This routine should take no longer than an hour. Your kidneys and health deserve an hour from you every morning. I understand many of us have hectic lives, but if you are busy, try to go to bed early and wake up one hour earlier the next morning. Trust me, it is worth it. An hour morning routine beats dialysis any time. This could be the first step in how to heal your kidneys naturally.

Start with water
At night the body loses a lot of water due to sweat and breathing. In fact, in a small study presented at an Associated Professional Sleep Societies meeting in 2009, healthy young men were found to lose ¼ lbs. per hour while asleep. That is about 2 lbs. in eight hours of sleep.
This is why you should start your day with water. The rest of the routine can be done according to your judgment and comfort. Start with drinking two glasses of filtered water if you are not on any water restrictions. This is even more important for patients with polycystic kidney disease and kidney stones. If you are wondering how much water to drink in PKD, check out our calculator here.
Exercise
Exercise for about 20 minutes. The type and duration of exercise depend on an individual’s level of functioning, endurance, and preference. Studies on exercise in chronic kidney disease (CKD) have included resistance training and aerobic activities that use large muscle groups continuously such as walking, cycling, and jogging.
High-intensity interval training (HIIT) was shown to offer superior benefits in individuals with metabolic disease. In the few studies of HIIT in CKD, it was found to be a safe and feasible option for individuals with CKD.
If you are new to exercising, start slow and try to gradually get to 20 minutes of strenuous exercise five to six days a week. It is best to alternate between aerobic exercise and resistance training. If you absolutely can’t fit exercise into your morning routine, you can move it to whatever time that works for you. The benefits are all the same. Consistency is the key here.
Download Your Copy!
Meditation
Meditate for at least 10 minutes. Meditation is a great way to start your day, manage stress, and stimulate your vagus nerve, which connects the brain with many vital organs including the kidneys. There are many ways to meditate and many types of meditations. Mindfulness meditation is the most tested in kidney patients. It has been shown to improve the quality of life, reduce anxiety and depression, reduce sympathetic overactivity, improve sleep, and improve blood pressure.
To practice mindfulness meditation such as Benson’s relaxation technique, sit in a comfortable position or on a meditation cushion, close your eyes, relax your shoulders and muscles, and focus on your breathing. Say a word with every exhalation. Pick a positive word like “gratitude.” As your mind starts racing to distract you from your breath, try to return to the word you chose. Practicing this every day will make you better at it, and you will be able to reap all the benefits with consistency.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Journaling
Journaling is a great way to write your thoughts and plan your day. It has similar benefits to meditation. In fact, journaling was found to reduce stress and depression, boost the immune system, and improve memory among many other benefits. You can write whatever comes to mind. Think of it as a thought download. This should also not take more than 10 minutes.
Meditation and journaling can also be moved to a different time of the day for busy individuals with similar benefits. If you are very busy, consider meditation in the morning and journaling in the evening.
Cold shower
Taking a cold shower has many benefits for kidney patients. It increases endorphins and fights off depression, and it improves metabolism and circulation. Most importantly, it stimulates the vagus nerve and improves renal circulation. It may also soothe itchy skin, which is common in CKD patients. Cold showers help with post-workout muscle soreness. The benefits of a cold shower begin when the water temperature dips to 60 degrees Fahrenheit. This is 40 degrees lower than the typical steamy shower.
Now, I know it will be hard to turn that shower knob from hot to cold in the morning. The way to do it is to start with your hot shower. Then gradually lower the temperature at the end of the shower every day by turning the knob to cold. Give your body time to adjust. You can start with 30 seconds under the cold and gradually increase it to a maximum of three minutes. A quick shower for kidney health takes 10 minutes.
Plant-based breakfast
A good plant-based breakfast can be a super healthy way to start your day. A plant-based or plant-dominant diet has been linked to better kidney health and to slow the progression of kidney disease. There are many options that you can prepare in less than 10 minutes or plan the day before. You can find a few recipes in this e-book.
The bottom line
Your kidneys and health deserve an hour from you every morning. This morning routine can improve kidney health and help slow the progression of early kidney disease. If you’re wondering how to heal kidneys, this routine could serve as the first step, or it could be all you need for heathier kidneys. You can modify it to fit your schedule so that you’re at least doing some of the suggested morning actions every day. If you’ve been wondering how to improve kidney function, this morning routine is an important step in the right direction.
The post How To Improve Kidney Function with a Morning Routine appeared first on Integrative Kidney.]]>
3 years of lifestyle interventions improved exercise capacity and decreased the losses in neuromuscular and cardiorespiratory fitness in CKD patients
In this study, researchers randomized 161 patients with stage 3-4 CKD to either get usual care or usual care plus lifestyle “intervention” for 3 years.
The lifestyle intervention comprised of care from a multidisciplinary team, including a nephrologist, nurse practitioner, exercise physiologist, dietitian, diabetes educator, psychologist, and social worker.
The patients were coached for 8 weeks and then followed for 34 months with a home-based program.
The study did not look at the progression of CKD but it found that a 3-year lifestyle intervention doubled the percentage of CKD patients meeting physical activity guidelines, improved exercise capacity, and decreased the losses in neuromuscular and cardiorespiratory fitness.
It appears that the study mainly focused on exercise. So imagine the benefit of a comprehensive lifestyle modification plan that includes nutrition, exercise, stress management, sleep improvement, and attention to toxin exposure and gut-kidney connection. That’s what we focus on.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Curcumin powder did not improve markers of vascular dysfunction in PKD
This is essentially a negative study.
It demonstrated that Curcumin powder did not improve markers of vascular dysfunction in children and young adults with PKD. The study was conducted for only one year using a dose of 25 mg/kg per day of curcumin.
This is a classic supplement or nutrient study that is usually underpowered or conducted for short periods of time for a disease that takes years or even decades to evolve. Nevertheless, the study proved that short-term use of curcumin is not beneficial for vascular health for young patients with polycystic kidney disease.
Low serum zinc levels were associated with infections in CKD patients
This did not really need research but it is now studied and it is official: Low zinc levels in patients with CKD lead to infection (..well among other things).
This retrospective study analyzed data from 299 CKD patients who had serum zinc levels checked to evaluate anemia. They used the level of 50 mcg/dl as the cutoff between low or “high” zinc values.
Low serum zinc values remained an independent risk factor for infection-related hospitalization. This was especially true for patients taking proton pump inhibitors (PPIs) medications.
Read about the effect of Zinc on kidney health in this blog.
Download Your Copy!
Join here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to here your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post January Research and News appeared first on Integrative Kidney.]]>
Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>
Altered vitamin K biodistribution may decrease the benefit of vitamin K2 supplementation in advance CKD
In this study, investigators sought to determine if there are other causes for vitamin K deficiency in advanced CKD beyond decreased dietary intake. They compared vitamin K uptake and distribution into circulating lipoproteins after a single administration of vitamin K1 plus K2 (MK-4 and MK-7) between patients on dialysis and healthy individuals.
They found that patients with uremia and advanced kidney disease don’t incorporate MK-7 well into HDL and LDL particles compared to healthy individuals. In addition, the combination of a statin and PPI was associated with signs of functional vitamin K2 deficiency in these patients.
In essence, patients with advanced kidney disease may not benefit as well from vitamin K2 supplementation. This highlights the importance of optimizing vitamin K2 status at earlier stages in CKD.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Higher levels of deoxycholic acid were associated with a higher risk of progression in CKD
Deoxycholic acid is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. Intestinal bacteria metabolize the primary bile acid, cholic acid, into deoxycholic acid (DCA).
Researchers studied 3,147 CRIC study participants who had fasting DCA levels. DCA levels above the median were independently associated with higher risks of ESKD and all-cause mortality.
This study highlights the importance of the microbiome and dysbiosis in the progression of kidney disease as we discussed in our blog.
Download Your Copy!
The higher number of medications a kidney patient takes the faster her kidney disease progresses
In a study performed in Japan of 1117 CKD patients under nephrological care, the use of a higher number of medications was associated with an increased risk of kidney failure, cardiovascular events, and all-cause mortality in patients with CKD. This is one of the major reasons we advocate for lifestyle modifications and coaching as the first and major step in the management of kidney disease.
Subscribe here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post December Research and News appeared first on Integrative Kidney.]]>
Low magnesium levels have been associated with a number of adverse events, such as high risk for heart disease. However, little is understood about magnesium and kidney health. Here, we will discuss the potential benefits of magnesium on the kidneys. This is one of two articles on magnesium and kidneys. For more on how to test and treat kidney patients with magnesium deficiency, see part two, “Magnesium Deficiency: Assessment and Management for Better Kidney Health.”

Dietary sources of magnesium
A daily intake of 3.6 mg/kg is necessary to maintain magnesium balance in humans under normal conditions. This is estimated to be between 320 to 420 mg/day (13–17 mmol/day) for adults. Sadly, there has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is due to depletion of magnesium in soil over time. This, along with the rise of ultra-processed food, sodas, and taking medications such as proton pump inhibitors and diuretics that deplete magnesium levels (polypharmacy), has led to rising prevalence of magnesium deficiency.
Traditionally, the highest food sources of magnesium are:
- Leafy greens (78 mg/serving on average)
- Nuts (80 mg/serving on average)
- Pumpkin seeds have the highest level of magnesium per serving (156 mg).
- Whole grains (46 mg/serving on average)
A complete list of foods high in magnesium can be found here.
Can Magnesium Help Kidney Function?
There are many potential benefits of magnesium for kidney health including improving blood pressure control, insulin sensitivity, bone health, vascular health, and preventing kidney stones. Let’s explore the data.
Magnesium and blood pressure control
Magnesium supplementation may help reduce blood pressure (BP) by increasing the production of nitric oxide. Nitric oxide acts as a signaling molecule that helps relax blood vessels, which lowers BP. In fact, a review of 34 studies showed that supplementing magnesium with an average dose of 368 mg per day for 3 months can decrease systolic BP by 2.00 mmHg and diastolic BP by 1.78 mmHg. This supplementation was accompanied by 0.05 mmol/L increase in serum magnesium levels.
Download Your Copy!
Magnesium and insulin sensitivity
Diabetes is one of the major risk factors for kidney disease worldwide. Higher dietary intake of magnesium has been correlated with lower diabetes incidence. A review of 18 studies in people with diabetes showed that magnesium supplements reduced fasting plasma glucose levels. In people who are at high risk for diabetes, magnesium supplementation significantly improved plasma glucose levels after a 2-hour oral glucose tolerance test. These effects are thought to be due to the effects of magnesium on insulin receptors and signaling that allows for improvement in glucose transport and utilization.
Magnesium and vascular health
Magnesium levels have been associated with a lower incidence of cardiovascular disease. In fact, supplementing with magnesium was associated with improvement in vascular flow and endothelial function. Endothelial function refers to the lining of the blood vessels, which is involved in regulating blood vessel health and blood clotting.
Studies in patients receiving dialysis have shown that having a lower serum magnesium level is a significant risk for cardiovascular mortality. Laboratory data show that magnesium inhibits high phosphate-induced calcification of vascular smooth muscle cells. Calcification of arteries is a strong predictor of heart disease and heart-disease-related death.
Magnesium and vitamin D
Magnesium is essential to vitamin D metabolism. Vitamin D that we eat or make in our skin from sun exposure circulates in the blood and is bound to vitamin D binding protein (VDBP). VDBP binding activity depends on adequate magnesium levels. In addition, magnesium is an essential cofactor for the enzymes that activate vitamin D. Studies have demonstrated that magnesium deficiency is associated with impaired vitamin D metabolism.
On the other hand, taking large doses of vitamin D can induce severe depletion of magnesium. This is thought to be due to the overutilization of magnesium. Therefore, adequate magnesium supplementation should be an important part of vitamin D therapy.
Adequate magnesium supplementation should be an important part of vitamin D therapy.
Share on X
Magnesium and bone health
Besides magnesium’s effects on vitamin D metabolism, it is an essential component of hydroxyapatite, an essential component of bone and teeth. In fact, 60% of total Mg is stored in the bone. Low magnesium intake was found to be associated with lower bone mineral density in postmenopausal women. Magnesium deficiency contributes to osteoporosis directly by acting on crystal formation and on bone cells and indirectly by impacting the secretion and the activity of parathyroid hormone (PTH) and by promoting oxidative stress and inflammation.
In addition, a review of 8 studies looked at magnesium and chronic kidney disease (CKD). The study investigated magnesium supplementation on parameters of CKD-related mineral bone disease (CKD-MBD). Mg supplementation improved PTH levels and carotid intima-media thickness (CIMT). Low serum Mg levels were also found to impact PTH and worsen osteoporosis in CKD patients, particularly with diabetes.
Magnesium and kidney stones
Mg acts as an inhibitor of calcium oxalate crystallization and stone formation in the urine. It also decreases the absorption of dietary oxalate in the gut. Mg supplementation in patients with kidney stones was found to decrease the incidence of stone formation even in patients without signs of Mg deficiency.
Magnesium as a phosphate binder
Hyperphosphatemia (high phosphate level) is common in advanced kidney disease. Many kidney patients with stage 4 and above use binders that bind phosphate (or “phosphorus,” as it is commonly known) in the food and prevent it from getting absorbed. High phosphate levels have been associated with poor bone and vascular health in kidney patients. In fact, higher dietary phosphate load can be seen in earlier stages of CKD, and it can do harm even before it is detected.
Magnesium carbonate has been successfully used as a phosphate binder. Magnesium based phosphate binders were also found to reduce vascular calcifications in rats with kidney disease. Iron-magnesium hydroxycarbonate was also studied and found to be well tolerated and can effectively lower phosphate levels in dialysis patients. It is essential to know that most of the magnesium used as a phosphorus binder will not be absorbed.
The bottom line on magnesium and kidneys
Magnesium is essential to many biological functions. It has many health benefits for kidney, bone, and vascular health. Optimizing magnesium status is, therefore, an important step in the integrative approach to kidney health. In part two of this blog, “Magnesium Deficiency: Assessment and Management for Better Kidney Health,” we will discuss practical steps for figuring out a person’s actual magnesium status, the best form of magnesium to take, and the dose I recommend for each condition.
The post Magnesium and kidneys appeared first on Integrative Kidney.]]>We combed through multiple medical journals looking for the latest research on the Integrative approach to kidney health. We know your time is valuable so we curated and summarized these studies for you. Welcome to the November edition of InKidney Research and News.

Is phosphorus toxic to the kidneys?
In this detailed study, researchers found that excessive intake of phosphate (phosphorus) increases a substance called FGF-23. One of FGF-23 main tasks is to maintain phosphorus balance in the blood. It does that by increasing the excretion of phosphorus in the urine. This study found that the presence of high phosphorus in the kidney tubules causes them to bind with calcium forming tiny crystals that are very toxic to the tubular cells and leads to fibrosis. Investigators found that a level of FGF-23 above 53 pg/mL is associated with progression of kidney disease in humans.
Coffee and caffeine consumption and the risk of kidney stones
Investigators used Mendelian randomization to study the link between coffee consumption and the risk of kidney stones. They looked into the UK and Finland genetic databases and tried to find the association between coffee drinking genotype and kidney stone phenotype.
The study found that genetically predicted coffee and caffeine consumption was associated with a lower risk of kidney stones. This supports other observational studies that indicated the same.
But of course, the study means that consumption of black coffee is associated with decreased risk for kidney stones. It does not addresses latté or triple caramel Frappuccino. So, as always drink coffee in moderation.
Download Your Copy!
One in every 5 CKD patients receives a nephrotoxic medication
This study published ahead of print looked into the use of nephrotoxic medications in the US and Sweden. They studied a total of 74,135 patients with stage 3-5 CKD. In one year, one in every 5 patients with CKD, received a nephrotoxic medication.
Subscribe here to receive a FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
The post November Research and News appeared first on Integrative Kidney.]]>
Lifestyle modifications do indeed prevent CKD and delay its progression
This study is coming from Japan. Researchers found that two years of healthy lifestyle choices prevent against the development of kidney disease and protein in the urine. Lifestyle modifications also slowed the decline in kidney function.
They studied 451,534 people (277,494 men and 174,040 women) aged 20–79 years. Healthy lifestyle choices in this study were defined as: 1. No smoking 2. Healthy eating habits, and 3. Low BMI
Now, we all know that there is more to lifestyle modifications than this but this is definitely a start.
Is Urea by itself toxic?
The authors in this study published in NDT definitely think so. They noted that the accumulation of uremic toxins is a risk factor for mortality and accelerated atherogenesis in patients with CKD. The researchers found that a post-translational protein modification called carbamylation is a predictor of CKD progression. They also noted that urea by itself promotes the carbamylation of proteins.
So urea is not just a benign marker and should be classified as a uremic toxin. The level of protein carbamylation may be used as a marker for risk stratification of CKD outcomes.
Download Your Copy!
Magnesium, bones, and kidneys… What?!
This study is coming from the dialysis world. It’s a meta-analysis that examined the effects of magnesium supplementation on chronic kidney disease mineral bone disease (CKD-MBD). As the researchers mentioned, magnesium supplementation in dialysis patients is controversial. However, this study showed that it helps by regulating calcium and parathyroid hormone and it decreases carotid intima-media thickness.
This is in the dialysis population. So paying attention to magnesium balance and supplementation may have a much better role in bone and vascular health in earlier stages of CKD. Optimizing vitamin D, vitamin K2, vitamin A, calcium, phosphorus , and magnesium is very important to improve bone and vascular health in CKD patients.
Subscribe here to receive a monthly update on the latest research in Integrative Nephrology.
The post October Research and News appeared first on Integrative Kidney.]]>
As mentioned in a previous blog, the integrative approach to many kidney diseases should be personalized, yet comprehensive. It starts with understanding an individual’s genetic predisposition, current and previous lifestyle choices and exposures, triggers of the current disease process, and unique nutritional status. In addition, it’s important to identify an individual’s spiritual, mental and emotional state. This type of approach can help the provider develop a personalized plan that includes attainable lifestyle modifications for the patient.
The ability of the patient to make these changes is the cornerstone of this tailored management plan. Although implementing and adhering to lifestyle modifications can be challenging, it’s not a reason to quit. In order to understand how to overcome these challenges, we talked about the brain and implementing changes in this blog. Understanding synaptic pruning and neuroplasticity can help us in this process.
Implementing Lifestyle Modifications
Now let’s utilize what we discussed above to help patients implement the lifestyle modifications we recommend for those with kidney disease. Of course, willpower varies from person to person. So, what we are suggesting here might not be for everyone, but we think the information is helpful.
Think about it
Now that we know that the brain is hardwired to follow habits, we can approach lifestyle habits without judging or beating ourselves up. The first step in implementing lifestyle modifications is to think about these changes. It will help to write down or talk to someone about the thoughts associated with these changes. Think about lifestyle habits that are currently working and any that are not in order to help identify patterns. Identifying these patterns can be a good starting point to help the brain perceive the need for change. Remember, the brain wants to be efficient and it will always default to the “pre-wired” unhealthy habits if we let it. So, spending extra energy and utilizing that pre-frontal cortex to teach the brain new things is required in the beginning.
Pick one thing
Once patterns have been identified, it’s best to implement one change at a time to avoid overwhelming the brain. It can be helpful to work with a provider or coach to devise a plan of action. Oftentimes, it is beneficial to start with easier changes, as successful implementation of the new habits can help build momentum. For example, an individual may decide an easy change would be to not snack or drink sugary beverages/alcohol after dinner. It is important to note that what is easy for one person may not be as easy for another, so personalization of an action plan is critical.
Plan it
Having a plan can make or break a person’s ability to stick with the new habits. It’s easy for the brain to default to old habits when there isn’t a plan in place. For example, using the weekend to plan out and/or prepare meals for the upcoming week can help ensure healthy eating habits during the week for someone who orders take-out when they get home tired after work. Taking the guess work out of things can set someone up for success.
Learn from mistakes
Let’s face it, mistakes can happen. They don’t indicate failure or give reason to quit. There may have been some variables that were not accounted for. It’s important to learn from mistakes and improve the planning process for the future.
Repeat, repeat, repeat
The most important step in making these modifications permanent is repetition. Repetition creates new patterns and neural pathways via synaptic pruning so that the brain will default to them instead of the previous unhealthy habits. The amount of time this takes depends on the individual’s willpower and commitment. This is not going to be easy but, as they say practice makes perfect.
The Bottom Line
Implementing lifestyle modifications is crucial in the fight against the chronic kidney disease epidemic and foundational to the integrative approach to kidney health. It can be a challenging process but understanding the brain’s efficacy and patterns can help individuals make the changes needed to lead a healthier lifestyle.
The post Implementing Lifestyle Modifications for Kidney Health appeared first on Integrative Kidney.]]>
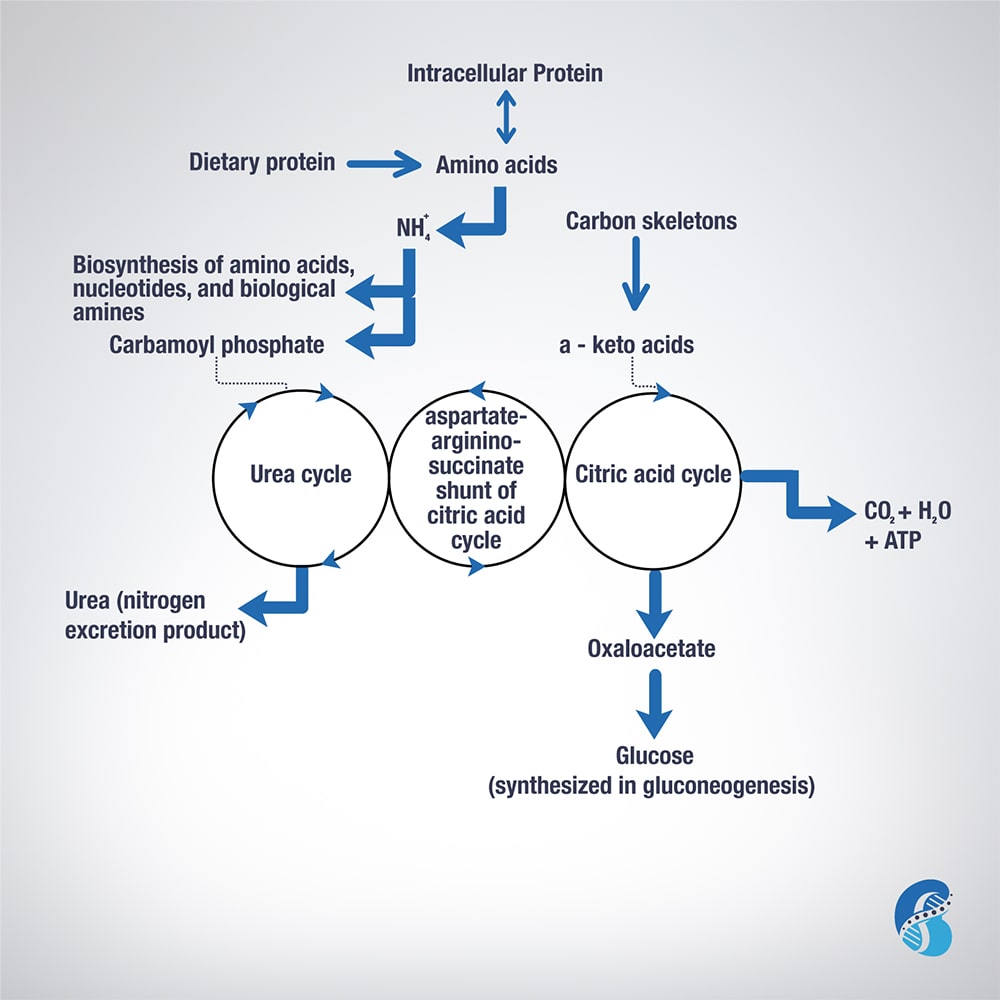
Excess AA in a protein-rich diet is metabolized through a process called transamination. This involves the removal of the nitrogen (or the amino group NH2 side chain) and replacing it with a keto group carbon structure forming a ketoacid or ketoacid analogue.
Apart from lysine and threonine, every EAA has a keto-analogue. The ketoacid then undergoes oxidation to carbon dioxide, water, and a three to four-carbon unit that can be converted by the liver into glucose (by gluconeogenesis) or into ketone bodies for energy.
The conversion of an amino acid to ketoacid depends on the enzyme aminotransferase. This enzyme is reversible (meaning it can convert amino acid to ketoacid or a ketoacid to amino acid). The speed and direction of this conversion depends on the amount of amino acids available, the amount of keto-acids formed, and the availability of the cofactors needed to power the process. These cofactors are nutrients that facilitate the reactions. Vitamin B6 is a major cofactor for this enzyme. Other cofactors needed in the amino acid breakdown (catabolism) include biotin, folate, and B12.
Now, remember that amino group (-NH2)? That can be used in the biosynthesis of non-essential AA and the excess ends up metabolized in the urea cycle to form urea and other nitrogen waste products that are excreted by the kidneys. It is worth noting that the AA arginine acts as a regulator of the urea cycle. Remember, arginine is essential for the production of nitric oxide, which plays an important role in blood pressure management. Issues with the urea cycle and nitric oxide balance may explain why kidney patients have blood pressure problems and may not feel well with arginine supplementation.
Overall, this process is summarized in this figure.
The post Biochemistry of Amino Acids Metabolism appeared first on Integrative Kidney.]]>
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Kidney stone formation (also called urolithiasis or nephrolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. Anyone who’s experienced them can attest that stones are often very painful. When left untreated, they may contribute to more serious conditions including urinary tract obstruction and permanent kidney damage or CKD.
Read more about the etiology and prevalence of kidney stones here.
Conventionally, the approach to treatment is a multi-pronged approach and may include medication, dietary and lifestyle interventions, surgical removal, and using ultrasonic waves to break up stone. However, dietary guidelines tend to focus on stone composition instead of on the underlying pathology. In this series, we’ve been discussing the combination of upstream risk factors that impact risk of stone formation.
Factors that impact the integrative approach to kidney stones include:
- Type of stone
- Environment
- Diet
- Hydration and electrolyte balance
- Microbiome and gut health
- Genetics
Diet is a topic that deserves a deeper dive! We’ll focus on it here today.
Dietary Approach to Kidney Stone Prevention
The Diet Controversy and Kidney Stones
Dietary interventions are common in addressing kidney stones. The reality is, we need a more comprehensive approach when it comes to preventing kidney stone formation and kidney damage. Dietary approach to kidney stone prevention is one aspect of this comprehensive integrative approach.
First, let’s start by outlining the impact of the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
- Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
- Increased intake of processed/refined foods like cereals, crackers, baked goods, etc.
- Processed, fried, conventionally raised, high-nitrate animal protein
- Low intake of fiber and fresh produce in general
- A “beige” diet (low in phytonutrients and antioxidants) lacking color and low in enough variety of colorful fruits and vegetables
- Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
Interestingly, studies have shown that eating more fresh produce is protective from kidney stone formation. When we consider that the SAD is low in protective foods and the key nutrients found to be helpful in preventing urolithiasis. Let’s examine these factors that impact kidney stones in more detail.
Macronutrient Balance for Kidney Stones (Protein, Fat & Carbs)
Protein
For a long time, it was assumed that stone formation was at least in large part due to excessive protein intake. As a result, those at risk for stone formation were instructed to eat a low protein diet to prevent urolithiasis. However, there are underlying factors that cause a domino effect impacting how the products of protein breakdown lead to stone formation. This includes micronutrient and electrolyte balance, metabolic disorders, digestive abnormalities, and of course dysbiosis (we’ll discuss these in detail in another blog).
It’s true that excessive protein intake can lead to more acidic urine and increased uric acid production (a risk factor for uric stone formation). Elevated protein metabolism alone isn’t a problem in isolation, there’re other upstream factors to consider that make certain individuals more susceptible to stone formation. Furthermore, the source of protein seems to be significant. Animal protein sources seem to play a bigger significance in risk of stone recurrence, while plant-proteins might be protective.
Carbohydrates and Fiber
Though excessive consumption of carbohydrates isn’t recommended, excessive restriction of carbs in people prone to kidney stones should be avoided. We suggest avoiding sources of simple sugars, fructose and high fructose corn syrup (that includes sugar sweetened beverages, pastries, sodas, and even the so-called healthy sweetener agave). Fructose consumption in particular seems to increase production of excess uric acid and simultaneously reduces its excretion in the urine.
Instead, we want to opt for more nutrient-dense, high-fiber, low-glycemic carbohydrates, including root vegetables, berries, whole grains and legumes. Increased consumption of fiber helps to promote improved microbiome health and diversity, which may play a significant role in kidney stone formation risk. Furthermore, there is interesting research that indicates that individuals who aren’t getting enough dietary vitamin C to meet their needs while on a low-carb diet may increase their risk for developing certain types of kidney stones.
This begs the question, what other nutrient deficiencies might contribute to stone formation when macronutrients might not be optimized?
Fats and Essential Fatty Acids
In order to balance a moderate protein and carb intake, some individuals might benefit from increasing calories from fat. Healthy fats include omega 3 sources like fish and fish oil, nuts, seeds, olive oil, and avocado. Grass-fed ghee might be especially beneficial due to a combination of anti-inflammatory profile, vitamin A, and gut healing benefits of the butyric acid component.
Independently, increased fat intake has not been associated with kidney stone formation. However, when coupled with a SAD diet full of simple carbohydrates and fried and processed foods, and excessive protein intake that seems, at least in part, to be a contributing factor. At the same time, when eating a high-fat diet as part of a ketogenic approach, excessive restriction of carbohydrates seems to contribute to stone formation in individuals with existing risk factors. In those cases, it’s recommended to work with a nutritionist to ensure that you’re balancing your macronutrients effectively to avoid kidney stones.
Micronutrient Balance
Fat-soluble vitamins
We can’t talk about dietary approach to kidney stone prevention without discussion fat soluble vitamins. Fat soluble vitamins include vitamins A, D, E, and K. Deficiencies in D, A and K in particular have been associated with kidney stone formation. This might at least in part be due to the fact that these vitamins play a major role in calcium metabolism and bone mineralization. When D and K are deficient, calcium from diet is deposited in arteries instead of in bones, leading to calcification and stone formation.
In fact, vitamin K depletion might be an independent risk factor for kidney stone formation. When individuals form kidney stones, a vitamin-K dependent protein (matrix Gla protein) is secreted in inactive form. This protein when activated with vitamin K can inhibit the growth of calcium oxalate crystals. When vitamin K isn’t available, more crystals may form.
To improve dietary intake of these nutrients, foods like eggs (especially the yolks), organ meats, ghee, natto (fermented soybeans), and full-fat cheeses (assuming you’re able to tolerate dairy) can be helpful to include in your diet. Daily sun exposure (at least 20 minutes a day) can also help promote increased vitamin D levels. However, supplementation is often indicated. Issues with absorbing fats and fat-soluble vitamins due to digestive issues are common, a nutritionist or a clinician trained in Integrative and Functional Medicine can help you determine if supplementation is needed.
Oxalates
Oxalates are front and center in the dietary approach to kidney stone. In fact, calcium oxalate stones comprise most kidney stones (75%). There’s some evidence that limiting consumption of oxalates can help reduce stone formation. Food containing high levels of oxalates tend to be generally considered healthy, including spinach, beets, nuts and seeds, to name a few examples.
What makes these foods healthy for some people, and contribute to stone formation in others?
One reason might be inadequate calcium intake from the diet. Food sources like broccoli, sardines and canned salmon, and leafy greens like kale and collard greens can be good low-oxalate, calcium-rich food sources. When consumed in adequate amounts, calcium binds to oxalates in the intestines, preventing it from being absorbed and reducing formation of stones.
Another important consideration is the composition of the microbiome. When oxalate-degrading bacteria in the gut is inadequate, it can increase risk of developing kidney stone disease. More about that in a future article.
Vitamin C
Ascorbic acid has been associated with formation of kidney stones. Excessive intake of vitamin C might increase oxalate production. Some studies have shown higher intake of vitamin C is associated with increased risk of kidney stones, but only in people predisposed to forming oxalate-stones. Regardless, caution should be used in supplementing vitamin C within the context of the larger, integrative picture.
Phytate
Phytate, sometimes called phytic acid, is an antioxidant compound found in certain foods like beans, lentils, nuts, and seeds. Some have claim that phytate intake might prevent the absorption of certain nutrients, but actually it might be protective for kidney stone! One study found that women who consumed more phytate had reduced risk of kidney stone formation. Other studies have come to the same conclusion. This might account to the benefit of eating these particular foods, which happen to be good plant-based protein sources, in place of animal protein.
Salt and Other Electrolytes
Calcium is a controversial topic when it comes to risk of kidney stone formation. Surprisingly, higher intake of dietary calcium (from food sources like leafy greens, salmon, and legumes, for example) is associated with lower risk of stone formation. On the other hand, taking calcium supplements may be linked with increasing risk of kidney stone formation (this might be gender specific to men).
Furthermore, the conversation often fails to address the nuance of calcium consumption in the form of dairy, which might not be superior over plant-based forms of calcium, or to account for the impact of vitamin D deficiency.
Magnesium is another electrolyte that is often overlooked. Magnesium is part of hundreds of the body’s biochemical reactions, so adequate intake is essential. Furthermore, ensuring adequate intake of magnesium can help prevent the formation of calcium oxalate stones by destabilizing the bond between the two compounds causing them to split.
Food sources of magnesium include swiss chard, spinach, almonds, and pumpkin seeds. Many of those foods might be contraindicated in a low-oxalate diet, so supplementation might be indicated before gradually adding those foods back as part of a comprehensive approach. Supplementing with chelated forms of magnesium (like magnesium citrate, malate, or bisglycinate) or making time for an epsom salt bath (magnesium sulfate in the salts absorbs through the skin).
But the most important electrolyte in the dietary approach to kidney stone is sodium. Excessive sodium (salt) consumption has been associated with stone formation, particularly when fluid intake is inadequate. High sodium excretion increases calcium deficiency by increasing excretion through the urine. Reducing excessive intake of sodium, especially from processed foods like cured meats, can be helpful, along with ensuring adequate intake of potassium-rich foods like broccoli, leafy greens, squash, potatoes, mushrooms, bananas, cantaloupe, and grapefruit to name a few.
More about the impact of electrolyte balance on kidney stone formation in a future blog.
Water (the forgotten macronutrients)
Inadequate water intake is one of the most significant contributing factors to kidney stone formation. The process of excretion of oxalate consumes salt and water, as a result dehydration is more likely.
Taking mineral compounds containing citrate (like magnesium citrate or potassium citrate) can prevent the formation calcium oxalate and uric acid stones while balancing mineral needs. Adding lemon juice or apple cider vinegar (4oz per day) to water might also help prevent urolithiasis through the same mechanism. Your Integrative or Functional Medicine clinician can help create a plan that best fits your needs.
The Bottom Line
Multiple factors impact kidney stone formation, including dietary factors. It seems there’s no single food or food component that causes stone formation. Rather, there are likely a combination of factors including nutrient depletion, dehydration, and poor food choices combined with environmental factors, genetics, and gut integrity and microbiome balance which combined play a role.
Therefore, the integrative approach to addressing kidney stones must account for the constellation of these factors. Practitioners working with individuals to prevent kidney stone formation should formulate a personalized approach that modifies all relevant components in their integrative approach. We’ll explore these connections in future blogs in this series.
The post Dietary Approach to Kidney Stone Prevention appeared first on Integrative Kidney.]]>





