Polycystic kidney disease (PKD) is the most common inherited kidney disease affecting one in 800 people. There have been several advances in our understanding of PKD since our last blog. In a previous blog, we discussed the genetics of PKD and how genetic variants translate into a disease. In this blog, we will discuss recommendations for water, salt, and caffeine intake to reduce cyst growth. You will find out if ketogenic diet or intermittent fasting is the best diet for polycystic kidney disease.
PKD at the Cellular Level
To determine the best PKD diet and lifestyle recommendations, we must understand what is happening at the cellular level. In essence, abnormalities in PC1 or PC2 proteins will activate two pathways inside the cell. First, it activates the cAMP pathway which regulates fluid transport. Second, it changes the way cells make energy from sugar (called glycolysis). When PC1 or PC2 proteins don’t work well, they switch from aerobic (with oxygen) to anaerobic (without oxygen) glycolysis. This is similar to the shift that occurs in cancer cells.
While anaerobic glycolysis produces less energy from glucose, it is faster than aerobic glycolysis. This faster energy production allows the cells to grow faster. However, it also leads to a critical dependence on glucose.1
How Much Water to Drink in PKD
Vasopressin, also called anti-diuretic hormone (ADH), is a hormone that is produced by the brain and excreted by the pituitary gland. It plays an important role in the body’s fluid and sodium balance, blood pressure, and kidney function. ADH was found in several studies to promote cyst growth by stimulating the production of cAMP. Further proving the role of ADH in polycystic kidney disease, cyst growth was inhibited in both forms of PKD by administering ADH receptor blockers (medications that block ADH).
Dehydration can increase ADH and make cysts worse. Drinking large amounts of water leads to a significant decrease in the progression of PKD. The optimal amount of water to drink in PKD patients depends on the amount of salt and food they eat. The simplest way to assess this is by checking a 24-hour urine osmolality. You can use our calculator to assess the amount of water you should drink daily based on your 24-hour urine osmolality. Alternatively, measuring first morning urine osmolality can be used. The target for that is keeping the level less than 280 mOsm/kg.
In addition to dehydration, other factors that lead to the production of ADH include stress, pain, and surgery. These factors, therefore, can indirectly lead to cyst growth and further harm kidney function.
Download Your Copy!
Polycystic Kidney Disease and Caffeine
In the laboratory, caffeine increased cAMP accumulation in the cells. Yet, in human clinical trials, caffeine intake was not directly associated with kidney size in patients with PKD. An analysis in one clinical trial found no significant association between the consumption of caffeine from coffee and tea on the progression of ADPKD.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Calorie-restricted and Keto Diets for Polycystic Kidney Disease
Kidney cysts thrive on sugar or glucose, as discussed earlier. Therefore, calorie-restricted and ketogenic diets can be beneficial for patients with PKD. Ketogenic diets induce a state that resembles starvation by providing high fat content and low protein and carbohydrate content. Time-restricted feeding such as intermittent fasting can have similar effects. Studies on animals demonstrated that ketosis could decrease cyst growth in PKD. These effects were thought to be mediated by a strong inhibition of mTOR.2
It is worth mentioning here that metformin can activate AMPK and is currently being studied in PKD.
To our knowledge, there is no published human study to confirm the effect of ketogenic diets and intermittent fasting on PKD. However, given the strong benefit of ketosis on cyst growth, we generally recommend a plant-based ketogenic diet or intermittent fasting in addition to a large water intake for patients with PKD.
Dietary, Lifestyle, and Water Recommendations for PKD
The phrase “our genes are not our destiny” describes how nutrition and lifestyle factors can have a positive impact on genetic expression for certain diseases. Drinking large amounts of filtered water, restricting salt intake, a ketogenic diet, or intermittent fasting can have significant benefits in patients with PKD. So far, limiting caffeine is not necessary to improve PKD, but it has other health benefits. Finally, stress reduction techniques can help to lower ADH and reduce kidney cysts.
Ongoing Studies of Diet and Lifestyle PKD Treatments
- PREVENT-ADPKD trial is looking at water intake and the progression of ADPKD. It should be released in late 2021.
- DRINK trial is investigating water intake in ADPKD. It should also be released in late 2021 or early 2022.
- METROPOLIS trial is comparing Metformin and Tolvaptan for the treatment of ADPKD. It should be completed in early 2022.
- Time-restricted feeding in ADPKD is currently recruiting subjects and is estimated to be completed by end of 2022.
- Keto-ADPKD trial is looking at how the ketogenic diet affects the progression of ADPKD. It should be completed in mid-2022.
- Curcumin Therapy to treat vascular dysfunction in ADPKD was completed in late 2020. It should be released soon.
1 It is noteworthy that glycolysis is inhibited by a cellular messenger called AMP-activated protein kinase (AMPK). It is activated by another messenger called mTOR. Metformin activates AMPK.
2 Activation of AMPK and inhibition of mTOR can theoretically inhibit glycolysis in cells that line the cysts in PKD. However, studies that looked at medications that inhibit mTOR such as sirolimus, did not show any benefit in PKD.
The post Diet and Lifestyle Modifications in PKD appeared first on Integrative Kidney.]]>

Hereditary Kidney Disease: The Genetics of PKD
PKD is a group of two genetic disorders that affect the kidneys and cause the formation of multiple fluid-filled cysts of various sizes. As these cysts grow, they squeeze and destroy normal kidney tissue, eventually leading to loss of kidney function. PKD is an inherited disease, meaning it’s passed down genetically from parent to child, just like eye or hair color. Polycystic kidney disease can be autosomal-dominant (ADPKD) affecting 50% of offspring. Or it can be autosomal-recessive (ARPKD), affecting 25% of offspring.
Mutations in one of two genes (PKD1 or PKD2) account for most cases of ADPKD. Polycystic kidney disease 1 gene (PKD1) mutations are the most common. About 80 percent of patients affected by ADPKD have a PKD1 mutation. PKD2 gene is the cause of up to 20% of ADPKD cases and it is usually associated with milder course.
The PKD1 and PKD2 genes provide the blueprints for important kidney and liver proteins called polycystin-1 (PC1) and polycystin-2 (PC2). These proteins are crucial for the structure of the kidney’s tubular cells, which filter and clean the blood. PC1 and PC2 influence healthy growth and fluid secretion in these cells. However, in people with hereditary kidney disease, abnormal genetic blueprints lead to the production of abnormal proteins. When these kidney proteins don’t work properly, cysts accumulate and damage the kidney.
Cysts that form in polycystic kidney disease usually occur when the cells lining the tubules of the kidney start growing out of control (called proliferation). These outgrowths bulge and eventually separate into cysts. As the cysts grow, they transport fluid across their lining, forming a fluid-filled sack much like a balloon. So, when we think about PKD, we should remember two processes: cell proliferation (cell growth) and fluid secretion into the cysts.
Truncating vs. Non-truncating mutations
Sometimes a change in the DNA sequence of a gene results in the creation of an “early stop.” This early stop functions to end the translation of the gene into a protein in our cell factory. This produces a shortened or truncated protein. This type of variant can have serious functional consequences. They are called truncating mutations.
The relationship between genetic variants and prognosis in PKD is not completely understood. In a study that looked at the “renal survival” in 741 patients with ADPKD, PKD2 mutations were associated with approximately 20 years longer survival than PKD1 mutations. In addition, the type of PKD1 mutation, not its position, correlated strongly with renal survival. The median age at onset of kidney failure was 55 years for carriers of a truncating mutation and 67 years for carriers of a non-truncating mutation. This observation allows the integration of genic and allelic effects into a single scheme, which may have prognostic value.
This points to the importance of genetic testing even in a genetic disease with an obvious clinical presentation such as PKD. Identifying patients with truncating mutations may help selecting those who require more aggressive therapies.
PKD at the Cellular Level
To determine the best PKD diet and lifestyle recommendations, we must understand what is happening at the cellular level. In essence, abnormalities in PC1 or PC2 proteins will activate two pathways inside the cell. First, it activates the cAMP pathway which regulates fluid transport. Second, it changes the way cells make energy from sugar (called glycolysis). When PC1 or PC2 proteins don’t work well, they switch from aerobic (with oxygen) to anaerobic (without oxygen) glycolysis. This is similar to the shift that occurs in cancer cells.
While anaerobic glycolysis produces less energy from glucose, it is faster than aerobic glycolysis. This faster energy production allows the cells to grow faster. However, it also leads to a critical dependence on glucose.1
In the next blog we will discuss the 2021 update about diet and Lifestyle Treatments to Improve Polycystic Kidney Disease (PKD).
1 It is noteworthy that glycolysis is inhibited by a cellular messenger called AMP-activated protein kinase (AMPK). It is activated by another messenger called mTOR. Metformin activates AMPK.
The post Polycystic Kidney Disease Genetics appeared first on Integrative Kidney.]]>
We discussed the effect high intake of animal protein has on kidney health in a previous blog. According to the National Institute of Health, the Dietary Reference Intake (DRI) of protein for the average person is 0.8-1.6 g/kg/day (or 0.36-0.75 g/lb/day). However, according to the Kidney Disease Improving Global Outcomes (KDIGO) guidelines, kidney patients should limit their protein intake. A low protein diet (LPD) by the KDIGO guidelines is 0.8 g/kg/day (or 0.36 g/lb/day). That means a kidney patient who weighs 150 lbs (68 kg) has a dietary protein target of approximately 54 grams of protein per day (0.8g/kg x 68kg). Furthermore, it’s suggested that at least half of that comes from plant-based sources. Protein intake, along with other related nutrients, should be tracked closely through nutrient intake analysis, ideally by a nutritionist.
Different dietary protein regimens have been proposed in the literature for patients with Chronic Kidney Disease (CKD):
- A low-protein diet (LPD), with protein target of 0.6-0.8 g/kg/day;
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with essential amino acids, or
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with nitrogen-free keto-analogues of essential amino acids.
The controversy in the literature, however, has always been whether or not lowering the targeted protein intake puts patients at risk for nutritional deficiency, including essential amino acids and other nutrients often found in high protein foods like folate, B12, carnitine, and zinc. And in that case, is supplementation among these patients necessary and useful to maintain adequate nutrition?
Let’s focus on amino acids, but first let’s review.
What are essential amino acids?
Amino acids (AA) are the basic organic building blocks of protein. There are a total of twenty-two AA, each composed of a core structure made of nitrogen, carbon, hydrogen and oxygen, and variable side chain groups unique to each AA.
Of the twenty-two AA, there are nine essential amino acids (EAA). They’re called “essential” because the human body cannot make them, and therefore we must get from our diet. These EAA are:
- Histidine
- Leucine
- Isoleucine
- Valine
- Lysine
- Threonine
- Methionine
- Phenylalanine
- Tryptophan
The remaining thirteen AA are termed “unessential” not because they’re not important, but because the body can make them through various biochemical processes as long as the necessary building blocks are available from the diet.
When restricting protein intake in kidney patients, it is important that the patient receive adequate intake of EAA since adequate levels of these essential nutrients can only be maintained through food. This can be challenging to keep balanced, which is why we suggest working with a nutritionist to track and support adequate intake.
Using ketoacid analogues with very low protein diet for kidney health
To understand the relevance of ketoacid analogues in CKD, we need a biochemistry lesson in AA metabolism. Biochemistry isn’t everyone’s thing, so we summarized it here.
One thing to remember about AA metabolism is that it requires cofactors which are nutrients that facilitate the reactions. Vitamin B6 is a major cofactor here. Other cofactors needed in the amino acid breakdown (catabolism) include biotin, folate, and B12.
How do ketoacid-analogue supplements help in kidney disease?
Theoretically, the process described above can be used in reverse to reduce circulating nitrogen and the formation of nitrogen waste products (i.e., like urea), and, therefore, decrease the burden on the diseased kidneys. Supplementing ketoacid analogues of EAA in kidney patients on a LPD and VLPD, might simultaneously achieve two goals: 1. It closes the gap on adequate EAA nutritional need 2. It decreases the toxic nitrogen waste products contributing to disease complications. There is also some evidence that certain analogues can decrease protein breakdown in the body.
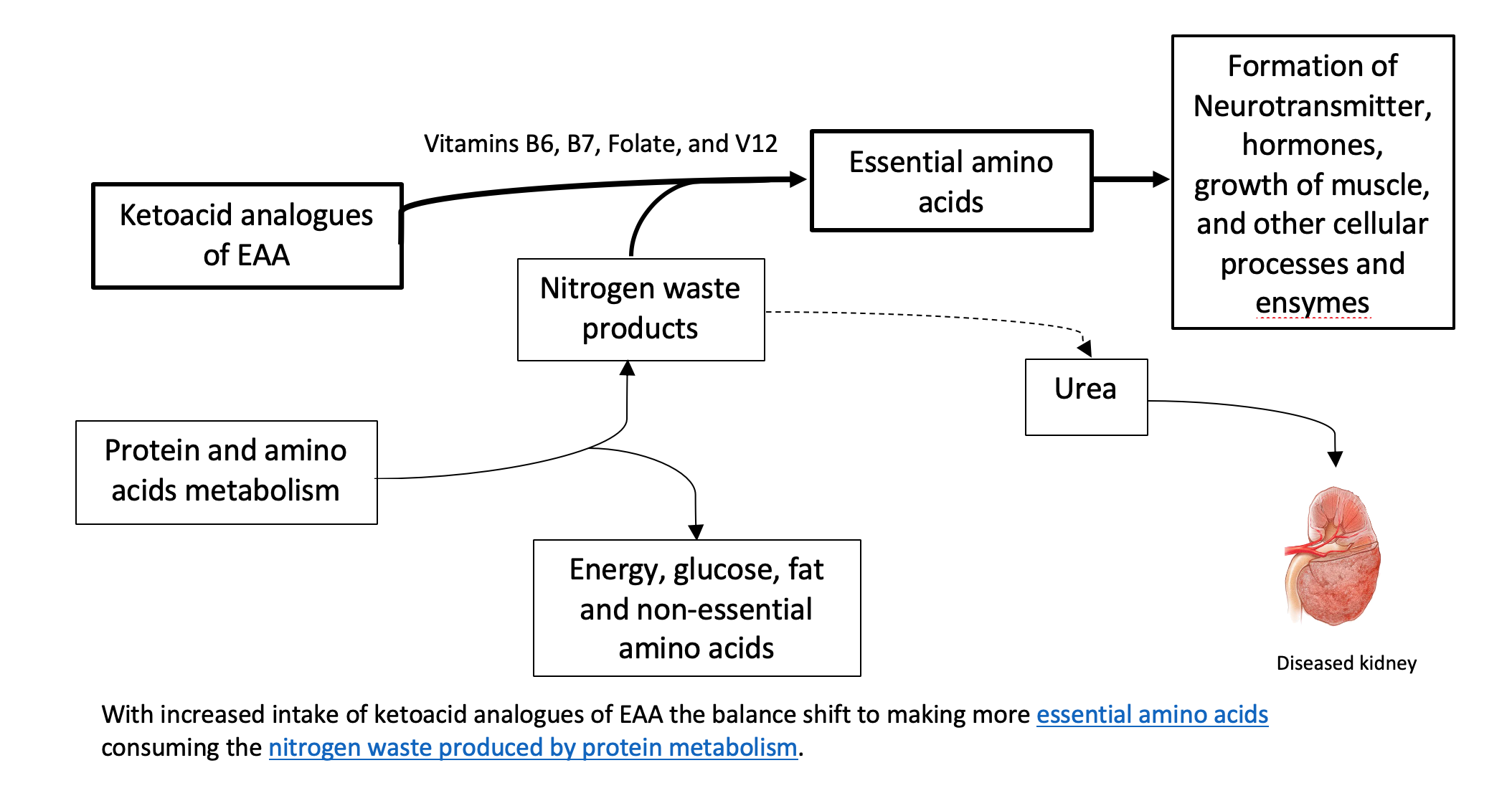
Some studies have demonstrated that very low protein diets supplemented with ketoacid analogues decrease waste product generation in kidney disease and prevent protein-energy decline. Supplementation of keto-analogues in kidney patients was found to decrease protein in the urine as well as improve bone health, insulin sensitivity, and blood pressure control. In addition, these dietary protein restrictions with keto-analogue supplements have been found to delay symptoms of kidney failure and the need for dialysis.
However, one of the major concerns of supplementing ketoacids in kidney patients on VLPD is that it does not address the other micronutrients and cofactors that need to be supplemented. it also does not take into account the carbohydrate and healthy fat content of a healthy diet.
Ketoacid analogue preparations
There are a few different preparations available in Europe, but only one available in the US. If one looks at the content of these preparations, it will become obvious that the kidney patient will need several pills of ketoacid daily to achieve an adequate daily dose. This makes this therapy very expensive and impracticable.
Luckily, there’s a way to use diet to balance macronutrients to induce the same effect. A low protein diet, with the majority of protein coming from whole food plant sources. When consuming animal protein, we recommend organic and grass-fed sources. Strictly vegetarian patients can still get adequate sources of EAA if they plan their diet well with the help of a nutritionist. If AA supplements are determined to be needed, one should aim to achieve the recommended daily intake from a trusted source (with good quality control). According to the World Health Organization, the recommended daily dose of each of the EAA is as follows:
| Essential Amino Acid | Recommended daily intake |
| Lysine | 30 mg/kg/day |
| Leucine | 39 mg/kg/day |
| Isoleucine | 20 mg/kg/day |
| Valine | 26 mg/kg/day |
| Threonine | 15 mg/kg/day |
| Phenylalanine | 25 mg/kg/day |
| Tryptophan | 4 mg/kg/day |
| Histidine | 10 mg/kg/day |
| Methionine | 10 mg/kg/day |
The bottom line on very low protein diet for kidney health
High animal protein intake is associated with faster progression of CKD, likely due to excess production of nitrogen byproducts from protein breakdown. Following a whole food plant-dominant diet can be helpful to delay the progression of kidney disease. The use of ketoacid analogues as supplements may not be practical. In order to assure that the patient is getting adequate sources of EAA and other necessary nutrients, dietary restrictions should be implemented with the help of an experienced nutritionist, preferably one trained in a personalized approach to kidney health.
The post Very Low Protein Diet for Kidney Health appeared first on Integrative Kidney.]]>





