BP) by increasing the production of nitric oxide. Nitric oxide acts as a signaling molecule that helps relax blood vessels, which lowers BP. In fact, a review of 34 studies showed that supplementing magnesium with an average dose of 368 mg per day for 3 months can decrease systolic BP by 2.00 mmHg and diastolic BP by 1.78 mmHg. This supplementation was accompanied by 0.05 mmol/L increase in serum magnesium levels.
Download Your Copy!
Magnesium and insulin sensitivity
Diabetes is one of the major risk factors for kidney disease worldwide. Higher dietary intake of magnesium has been correlated with lower diabetes incidence. A review of 18 studies in people with diabetes showed that magnesium supplements reduced fasting plasma glucose levels. In people who are at high risk for diabetes, magnesium supplementation significantly improved plasma glucose levels after a 2-hour oral glucose tolerance test. These effects are thought to be due to the effects of magnesium on insulin receptors and signaling that allows for improvement in glucose transport and utilization.
Magnesium and vascular health
Magnesium levels have been associated with a lower incidence of cardiovascular disease. In fact, supplementing with magnesium was associated with improvement in vascular flow and endothelial function. Endothelial function refers to the lining of the blood vessels, which is involved in regulating blood vessel health and blood clotting.
Studies in patients receiving dialysis have shown that having a lower serum magnesium level is a significant risk for cardiovascular mortality. Laboratory data show that magnesium inhibits high phosphate-induced calcification of vascular smooth muscle cells. Calcification of arteries is a strong predictor of heart disease and heart-disease-related death.
Magnesium and vitamin D
Magnesium is essential to vitamin D metabolism. Vitamin D that we eat or make in our skin from sun exposure circulates in the blood and is bound to vitamin D binding protein (VDBP). VDBP binding activity depends on adequate magnesium levels. In addition, magnesium is an essential cofactor for the enzymes that activate vitamin D. Studies have demonstrated that magnesium deficiency is associated with impaired vitamin D metabolism.
On the other hand, taking large doses of vitamin D can induce severe depletion of magnesium. This is thought to be due to the overutilization of magnesium. Therefore, adequate magnesium supplementation should be an important part of vitamin D therapy.
Adequate magnesium supplementation should be an important part of vitamin D therapy.
Share on X
Magnesium and bone health
Besides magnesium’s effects on vitamin D metabolism, it is an essential component of hydroxyapatite, an essential component of bone and teeth. In fact, 60% of total Mg is stored in the bone. Low magnesium intake was found to be associated with lower bone mineral density in postmenopausal women. Magnesium deficiency contributes to osteoporosis directly by acting on crystal formation and on bone cells and indirectly by impacting the secretion and the activity of parathyroid hormone (PTH) and by promoting oxidative stress and inflammation.
In addition, a review of 8 studies looked at magnesium and chronic kidney disease (CKD). The study investigated magnesium supplementation on parameters of CKD-related mineral bone disease (CKD-MBD). Mg supplementation improved PTH levels and carotid intima-media thickness (CIMT). Low serum Mg levels were also found to impact PTH and worsen osteoporosis in CKD patients, particularly with diabetes.
Magnesium and kidney stones
Mg acts as an inhibitor of calcium oxalate crystallization and stone formation in the urine. It also decreases the absorption of dietary oxalate in the gut. Mg supplementation in patients with kidney stones was found to decrease the incidence of stone formation even in patients without signs of Mg deficiency.
Magnesium as a phosphate binder
Hyperphosphatemia (high phosphate level) is common in advanced kidney disease. Many kidney patients with stage 4 and above use binders that bind phosphate (or “phosphorus,” as it is commonly known) in the food and prevent it from getting absorbed. High phosphate levels have been associated with poor bone and vascular health in kidney patients. In fact, higher dietary phosphate load can be seen in earlier stages of CKD, and it can do harm even before it is detected.
Magnesium carbonate has been successfully used as a phosphate binder. Magnesium based phosphate binders were also found to reduce vascular calcifications in rats with kidney disease. Iron-magnesium hydroxycarbonate was also studied and found to be well tolerated and can effectively lower phosphate levels in dialysis patients. It is essential to know that most of the magnesium used as a phosphorus binder will not be absorbed.
The bottom line on magnesium and kidneys
Magnesium is essential to many biological functions. It has many health benefits for kidney, bone, and vascular health. Optimizing magnesium status is, therefore, an important step in the integrative approach to kidney health. In part two of this blog, “Magnesium Deficiency: Assessment and Management for Better Kidney Health,” we will discuss practical steps for figuring out a person’s actual magnesium status, the best form of magnesium to take, and the dose I recommend for each condition.
The post Magnesium and kidneys appeared first on Integrative Kidney.]]>
By Majd Isreb, MD, FACP, FASN, IFMCP
What is vitamin A?
Vitamin A is an essential fat-soluble nutrient known for its role in good vision. It is also needed for cellular growth and differentiation. Since skin and gut cells are some of the fastest-growing cells in the human body, they are most sensitive to vitamin A deficiency. Vitamin A is also important for cells of the immune system. Vitamin A plays an important role in these processes:
- Growth and differentiation of all cells
- Embryonic development
- Organ formation in utero
- Normal immune function
- Eye development and vision
- Red blood cell production
Chemically, vitamin A is a group of organic compounds that share a beta-ionone ring with an isoprenoid chain. These compounds are often referred to as “retinoids.” The name of the retinoid depends on the number of the rings, the size of the isoprenoid chain, and the end group. These include -carotene, -carotene, retinal, retinol (which is vitamin A per se), all-trans-retinoic acid, all-trans-retinyl ester, 9-cis-retinoic acid, and 11-cis-retinal. Now, forget about all these different compounds, and let’s simplify things by calling them all vitamin A.
Diet and vitamin A
Vitamin A is consumed in the diet either as preformed vitamin A or as a precursor provitamin A carotenoid. Provitamin A carotenoids are converted to vitamin A in human intestinal cells. Sources of preformed vitamin A include eggs, liver, butter, milk, and fortified cereals. Provitamin A carotenoids are mainly found in vegetables such as carrots, spinach, collards, pumpkins, and squash. The most common type is beta-carotene. On average, the standard American diet provides approximately 700-800 micrograms of “retinol activity equivalents (RAE)” per day. Most of that comes as preformed vitamin A. The recommended dietary allowance of RAE for men and women is 900 and 700 micrograms/day, respectively. This makes deficiency of vitamin A rare. I will not discuss the complex absorption of vitamin A in the gut and storage in the liver and cellular mechanisms of action in detail here. These are described in detail elsewhere. However, it is important to remember that the presence of fat in the diet greatly enhances vitamin A absorption since it is fat-soluble. It is also worth noting that excessive intake of preformed vitamin A can cause elevated vitamin A levels. But elevated vitamin A does not usually occur after increased intake of provitamin A carotenoids. This is because the conversion of carotenoids to vitamin A is regulated by a negative feedback loop. When adequate levels of vitamin A are present, the body downregulates the production of vitamin A from carotenoids.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Effects of vitamin A on the bone
Before we discuss the effects of vitamin A on bone in detail, we should review the processes of bone formation and maintenance.
Bone formation and maintenance
Our bones contain a small number of cells surrounded by a mesh of collagen fibers that provide a scaffolding for salt crystals. These salt crystals are made of calcium, phosphate, and carbonate which combine to create, the so-called, hydroxyapatite. The latter incorporates other salts like magnesium hydroxide, fluoride, and sulfate as it hardens, or calcifies, on the collagen scaffolding. Hydroxyapatite crystals give bones their hardness and strength, while collagen fibers give them flexibility. There are four types of cells that are found within the bone: osteoblasts, osteocytes, osteogenic cells, and osteoclasts. In adults, two processes are responsible for changes in the skeleton: modeling and remodeling. Bone modeling describes the process of new bone formation or bone resorption on a given bone surface. This process is important for bone growth and shaping during childhood and adolescence. Bone remodeling, on the other hand, is the process that is used to maintain and renew healthy bones during adulthood. In other words, in bone modeling either bone formation or bone resorption occurs, while in bone remodeling both bone resorption and bone formation occur together. For remodeling to occur the bone must be “dissolved,” and then a new bone is formed. In this process, osteoclasts dissolve old bone tissue at specific sites. This process is called resorption. Subsequently, new bone tissue is formed by the osteoblasts. Even though it may seem counterintuitive, bone resorption (breaking down old bone) is necessary for building new, healthy bone. So, in essence, modeling leads to the formation of new bone tissue where needed, while remodeling helps maintain and strengthen existing bone tissue.
Download Your Copy!
The role of vitamin A
Vitamin A has two different effects on bone depending on the dose. While adequate vitamin A intake was shown to maintain healthy bones, high levels of vitamin A have been shown to cause the opposite. Let me explain.
Good effects
Studies have shown that vitamin A is important for bone resorption. This is essential for maintaining healthy bone by remodeling as we discussed. In fact, adequate intake of vitamin A has been found to improve bone mineral density and decrease the risk of fractures.
Not-so-good effects
In the early 1900s, researchers found more osteoclasts in the bones of animals with high vitamin A levels. Later animal studies showed that excess vitamin A led to the formation of bones with a “moth-eaten appearance.” It was also demonstrated that vitamin A stimulated bone resorption. Recently, it was noted that vitamin A can stimulate mineral release and bone degradation in mouse bones. These effects were blocked by osteoclast inhibitors such as bisphosphonate and calcitonin. In essence, vitamin A can increase osteoclast formation and differentiation causing increased bone resorption. This is good for the maintenance of healthy bones but becomes harmful when there is excessive resorption at high levels of vitamin A. The evidence that supports this comes from studies that showed an increased risk of hip fractures in the lowest and highest vitamin A blood levels. There are other studies that also showed that a high daily intake of vitamin A was associated with decreased bone mineral density. In the NHANES study in the US, for example, daily vitamin A intake of more than 3,000 micrograms was associated with an increased risk of hip fracture. However, there was no increased risk of fractures with high beta-carotene intake. This low-dose versus high-dose phenomenon has been seen with other nutrients and is described as a U-shaped hormetic response. At low doses, there are symptoms of nutritional deficiency but at high doses, there are symptoms of toxicity.
The interaction between vitamin A & vitamin D3
As we noted before, nutrients don’t work solo. Optimal bone health requires optimization of vitamin D, vitamin K2, calcium, phosphorus, and magnesium. There have been reports of vitamins D and A opposing each other. Some studies showed that vitamin D protects against vitamin A toxicity. On the other side, excess vitamin A was also shown to reduce the effects of vitamin D toxicity. In humans, high vitamin A intake was found to decrease the ability of vitamin D to enhance calcium absorption in the gut. Studies have shown that the negative effects of excess vitamin A on bone mineral density and fracture risk are amplified when accompanied by vitamin D deficiency.
Vitamin A and kidney health
Several studies have shown that vitamin A levels increase with worsening kidney function and advanced CKD. This, along with the prevalence of vitamin D deficiency in this population, adds another layer of complexity to bone problems in kidney disease. This common complication of kidney disease is called chronic kidney disease-associated mineral bone disease (CKD-MBD). In CKD-MBD, there is abnormal bone turnover and increased vascular and soft tissue calcifications. Increasing intake of preformed vitamin A at the advanced stage of CKD can lead to worsening bone abnormality and elevated calcium levels in the blood. This, in turn, can increase the risk of vascular calcifications. Therefore, supplementing vitamin A in patients with advanced CKD is not generally recommended on a regular basis. Natural vitamin A intake through a diet high in carotenoids should be sufficient.
Assessing vitamin A status
Unfortunately, it is difficult to assess vitamin A status in an individual. This is because most vitamin A is stored in the liver and is released as needed to the blood. The two common ways to measure vitamin A status are measuring serum retinol and retinyl ester concentrations. There are also labs that measure beta-carotene levels. Serum retinol levels are only helpful if they are very low or very high. Levels < 1.05 micromol/L indicate vitamin A insufficiency. It has been suggested to use the ratio of serum retinyl esters to total serum vitamin A (retinol plus retinyl esters) as a marker for excess vitamin A. Serum retinyl ester levels exceeding 10% of total serum vitamin A may reflect excess vitamin A stores and potential toxicity.
How much vitamin A should I take?
Considering the above, we recommend our patients get most of their vitamin A from the diet (either as preformed vitamin A or provitamin A carotenoids). CKD patients can eat eggs, liver, butter, milk, carrots, spinach, collards, squash, and pumpkin with guidance from their dietitian/nutritionist and nephrologist. The following recommendations for CKD patients are based on anecdotal practice since the literature doesn’t support specific recommendations. Patients with stage I-IIIa CKD may take up to 2500-3000 IU of supplemental vitamin A (or RAE 900 microgram/day for men and 700 microgram/day for women.) Patients with stage IIIb-IV CKD should decrease their intake of vitamin A supplements by 50%. Supplementation with vitamin A is not recommended for patients with an estimated GFR of less than 20 ml/min. If supplementation is desired during periods of sickness (for example, a respiratory illness) to boost the immune system, we recommend using beta-carotene supplements instead of preformed vitamin A supplements.
The Bottom Line
Vitamin A has a great impact on bone health. It is essential for bone resorption and remodeling. However, excess vitamin A intake can lead to bone weakness and fractures. In patients with advanced CKD, vitamin A tends to accumulate, and supplementation is not recommended. If supplementation is desired to boost the mucosal immune system during periods of sickness and high demand, look for supplements that provide much of their vitamin A in the form of beta-carotene. The individualized integrative approach to CKD-MBD necessitates careful nutritional assessment and evaluation of vitamin A levels in addition to vitamins D, K, magnesium, calcium, and phosphorus. This can help develop a tailored lifestyle and nutrition plan that can be incorporated into the medical management of CKD.
The post Vitamin A in Bone Disease Associated with Chronic Kidney Disease appeared first on Integrative Kidney.]]>
Daily requirements of phosphorus
There is no doubt that the average person requires phosphorus in the diet to live a healthy life. On average, the normal person requires 900-1250 milligram of phosphorus a day. Higher intake is especially required for growing children and pregnant women. Unfortunately, the standard American diet often contains higher amounts of phosphorus.
Role of phosphorus
Phosphorus plays an essential role in bone and teeth formation. It is also required for metabolism and energy production in the body. It is crucial for normal heart and nerve functions. Phosphorus serves as a building block for many structural and functional proteins. Because of its important role, the gut, hormones, kidneys, bones, and even vitamin D are involved in regulating phosphorus levels in the body.
The different types of dietary phosphorus
To simplify, we divide dietary phosphorus into two major categories. The first is organic phosphorus which comes “naturally” from animal and vegetarian proteins. The second is inorganic phosphorus, which is present in food additives.
How the kidneys handle phosphorus
The kidneys are critical for keeping a balanced, steady state of phosphorus in the body. Most of the phosphorus ingested in food gets absorbed in the gut. Some of it leaves the body in the stool. Circulating phosphorus gets filtered by the glomeruli. In this process, tiny capillaries in the kidney take phosphorus out of the blood. However, the kidney tubules help reclaim that phosphorus. They add phosphorus back to the blood, a process called reabsorption. The glomeruli and kidney tubules work together to filter waste out of the blood, but also recycle nutrients that are needed, like phosphorus. The end result is that the amount of phosphorus excreted in the urine equals that absorbed in the gut.
Hormonal control of phosphorus balance
The reabsorption of phosphorus in the kidneys depends on several hormones. The parathyroid hormone (PTH) decreases phosphorus reabsorption. When phosphorus isn’t recycled, more phosphorus leaves the body through the urine. On the other hand, 1,23 (OH) vitamin D increases phosphorus reabsorption, meaning that the body holds on to more phosphorus. Vitamin D also increases phosphorus absorption by the gut.
Bones also influence phosphorus levels. They produce fibroblast growth factor 23 (FGF-23), which decreases phosphorus reabsorption. Simply put, FGF-23 tells the kidneys to excrete phosphorus in the urine, instead of holding on to it. The following picture summarizes phosphorus balance.
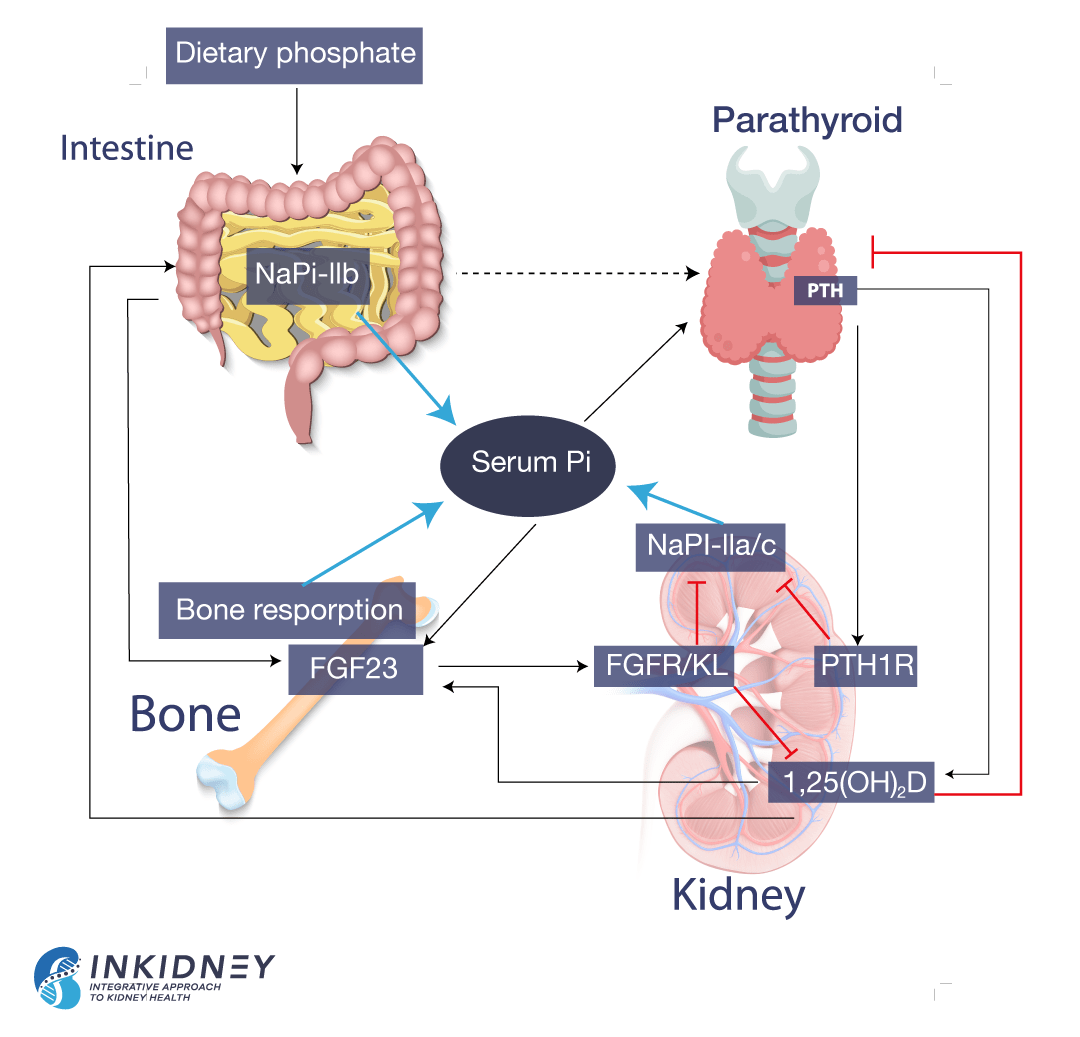
Phosphorus in kidney disease
In kidney disease, the ability of the kidney to excrete phosphorus progressively declines. Data shows that the decline begins early in kidney disease. In response, there is an increase in the production of PTH and FGF-23. Both hormones attempt to rid the body of phosphorus by decreasing the reabsorption of phosphorus by the kidneys. In the beginning, this effort maintains normal phosphorus in the blood. But as kidney disease advances, the ability of the kidneys to excrete phosphorus decreases further. Thus, in advanced kidney disease both PTH and FGF-23 are high, but phosphorus accumulates in the blood.
Elevated phosphorus binds to calcium and deposits in the blood vessels and under the skin. This leads to inflammation. In addition, several studies have shown that elevated levels of PTH and FGF-23 are associated with poor outcomes in kidney patients. Elevated levels of FGF-23 lead to poor cardiovascular outcomes and progression of CKD. Elevated PTH causes poor bone health in kidney patients.
Dietary phosphorus in kidney disease
Recent data showed that patients on plant-based diet are less likely to develop kidney disease. In fact, a plant-based diet is now recommended for kidney disease patients. Some worry that plant-based diets lead to higher intake of phosphorus. Many practitioners believe that a diet high in legumes, nuts and seeds will increase intake of phosphorus.
Yet, when we look at serving size, animal protein and dairy have the highest content of phosphorus. Besides, phosphorus in vegetarian sources is bound to phytic acid. Humans don’t have the enzymes needed to digest phytic acid. So, we actually do not absorb a lot of the phosphorus that is present in plant-based foods.
Actually, we only absorb 40% of vegetarian phosphorus as compared to 60% of animal-based sources of phosphorus.
But the food industry is actually adding phosphorus to many foods. This is part of food processing. Some use it as a preservative. Some use it to prevent discoloration. Others use it to improve taste. You often see names that are not easily recognizable. These names include calcium phosphate, disodium phosphate, phosphoric acid, or sodium triphosphate. These types of phosphorus are the inorganic phosphorus. We absorb almost 100% of phosphorus as food additives.
This is why it is so important to look at the ingredients. Any ingredient that has the four letters, “Phos” in it means that it contains phosphorus. You can find great resources about dietary phosphorus here.
The bottom line
Higher intake of dietary phosphorus leads to worse kidney outcomes. Processed food has the highest absorbable phosphorus, followed by animal proteins. Vegetarian sources have the lowest absorbable phosphorus. This supports the use of a whole foods, plant-based diet in kidney disease.
The post Dietary Phosphorus and Kidney Health appeared first on Integrative Kidney.]]>
We discussed the effect high intake of animal protein has on kidney health in a previous blog. According to the National Institute of Health, the Dietary Reference Intake (DRI) of protein for the average person is 0.8-1.6 g/kg/day (or 0.36-0.75 g/lb/day). However, according to the Kidney Disease Improving Global Outcomes (KDIGO) guidelines, kidney patients should limit their protein intake. A low protein diet (LPD) by the KDIGO guidelines is 0.8 g/kg/day (or 0.36 g/lb/day). That means a kidney patient who weighs 150 lbs (68 kg) has a dietary protein target of approximately 54 grams of protein per day (0.8g/kg x 68kg). Furthermore, it’s suggested that at least half of that comes from plant-based sources. Protein intake, along with other related nutrients, should be tracked closely through nutrient intake analysis, ideally by a nutritionist.
Different dietary protein regimens have been proposed in the literature for patients with Chronic Kidney Disease (CKD):
- A low-protein diet (LPD), with protein target of 0.6-0.8 g/kg/day;
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with essential amino acids, or
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with nitrogen-free keto-analogues of essential amino acids.
The controversy in the literature, however, has always been whether or not lowering the targeted protein intake puts patients at risk for nutritional deficiency, including essential amino acids and other nutrients often found in high protein foods like folate, B12, carnitine, and zinc. And in that case, is supplementation among these patients necessary and useful to maintain adequate nutrition?
Let’s focus on amino acids, but first let’s review.
What are essential amino acids?
Amino acids (AA) are the basic organic building blocks of protein. There are a total of twenty-two AA, each composed of a core structure made of nitrogen, carbon, hydrogen and oxygen, and variable side chain groups unique to each AA.
Of the twenty-two AA, there are nine essential amino acids (EAA). They’re called “essential” because the human body cannot make them, and therefore we must get from our diet. These EAA are:
- Histidine
- Leucine
- Isoleucine
- Valine
- Lysine
- Threonine
- Methionine
- Phenylalanine
- Tryptophan
The remaining thirteen AA are termed “unessential” not because they’re not important, but because the body can make them through various biochemical processes as long as the necessary building blocks are available from the diet.
When restricting protein intake in kidney patients, it is important that the patient receive adequate intake of EAA since adequate levels of these essential nutrients can only be maintained through food. This can be challenging to keep balanced, which is why we suggest working with a nutritionist to track and support adequate intake.
Using ketoacid analogues with very low protein diet for kidney health
To understand the relevance of ketoacid analogues in CKD, we need a biochemistry lesson in AA metabolism. Biochemistry isn’t everyone’s thing, so we summarized it here.
One thing to remember about AA metabolism is that it requires cofactors which are nutrients that facilitate the reactions. Vitamin B6 is a major cofactor here. Other cofactors needed in the amino acid breakdown (catabolism) include biotin, folate, and B12.
How do ketoacid-analogue supplements help in kidney disease?
Theoretically, the process described above can be used in reverse to reduce circulating nitrogen and the formation of nitrogen waste products (i.e., like urea), and, therefore, decrease the burden on the diseased kidneys. Supplementing ketoacid analogues of EAA in kidney patients on a LPD and VLPD, might simultaneously achieve two goals: 1. It closes the gap on adequate EAA nutritional need 2. It decreases the toxic nitrogen waste products contributing to disease complications. There is also some evidence that certain analogues can decrease protein breakdown in the body.
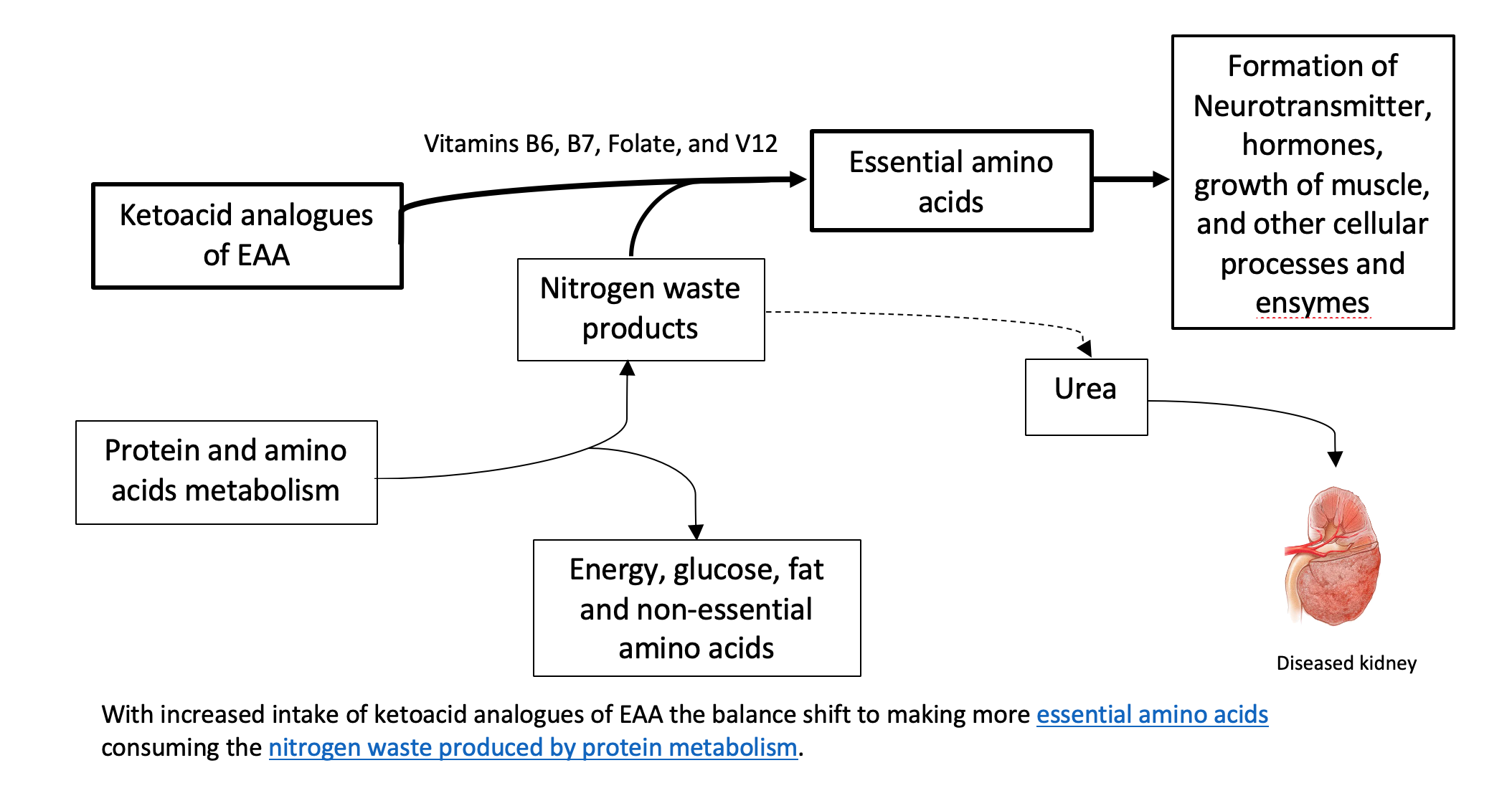
Some studies have demonstrated that very low protein diets supplemented with ketoacid analogues decrease waste product generation in kidney disease and prevent protein-energy decline. Supplementation of keto-analogues in kidney patients was found to decrease protein in the urine as well as improve bone health, insulin sensitivity, and blood pressure control. In addition, these dietary protein restrictions with keto-analogue supplements have been found to delay symptoms of kidney failure and the need for dialysis.
However, one of the major concerns of supplementing ketoacids in kidney patients on VLPD is that it does not address the other micronutrients and cofactors that need to be supplemented. it also does not take into account the carbohydrate and healthy fat content of a healthy diet.
Ketoacid analogue preparations
There are a few different preparations available in Europe, but only one available in the US. If one looks at the content of these preparations, it will become obvious that the kidney patient will need several pills of ketoacid daily to achieve an adequate daily dose. This makes this therapy very expensive and impracticable.
Luckily, there’s a way to use diet to balance macronutrients to induce the same effect. A low protein diet, with the majority of protein coming from whole food plant sources. When consuming animal protein, we recommend organic and grass-fed sources. Strictly vegetarian patients can still get adequate sources of EAA if they plan their diet well with the help of a nutritionist. If AA supplements are determined to be needed, one should aim to achieve the recommended daily intake from a trusted source (with good quality control). According to the World Health Organization, the recommended daily dose of each of the EAA is as follows:
| Essential Amino Acid | Recommended daily intake |
| Lysine | 30 mg/kg/day |
| Leucine | 39 mg/kg/day |
| Isoleucine | 20 mg/kg/day |
| Valine | 26 mg/kg/day |
| Threonine | 15 mg/kg/day |
| Phenylalanine | 25 mg/kg/day |
| Tryptophan | 4 mg/kg/day |
| Histidine | 10 mg/kg/day |
| Methionine | 10 mg/kg/day |
The bottom line on very low protein diet for kidney health
High animal protein intake is associated with faster progression of CKD, likely due to excess production of nitrogen byproducts from protein breakdown. Following a whole food plant-dominant diet can be helpful to delay the progression of kidney disease. The use of ketoacid analogues as supplements may not be practical. In order to assure that the patient is getting adequate sources of EAA and other necessary nutrients, dietary restrictions should be implemented with the help of an experienced nutritionist, preferably one trained in a personalized approach to kidney health.
The post Very Low Protein Diet for Kidney Health appeared first on Integrative Kidney.]]>
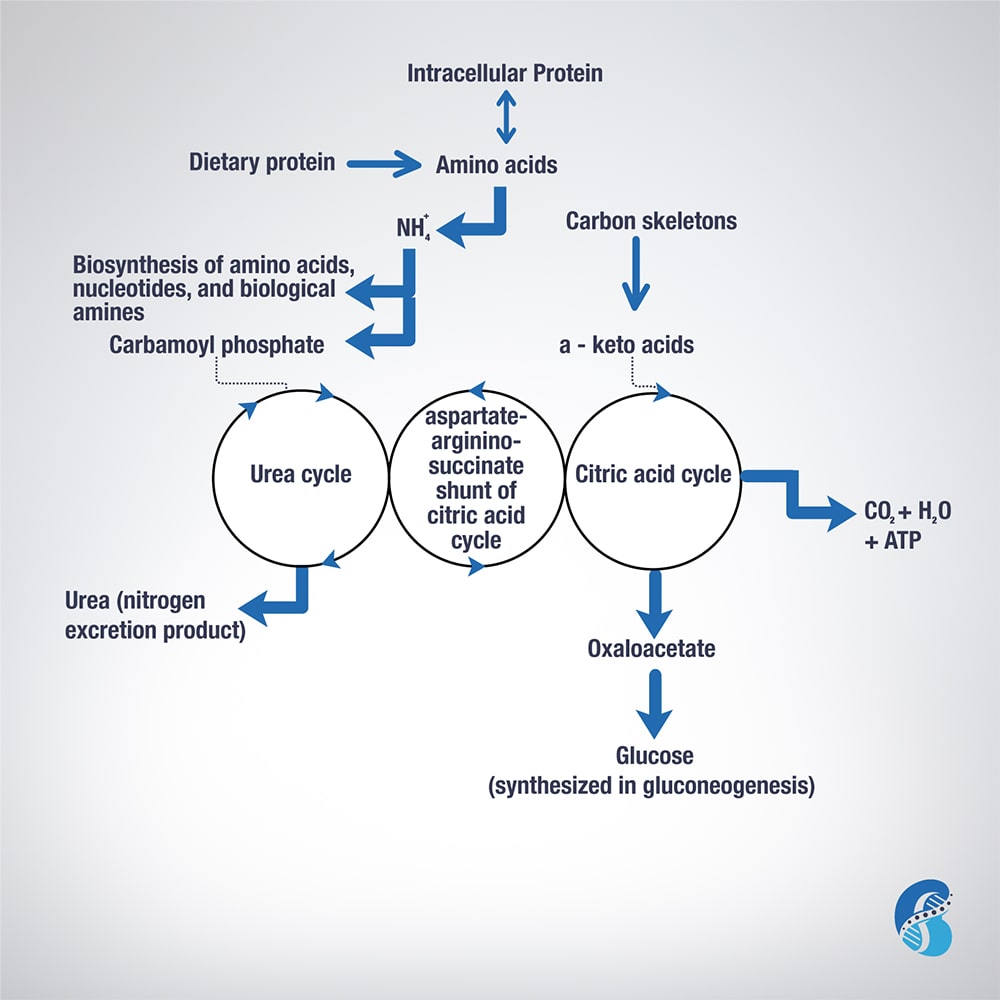
Excess AA in a protein-rich diet is metabolized through a process called transamination. This involves the removal of the nitrogen (or the amino group NH2 side chain) and replacing it with a keto group carbon structure forming a ketoacid or ketoacid analogue.
Apart from lysine and threonine, every EAA has a keto-analogue. The ketoacid then undergoes oxidation to carbon dioxide, water, and a three to four-carbon unit that can be converted by the liver into glucose (by gluconeogenesis) or into ketone bodies for energy.
The conversion of an amino acid to ketoacid depends on the enzyme aminotransferase. This enzyme is reversible (meaning it can convert amino acid to ketoacid or a ketoacid to amino acid). The speed and direction of this conversion depends on the amount of amino acids available, the amount of keto-acids formed, and the availability of the cofactors needed to power the process. These cofactors are nutrients that facilitate the reactions. Vitamin B6 is a major cofactor for this enzyme. Other cofactors needed in the amino acid breakdown (catabolism) include biotin, folate, and B12.
Now, remember that amino group (-NH2)? That can be used in the biosynthesis of non-essential AA and the excess ends up metabolized in the urea cycle to form urea and other nitrogen waste products that are excreted by the kidneys. It is worth noting that the AA arginine acts as a regulator of the urea cycle. Remember, arginine is essential for the production of nitric oxide, which plays an important role in blood pressure management. Issues with the urea cycle and nitric oxide balance may explain why kidney patients have blood pressure problems and may not feel well with arginine supplementation.
Overall, this process is summarized in this figure.
The post Biochemistry of Amino Acids Metabolism appeared first on Integrative Kidney.]]>







