
Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>
Altered vitamin K biodistribution may decrease the benefit of vitamin K2 supplementation in advance CKD
In this study, investigators sought to determine if there are other causes for vitamin K deficiency in advanced CKD beyond decreased dietary intake. They compared vitamin K uptake and distribution into circulating lipoproteins after a single administration of vitamin K1 plus K2 (MK-4 and MK-7) between patients on dialysis and healthy individuals.
They found that patients with uremia and advanced kidney disease don’t incorporate MK-7 well into HDL and LDL particles compared to healthy individuals. In addition, the combination of a statin and PPI was associated with signs of functional vitamin K2 deficiency in these patients.
In essence, patients with advanced kidney disease may not benefit as well from vitamin K2 supplementation. This highlights the importance of optimizing vitamin K2 status at earlier stages in CKD.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Higher levels of deoxycholic acid were associated with a higher risk of progression in CKD
Deoxycholic acid is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. Intestinal bacteria metabolize the primary bile acid, cholic acid, into deoxycholic acid (DCA).
Researchers studied 3,147 CRIC study participants who had fasting DCA levels. DCA levels above the median were independently associated with higher risks of ESKD and all-cause mortality.
This study highlights the importance of the microbiome and dysbiosis in the progression of kidney disease as we discussed in our blog.
Download Your Copy!
The higher number of medications a kidney patient takes the faster her kidney disease progresses
In a study performed in Japan of 1117 CKD patients under nephrological care, the use of a higher number of medications was associated with an increased risk of kidney failure, cardiovascular events, and all-cause mortality in patients with CKD. This is one of the major reasons we advocate for lifestyle modifications and coaching as the first and major step in the management of kidney disease.
Subscribe here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post December Research and News appeared first on Integrative Kidney.]]>
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Zinc: The Micromineral
Zinc is classified as a trace mineral because it’s typically needed in small doses to exert its biological effects. This nutritional mineral is involved in functions that include glycolysis (converting glucose into energy), digestion especially protein breakdown, bone formation, DNA metabolism, alcohol metabolism, angiotensin conversion, heme biosynthesis, and as an antioxidant protecting from reactive oxygen species (ROS) and the resulting cellular damage from inflammation. In fact, it’s so important, that it is the third most relevant intracellular ion (behind magnesium and potassium).
In addition, there are over 200 metal-containing enzymes, called metalloenzymes, responsible for catalyzing a variety of crucial reactions throughout the cells in our bodies. In fact, 20 distinct biological functions are associated with zinc metalloenzymes.
Zinc Absorption and Deficiency
Zinc is primarily absorbed in the small intestines via mechanisms called transcellular and paracellular transport. That means it can passively diffuse through tight junctions or by carrier mediated transporters. The latter means that absorption of zinc can be influenced by genetic SNPs which can result in decreased zinc transport and subsequently increased risk for deficiency.
Furthermore, absorption is reduced with aging and maldigestion/low stomach acidity. Drugs that inhibit stomach acid production, such as proton pump inhibitors (PPIs) and H2 antagonists (H2A) are thought to cause zinc depletion by reducing stomach acidity. This effect can be reversed by addressing the gut dysfunction through the 5R protocol.
Other drugs that may alter zinc status include angiotensin-converting enzyme inhibitors (ACEi), aspirin, thiazide and loop diuretics, long-term use of steroids, and fluoroquinolone, penicillamine, and tetracycline antibiotics. The degree of depletion is dependent on multiple factors, including individual differences, dose and length of therapy.
Assessment of zinc status through laboratory testing has been criticized as being unreliable. However, coupled with a comprehensive assessment of symptoms it can be very helpful in identifying mineral deficiency. Examples of symptoms of zinc deficiency include loss of/diminished taste and/or reduced sense of smell, frequent colds, slow wound healing, loss of appetite, presence of or worsening of acne/rosacea.
Zinc and kidney disease
The prevalence of zinc deficiency in chronic kidney disease (CKD) patients is well documented. Furthermore, zinc nutritional status is also altered in obesity. In fact, fasting plasma zinc concentrations are inversely correlated to BMI and plasma glucose levels.
Several inflammatory mechanisms are associated with zinc depletion, while adequate levels of zinc decreases formation of reactive oxygen species (ROS). ROS activate nuclear factor-κB (NF-κB) which induces the generation of inflammatory cytokines and adhesion molecules associated with various types of kidney diseases, including IgA nephropathy.
Superoxide dismutase (SOD), and enzyme which converts the oxidative superoxide into an inert product, hydrogen peroxide. The SOD enzyme requires zinc in order to work efficiently, and zinc deficiency reduces the activity of the enzyme and has been shown to increase inflammation-induced kidney damage, in part via this mechanism.
Another interesting associations include the impact of zinc deficiency on increased calcification in the development of kidney stones.
Zinc also plays an important role in protecting the kidneys from environmental toxins, especially heavy metal. Heavy metal toxicity, such as lead and cadmium, is significantly associated with kidney disease. Studies have shown that a deficiency in zinc, as well as other essential metals such as calcium or iron, can lead to increased absorption and toxicity of lead and cadmium. Therefore, providing yet another potential benefit of ensuring zinc repletion to protect against metal toxicity and its impact on kidney, as well as overall health.
Implications of zinc deficiency in cardiovascular and metabolic disease associated with KD
Zinc impacts kidney disease via indirect associations as well. Since cardiovascular disease (CVD) and diabetes are comorbid conditions associated with kidney disease, it makes sense that factors that impact them will also impact kidney disease.
It’s worth noting that various inflammatory markers that are triggered by deficiency of zinc, such as transcription of NF-κB, rise in IL-6, IL-2, and TNF-α, are associated with inflammatory/immune response that leads to systemic inflammation including atherosclerosis (vascular disease), a known complication of CKD.
Zinc has some interesting effects on blood lipids, including low density lipoproteins (LDL) and triglycerides (TG). Dyslipidemia is associated with increased risk of CVD. Zinc deficiency has also been associated with elevations in TG. Furthermore, maintaining adequate levels of zinc has a protective effect against atherosclerosis by inhibiting the oxidation of LDL by cells or transition metals.
In addition, there are multiple mechanism where zinc impacts metabolic disease. Interestingly, zinc is essential for insulin synthesis and release, and deficiency impairs the release of insulin contributing to insulin resistance and elevations in blood sugar.
Another hormone responsible for feeling of satiety (fullness after eating) is leptin, which is produced by fat cells. Leptin resistance is associated with overeating and obesity. Zinc depletion may impact leptin indirectly because leptin secretion is regulated by insulin. Furthermore, deficiencies in zinc impact the activity of peroxisome proliferator-activated receptors (PPARs). PPARs are associated with multiple metabolic pathways of lipid and glucose metabolism. They initiate cascade of mechanisms which activate atherogenesis, and are, therefore, central to metabolic disease, CVD, and kidney disease.
Food-based sources of zinc
Red meat, organ meats, and shellfish are considered the best sources of zinc, with levels significantly higher than vegetarian sources. Although not as high as animal sources, nuts and seeds are good sources of zinc. Although whole grains are good sources as well, refined grains are not because zinc is lost when the bran and germ coating is removed in processing. Unfortunately, although an essential part of a healthy diet, fruits and vegetables are not significant sources of this nutrient. Not surprisingly, vegetarians are at increased risk for zinc deficiency and should be screened routinely.
There are food interactions that reduce absorption of zinc. Phytic acid, naturally found in grains, may inhibit the absorption of zinc. Therefore it’s advisable to soak grains for 8-24 hours in filtered water and 1 tablespoon of lemon juice or apple cider vinegar prior to cooking to reduce levels of phytic acid and enhance absorption of zinc and other minerals. It’s also interesting to note that absorption of zinc is reduced by the intake of dairy, likely due to the high calcium content which is known to interfere with zinc uptake.
Supplementing zinc is a good option in cases of deficiency. There are multiple compounds available on the market. We recommend using zinc picolinate or zinc citrate. Zinc carnosine is useful as part of gut restoration protocol to address GERD and as part of a PPI tapering protocol. Note that the dosage and length of therapy should be customized to individual needs and tracked by your functional medicine provider or nutritionist. If you notice a metallic taste in your mouth, have your healthcare provider check your zinc levels to ensure they are not too high or toxic.
The Bottom Line
There are multiple suggested mechanisms contributing to the impact of zinc deficiency on kidney health. It’s no surprise considering the various mechanisms that involve zinc. It’s important to consider the impact of diet, medications, dysbiosis and intestinal permeability on zinc status and subsequently on protecting the kidneys from disease.
The post Zinc and Kidney Disease, Exploring the Links appeared first on Integrative Kidney.]]>
Angiotensin Converting Enzyme Inhibitors
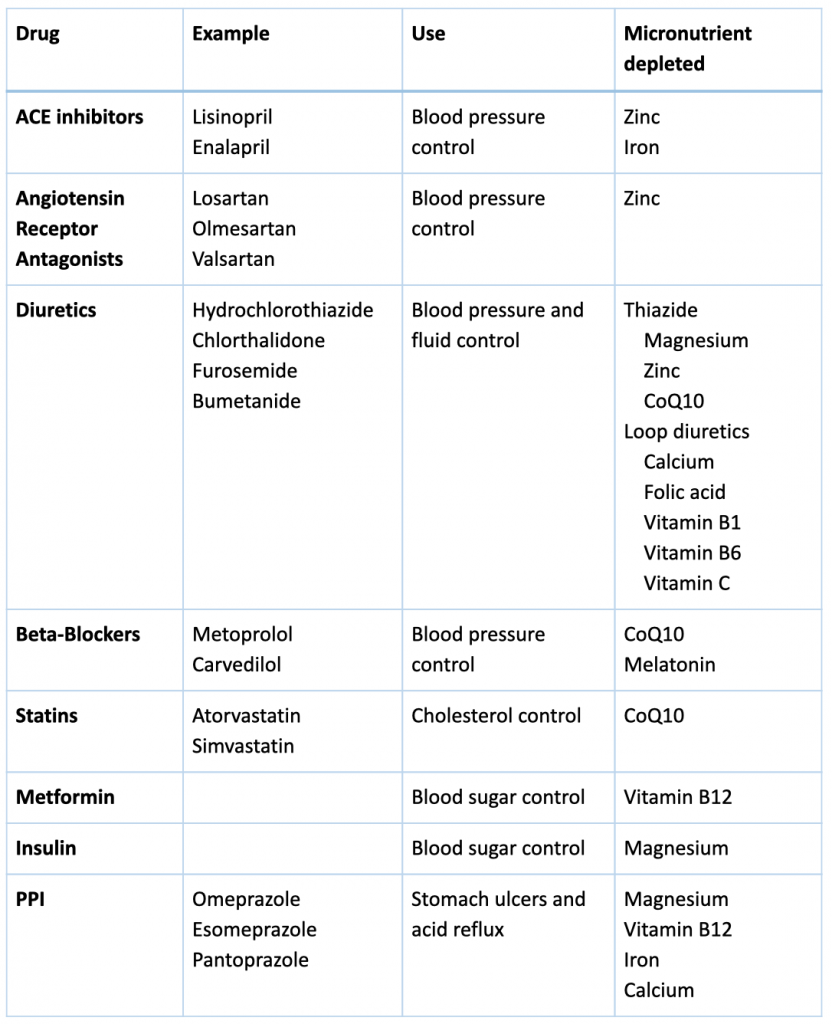
Angiotensin Converting Enzyme Inhibitors (ACEi) are one of the main classes of blood pressure lowering medications in Kidney disease. ACEi (such as lisinopril) have been found in multiple studies to improve outcomes in diabetic and non-diabetic kidney diseases. However, it’s well documented that ACEi can lead to increased levels of potassium in the blood. This elevation may have many serious effects, especially on the heart and the muscles. Furthermore, supplementing with arginine, a precursor of nitric oxide sometimes used to support blood pressure, while taking an ACEi may further increase the risk of increased blood potassium levels.
The use of ACEi also induces zinc deficiency. By binding zinc, ACEi cause an increase in zinc excretion in the urine. Zinc Deficiency has many negative effects on the body, including cardiovascular and metabolic disease risk.
Angiotensin Receptor Antagonists
Another kidney medications and micronutrients interaction involves angiotensin receptor antagonists (ARBs). Just like ACEi, Angiotensin Receptor Antagonists (such as losartan) are one of the most commonly used blood pressure lowering medications in kidney disease. Like ACEi, ARBs also lead to an increase in potassium so electrolyte balance should be monitored closely. Also similarly to ACEi, evidence suggests that ARBs can also lead to zinc deficiency.
Using the powerful antioxidant alpha-lipoic acid (ALA), which can be used as a supplement, has been found to enhance the effects of ARBs by decreasing inflammation and oxidative stress that leads to cardiovascular risk from atherosclerosis.
Diuretics
Diuretics, also known as water pills, are used in kidney patients for blood pressure and fluid control. They are one of the major causes of kidney medications and micronutrients interactions. There are multiple classes of medications that fall into this broad category, and each affects minerals like potassium, magnesium, calcium and various vitamins differently.
For example, hydrochlorothiazide (HCTZ) and furosemide, can lead to potassium and magnesium deficiency so these nutrients are monitored very closely in patients prescribed these drugs. However, their effect on calcium varies. Thiazide diuretics (such as HCTZ) increase calcium retention while loop diuretics (such as furosemide) induce calcium depletion.
HCTZ can increase zinc loss in the urine leading to zinc deficiency. Furthermore, HCTZ can lead to CoQ10 deficiency (by inhibition of NADH oxidase) an antioxidant compound produced in our cells associated with reduced function of cardiovascular muscle and reduced skeletal muscle strength (more about CoQ10 below under statins).
Loop diuretics, including furosemide, have been found to induce deficiency of folate, vitamins B1, B6, and C . Furthermore, drug-induced calcium loss in the urine associated with this class of diuretics can lead to increased risk of bone loss and increased risk for kidney stones.
Beta-blockers
Beta-blockers (BB), such as metoprolol, are commonly used blood pressure reducing medications because they have been found to improve cardiac outcomes.
However, these medications exert their effect while inhibiting enzymes that are dependent on CoQ10 (including NADH-oxidase and succinoxidase). While these medications do not lead to CoQ10 deficiency per se, the concomitant presence of a CoQ10 deficiency for any other reason can lead to greater inhibition of the heart muscle efficiency as well as leading to worsening heart failure.
In addition, BB (especially metoprolol) have been found to decrease the production of melatonin, a compound naturally produced by the body that acts as an antioxidant and sleep inducer. The production of melatonin in the evening as part of our natural sleep-wake cycle induces sleep, and therefore depletion of melatonin causes sleep disturbances. Poor sleep is a major risk factor for progression of kidney disease, diabetes, and hypertension. In patients taking these medications, melatonin supplementation was found to improve sleep quality.
Statins (HMG-CoA Reductase Inhibitors)
Statins, including atorvastatin and simvastatin among many others, are the most commonly used class of cholesterol lowering medications. One of the major drug-induced nutrient depletions of statins is due to the inhibition of the formation CoQ10. Along with insufficient vitamin D blood levels, depletion of CoQ10 along with certain genetic factors is associated with an increased risk of developing a common side effect referred to rhabdomyolysis (muscle breakdown).
CoQ10 is produced throughout the body in the energy powerhouse of the cells. It is a major part of energy production and metabolic process. ”Co” stands for coenzyme, referencing its action in assisting enzymes in an energy production process called the electron transport chain (ETC). The ETC is an essential part of how energy is derived from carbohydrates and fat. Therefore, depletion of CoQ10 not only increases oxidative stress, but also has a significant effect on the function of every organ in our body, and particularly impact the cardiovascular and renal system. .
In addition,statins have been shown to deplete trace elements, including zinc, copper, and selenium. Furthermore, statins while lowering serum fatty acid concentrations (LDL, TG, etc), they also negatively alter the blood levels of important heart-protective polyunsaturated fatty acids (PUFAs while increasing inflammatory arachidonic acid levels. These drug-nutrient interactions are inconsistent from patient to patient, likely due to genetic variations, agent chosen, and dose. Therefore, monitoring lab and symptoms of deficiency or imbalance is ideal.
Metformin
Metformin is used to improve blood sugar by improving insulin sensitivity. Although some kidney patients might be taking metformin, it is not recommended for use in advanced kidney disease due to the increased risk of lactic acidosis.
Metformin is known to decrease the absorption of vitamin B12 by inhibiting the secretion of intrinsic factor, a compound needed to absorb B12 from food. B12 is a water-soluble vitamin and is essential for many processes, including energy production, production of endogenous proteins used for various cell functions including DNA repair, production of blood cells, myelin (cells that make up the nervous system), and some antioxidants. Therefore, supplementing vitamin B12 in patients taking metformin may prevent anemia, maintain brain health, cell production, and support the detoxification pathways.
Insulin
Insulin which is injected to lower blood sugar in diabetics can lead to significant loss of magnesium in the urine. Low magnesium can lead to significant effect on vascular, bone and heart health. In fact, supplementing magnesium in diabetics can improve insulin response.
Proton Pump Inhibitors (PPIs)
Proton Pump Inhibitors (PPIs) are one of the most commonly used drug classes globally, including in patients with kidney disease. Drugs belonging in this class include omeprazole and pantoprazole, which are commonly used for “acid reflux”, decrease stomach acid production leading to nutrient malabsorption and dysbiosis. They can lead to a decrease in calcium, iron, magnesium and vitamin B12. It’s also well-established that they also directly contribute to kidney injury. Despite that, their use remains common and we should remain vigilant in general.
Bottom Line
Many kidney medications can lead to micronutrient deficiencies with negative effect on outcomes and organ functions. The following table helps summarize these for you with general suggestions for supplementation. We recommend that you work with your integrative medicine provider to address complementary therapy to optimize you treatment and nutritional status.

By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Nutrients don’t work solo, they function as part of a symphony with other nutrients to help our body perform at its best and prevent disease, including kidney damage and chronic kidney disease (CKD).
Nutrients don’t work solo, they function as part of a symphony with other nutrients to help our body perform at its best and prevent disease
Share on X
Vitamin D Background
Vitamin D3, also known as cholecalciferol, is a fat-soluble vitamin that plays a role as a pro-hormone. It Is naturally found in some foods like fish, egg yolks, and beef liver, but humans rely on making it through a process in the skin that requires sunlight exposure. For a full review of vitamin D visit our previous blog.
Vitamin D play a role in multiple aspects of health including bone mineral balance, neuromuscular function, immunity and autoimmune disease, cognitive health, and insulin regulation. Maintaining adequate vitamin D levels can be beneficial when managing or treating many conditions, including:
- Back or musculoskeletal pain
- Osteoporosis
- Frequent colds or flu
- Cardiovascular disease
- Diabetes
- Fatigue
- Seasonal Affective Disorder (depression)
Vitamin D interaction with other nutrients
Calcium and Phosphorous
Vitamin D is necessary for absorption of calcium and phosphorous from food sources in the intestines into the blood as well as regulating the amount of those minerals in circulation. This is a delicate balance, these two minerals are essential for bone remineralization and remodeling (bone building), skeletal muscle contractility, as well as kidney and cardiovascular health.
Vitamin K: K1 vs K2
Vitamin K is a general term for a group of compounds that function as part of the coagulation (blood clotting) pathways that help us regulate blood thinning and prevent risk of bleeding from cuts or injuries. Vitamin K is typically thought of for bleeding disorders, but proper balance may also be useful in the management and treatment of the following conditions:
- Atherosclerosis and ischemic heart disease
- Cirrhosis, hepatitis, and liver disease
- Osteoporosis ad bone loss
- Cancer
Vitamin K naming and structure is one of the most misunderstood topics in nutritional medicine, so let’s spend a few minutes understanding these foundations. Vitamin K is the general term used to describe a group of compounds that share a common ring structure. There are 2 subcategories to keep in mind, K1 and K2.
Vitamin K naming and structure is one of the most misunderstood topics in nutritional medicine. There are 2 subcategories to keep in mind, K1 and K2
Share on X
K1, also known as phylloquinone or phytonadione, is the primary form found in plants. Leafy vegetables are the main source of vitamin K1. This is the form that is utilized in the liver and involved in clotting. This is also the form that interacts with blood thinners like warfarin and why patients on blood thinners are told to carefully monitor their intake of leafy greens.
K2, referred to menaquinones, describes a group of varying chemical structures that are used outside the liver (in the bone and vascular wall). The number of isoprenyl units determine the name of the structure. So, a K2 with 4-isoprenyl units is MK4, and MK7 means there are 7 isoprenyl units. This is the form that is involved in bone health and the focus of kidney-relevant associations of this vitamin.
Vitamin K2 can be found in the diet in multiple forms (for example MK4, MK7, MK8, and MK9) in egg yolks, meats, cheese and other dairy products, natto (fermented soybeans), and seems to be much more readily bioavailable than K1 sources. In addition, menaquinones are synthesized by bacteria in the colon.
Vitamin K2 can be found in egg yolks, meats, cheese and other dairy products, natto (fermented soybeans), and is more readily bioavailable than K1. It is also synthesized by bacteria in the colon
Share on X
Considering what we already know about the impact of the microbiome of kidney health and risk factors, menaquinone might play a larger role in the gut-kidney connection than we’ve been giving it credit for. Though there are multiple factors that contribute to the circulating level of vitamin K in the body, there is speculation that dysbiosis (imbalance of healthy gut bacteria in the gut) may be a significant contributing factor to inadequate circulating levels of K2 contributing to cardiovascular, kidney and bone disease.
Supplementing Vitamin K2
Supplements containing vitamin K are available commercially as K1, MK4, and MK7. However, it’s important to note that safety and efficacy of each form is not equal and has contributed to significant confusion and inaccurate recommendations.
There’s little evidence supporting the supplementation of phylloquinone, or K1. This may be due to poor bioavailability and low rate of conversion to active forms. K2 on the other hand seems to be the superior form, with more recent data supporting the use of MK7.
In older studies, MK4 form seemed to be established as the most efficacious form when it comes to certain conditions like osteoporosis and hepatocellular carcinoma. However, MK7 seems to be more bioavailable and have a steadier blood concentration when compared to MK4 (and K1), meaning more stable blood concentrations and requiring lower doses. Additionally, the added benefit of MK7, particularly in osteoporosis, may in part be related to MK7 inhibition of NF-kB, an inflammatory mediator known to increase disease progression.
Like Vitamin D3, K2 helps to manage calcium deposition. Adequate levels of Vitamin K2 are required to deposit calcium into the bone and to prevent calcium deposition into soft tissue like cardiovascular arteries and the kidneys. So, you see, depletion of K2 along with vitamin D3 can be a risk factor for bone loss, cardiovascular and kidney disease and supplementation of the two combined might yield the best health outcomes.
So depletion of K2 along with vitamin D3 can be a risk factor for bone loss, cardiovascular, and kidney disease and supplementation of the two combined might yield the best health outcomes
Share on X
Manufacturers and companies selling vitamin K supplements often do not reveal whether contains MK-4 or MK-7. Therefore, it’s important to use formulations with full transparency and quality control. This is important for ensuring the highest efficacy, but also for safety concerns surrounding vitamin K supplements and that they may interfere with the stability of anticoagulant medications such as warfarin. This is also why it’s very important to work with your nutritionist or doctor to choose the correct product and dose. If you’re on a blood thinner, never make changes to your diet or supplements without consulting with the healthcare provider monitoring your medication and INR (test for bleeding tendencies).
Studies that examine the benefit of MK-7 supplementation showed a positive effect on cardiovascular calcification. Although MK-7 did not prevent CKD-associated hypertension and hypertrophy, it did demonstrate prevention of calcification of blood vessels. The recommended dose of MK-7 is typically 45μg/d, though doses of 100μg/d have been suggested likely based on studies of the less potent form of MK-4 which, as explained above, is not equivalent.
Although further studies are still underway, it’s safe to say that considering the low risk and cost, it’s worth optimizing vitamin D3 and K2 (MK7) levels in patients with associated risk factors for or diagnosed with CKD. Work with your nutritionist and nephrologist to personalize your intake and optimize your nutrient levels safely and effectively.
The post Vitamin K in Vascular, Kidney and bone Health appeared first on Integrative Kidney.]]>





