Colchicine for CKD!
In a multi-center, nested, case-control study in three Korean hospitals, patients with CKD stage 3 and 4 who are using drugs including colchicine, allopurinol, and febuxostat for high uric acid or chronic gout were studied over a period of 10 years. The progression of CKD was compared between 3085 compared to 11715 control patients.
Colchicine use was associated with a lower risk of adverse kidney outcomes in CKD patients with hyperuricemia, or chronic gout.
Unlike a study published two years ago in NEJM which excluded patients with advanced CKD, this study included patients with kidney function as low as 15 ml/min. Colchicine is known to anti-inflammatory. It also protects against kidney fibrosis.
There are concerns about myopathy and neuropathy with the intake of colchicine. It is, therefore, important to adjust the dose with advanced kidney disease and to be cautious when using it with patients who are on other myopathy-inducing drugs such as statin drugs.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Long sleep duration is associated with decline in kidney function
This study is retrospective, longitudinal cohort study included 82,001 participants who visited a primary care center in Japan. Patients were categorized into CKD risk groups and sleep duration categories according to their self-reported average nightly sleep duration. The relationship between average nightly sleep duration and the incidence of composite renal outcome was studied.
Researchers found that an average sleep durations ≥8 h/night were associated with an increased risk of kidney function decline over time.
There are many reasons that connect sleep problems with poor kidney function. We summarized these in this blog.
Risk of environmental heavy metal toxicity is higher in CKD
In a study of 5,638 NHANES participants, lead and cadmium levels were higher in patients with CKD than those without it. This was also associated with decreased urinary lead excretion. Each decrease in estimated GFR by 10 ml/min/1.73m2 was associated with 0.05 mcg/dL increase in lead levels and 0.02 mcg/dL of cadmium levels. This association was even stronger among black participants.
The study concluded that CKD increases the susceptibility to heavy metal environmental exposure by reducing its elimination.
Download Your Copy!
Join here to receive FREE monthly updates on the latest research in Integrative Nephrology, and tips on managing kidney disease straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post May Research And News appeared first on Integrative Kidney.]]>

3 years of lifestyle interventions improved exercise capacity and decreased the losses in neuromuscular and cardiorespiratory fitness in CKD patients
In this study, researchers randomized 161 patients with stage 3-4 CKD to either get usual care or usual care plus lifestyle “intervention” for 3 years.
The lifestyle intervention comprised of care from a multidisciplinary team, including a nephrologist, nurse practitioner, exercise physiologist, dietitian, diabetes educator, psychologist, and social worker.
The patients were coached for 8 weeks and then followed for 34 months with a home-based program.
The study did not look at the progression of CKD but it found that a 3-year lifestyle intervention doubled the percentage of CKD patients meeting physical activity guidelines, improved exercise capacity, and decreased the losses in neuromuscular and cardiorespiratory fitness.
It appears that the study mainly focused on exercise. So imagine the benefit of a comprehensive lifestyle modification plan that includes nutrition, exercise, stress management, sleep improvement, and attention to toxin exposure and gut-kidney connection. That’s what we focus on.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Curcumin powder did not improve markers of vascular dysfunction in PKD
This is essentially a negative study.
It demonstrated that Curcumin powder did not improve markers of vascular dysfunction in children and young adults with PKD. The study was conducted for only one year using a dose of 25 mg/kg per day of curcumin.
This is a classic supplement or nutrient study that is usually underpowered or conducted for short periods of time for a disease that takes years or even decades to evolve. Nevertheless, the study proved that short-term use of curcumin is not beneficial for vascular health for young patients with polycystic kidney disease.
Low serum zinc levels were associated with infections in CKD patients
This did not really need research but it is now studied and it is official: Low zinc levels in patients with CKD lead to infection (..well among other things).
This retrospective study analyzed data from 299 CKD patients who had serum zinc levels checked to evaluate anemia. They used the level of 50 mcg/dl as the cutoff between low or “high” zinc values.
Low serum zinc values remained an independent risk factor for infection-related hospitalization. This was especially true for patients taking proton pump inhibitors (PPIs) medications.
Read about the effect of Zinc on kidney health in this blog.
Download Your Copy!
Join here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to here your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post January Research and News appeared first on Integrative Kidney.]]>
Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>Low magnesium levels have been associated with a number of adverse events, such as high risk for heart disease. However, little is understood about magnesium and kidney health. Here, we will discuss the potential benefits of magnesium on the kidneys. This is one of two articles on magnesium and kidneys. For more on how to test and treat kidney patients with magnesium deficiency, see part two, “Magnesium Deficiency: Assessment and Management for Better Kidney Health.”

Dietary sources of magnesium
A daily intake of 3.6 mg/kg is necessary to maintain magnesium balance in humans under normal conditions. This is estimated to be between 320 to 420 mg/day (13–17 mmol/day) for adults. Sadly, there has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is due to depletion of magnesium in soil over time. This, along with the rise of ultra-processed food, sodas, and taking medications such as proton pump inhibitors and diuretics that deplete magnesium levels (polypharmacy), has led to rising prevalence of magnesium deficiency.
Traditionally, the highest food sources of magnesium are:
- Leafy greens (78 mg/serving on average)
- Nuts (80 mg/serving on average)
- Pumpkin seeds have the highest level of magnesium per serving (156 mg).
- Whole grains (46 mg/serving on average)
A complete list of foods high in magnesium can be found here.
Can Magnesium Help Kidney Function?
There are many potential benefits of magnesium for kidney health including improving blood pressure control, insulin sensitivity, bone health, vascular health, and preventing kidney stones. Let’s explore the data.
Magnesium and blood pressure control
Magnesium supplementation may help reduce blood pressure (BP) by increasing the production of nitric oxide. Nitric oxide acts as a signaling molecule that helps relax blood vessels, which lowers BP. In fact, a review of 34 studies showed that supplementing magnesium with an average dose of 368 mg per day for 3 months can decrease systolic BP by 2.00 mmHg and diastolic BP by 1.78 mmHg. This supplementation was accompanied by 0.05 mmol/L increase in serum magnesium levels.
Download Your Copy!
Magnesium and insulin sensitivity
Diabetes is one of the major risk factors for kidney disease worldwide. Higher dietary intake of magnesium has been correlated with lower diabetes incidence. A review of 18 studies in people with diabetes showed that magnesium supplements reduced fasting plasma glucose levels. In people who are at high risk for diabetes, magnesium supplementation significantly improved plasma glucose levels after a 2-hour oral glucose tolerance test. These effects are thought to be due to the effects of magnesium on insulin receptors and signaling that allows for improvement in glucose transport and utilization.
Magnesium and vascular health
Magnesium levels have been associated with a lower incidence of cardiovascular disease. In fact, supplementing with magnesium was associated with improvement in vascular flow and endothelial function. Endothelial function refers to the lining of the blood vessels, which is involved in regulating blood vessel health and blood clotting.
Studies in patients receiving dialysis have shown that having a lower serum magnesium level is a significant risk for cardiovascular mortality. Laboratory data show that magnesium inhibits high phosphate-induced calcification of vascular smooth muscle cells. Calcification of arteries is a strong predictor of heart disease and heart-disease-related death.
Magnesium and vitamin D
Magnesium is essential to vitamin D metabolism. Vitamin D that we eat or make in our skin from sun exposure circulates in the blood and is bound to vitamin D binding protein (VDBP). VDBP binding activity depends on adequate magnesium levels. In addition, magnesium is an essential cofactor for the enzymes that activate vitamin D. Studies have demonstrated that magnesium deficiency is associated with impaired vitamin D metabolism.
On the other hand, taking large doses of vitamin D can induce severe depletion of magnesium. This is thought to be due to the overutilization of magnesium. Therefore, adequate magnesium supplementation should be an important part of vitamin D therapy.
Adequate magnesium supplementation should be an important part of vitamin D therapy.
Share on X
Magnesium and bone health
Besides magnesium’s effects on vitamin D metabolism, it is an essential component of hydroxyapatite, an essential component of bone and teeth. In fact, 60% of total Mg is stored in the bone. Low magnesium intake was found to be associated with lower bone mineral density in postmenopausal women. Magnesium deficiency contributes to osteoporosis directly by acting on crystal formation and on bone cells and indirectly by impacting the secretion and the activity of parathyroid hormone (PTH) and by promoting oxidative stress and inflammation.
In addition, a review of 8 studies looked at magnesium and chronic kidney disease (CKD). The study investigated magnesium supplementation on parameters of CKD-related mineral bone disease (CKD-MBD). Mg supplementation improved PTH levels and carotid intima-media thickness (CIMT). Low serum Mg levels were also found to impact PTH and worsen osteoporosis in CKD patients, particularly with diabetes.
Magnesium and kidney stones
Mg acts as an inhibitor of calcium oxalate crystallization and stone formation in the urine. It also decreases the absorption of dietary oxalate in the gut. Mg supplementation in patients with kidney stones was found to decrease the incidence of stone formation even in patients without signs of Mg deficiency.
Magnesium as a phosphate binder
Hyperphosphatemia (high phosphate level) is common in advanced kidney disease. Many kidney patients with stage 4 and above use binders that bind phosphate (or “phosphorus,” as it is commonly known) in the food and prevent it from getting absorbed. High phosphate levels have been associated with poor bone and vascular health in kidney patients. In fact, higher dietary phosphate load can be seen in earlier stages of CKD, and it can do harm even before it is detected.
Magnesium carbonate has been successfully used as a phosphate binder. Magnesium based phosphate binders were also found to reduce vascular calcifications in rats with kidney disease. Iron-magnesium hydroxycarbonate was also studied and found to be well tolerated and can effectively lower phosphate levels in dialysis patients. It is essential to know that most of the magnesium used as a phosphorus binder will not be absorbed.
The bottom line on magnesium and kidneys
Magnesium is essential to many biological functions. It has many health benefits for kidney, bone, and vascular health. Optimizing magnesium status is, therefore, an important step in the integrative approach to kidney health. In part two of this blog, “Magnesium Deficiency: Assessment and Management for Better Kidney Health,” we will discuss practical steps for figuring out a person’s actual magnesium status, the best form of magnesium to take, and the dose I recommend for each condition.
The post Magnesium and kidneys appeared first on Integrative Kidney.]]>We combed through multiple medical journals looking for the latest research on the Integrative approach to kidney health. We know your time is valuable so we curated and summarized these studies for you. Welcome to the November edition of InKidney Research and News.

Is phosphorus toxic to the kidneys?
In this detailed study, researchers found that excessive intake of phosphate (phosphorus) increases a substance called FGF-23. One of FGF-23 main tasks is to maintain phosphorus balance in the blood. It does that by increasing the excretion of phosphorus in the urine. This study found that the presence of high phosphorus in the kidney tubules causes them to bind with calcium forming tiny crystals that are very toxic to the tubular cells and leads to fibrosis. Investigators found that a level of FGF-23 above 53 pg/mL is associated with progression of kidney disease in humans.
Coffee and caffeine consumption and the risk of kidney stones
Investigators used Mendelian randomization to study the link between coffee consumption and the risk of kidney stones. They looked into the UK and Finland genetic databases and tried to find the association between coffee drinking genotype and kidney stone phenotype.
The study found that genetically predicted coffee and caffeine consumption was associated with a lower risk of kidney stones. This supports other observational studies that indicated the same.
But of course, the study means that consumption of black coffee is associated with decreased risk for kidney stones. It does not addresses latté or triple caramel Frappuccino. So, as always drink coffee in moderation.
Download Your Copy!
One in every 5 CKD patients receives a nephrotoxic medication
This study published ahead of print looked into the use of nephrotoxic medications in the US and Sweden. They studied a total of 74,135 patients with stage 3-5 CKD. In one year, one in every 5 patients with CKD, received a nephrotoxic medication.
Subscribe here to receive a FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
The post November Research and News appeared first on Integrative Kidney.]]>
By Majd Isreb, MD, FACP, FASN, IFMCP
The nervous system and kidney health
The autonomic nervous system describes the system that controls involuntary actions, such as heartbeat and the widening or narrowing of blood vessels. It has two components: the sympathetic system (fight or flight system), and the parasympathetic system (or the rest and digest system).
It has been long established that the sympathetic system innervates all segments of the kidneys including the blood vessels and tubules. Stimulating the sympathetic system (fight or flight) can affect blood flow to the kidneys and lead to sodium retention at the level of the tubules, resulting in the progression of kidney disease. The kidneys are also innervated by small branches of the parasympathetic (rest and digest) vagus nerve.
What is the vagus nerve?
The vagus nerve is the longest nerve in the body. The word “vagus” means “wanderer” in Latin. This nerve is described as such because it connects the brain with many vital organs including the heart, lungs, gut, liver, spleen, and kidneys. It, therefore, affects breathing, digestion, and heart rate. Early studies also demonstrated that it also affects kidney blood flow.
Fascinating new evidence shows that the vagus nerve plays an important role in modulating inflammation in the body. When foreign particles are identified in various organs, “inflammatory signals” or neural messages are conveyed to the brain by the vagus nerve. The vagus nerve, then, transmits reflex signals back. These signals end up activating the splenic (spleen) nerve, which leads to a decreased inflammatory response and reduced inflammatory cytokines.
Simply put, the vagus nerve identifies triggers for inflammation and helps the brain regulate the inflammatory response. This is an evolving field called the cholinergic anti-inflammatory pathway (CAP).
Download Your Copy!
Stress worsens kidney disease
Our increasingly stressful lives can lead to increased cortisol levels, insulin resistance, and inflammation. Stress has been linked to imbalances between the sympathetic and parasympathetic systems. This is often described as autonomic dysfunction. These imbalances can also lead to the development and progression of kidney disease. Genetics, environment, and nutrition play important roles in augmenting these changes.
Autonomic dysfunction is a common finding in CKD. It is often measured by what is called heart rate variability (HRV).
Join us to end the kidney disease epidemic and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Heart rate variability as a measure of vagus nerve activity
HRV is simply a measure of the variation in time between each heartbeat. Because this variation is controlled by the autonomic system, HSV can be used to measure changes in this system. The vagus nerve plays an essential role here since it controls heart rate. Therefore, stimulation of the vagus nerve can affect HRV.
HRV can be measured by analyzing a long strip of electrocardiogram (EKG). However, several companies created “apps” and heart rate monitors that can measure it. Unfortunately, the accuracy of these apps and wearable devices is not yet clear. The easiest way to check HRV is to buy a chest strap heart monitor (Polar, Wahoo) and download a free app such as Elite HRV to analyze the data. Chest straps tend to be more accurate than wrist or finger wearables.
Finally, decreased HRV has been linked to bad outcomes in hypertension, systemic inflammation, depression, and CKD. Low HRV can increase the risk of sudden cardiac death. Therefore, there is increasing interest in manipulating heart rate variability by stimulating the vagus nerve.
Activate the vagus nerve to calm inflammation in CKD
Vagus Nerve Stimulation
CKD is a state of chronic inflammation that is associated with heart disease and poor outcomes. As mentioned above, the vagus nerve plays an important role in detecting and calming inflammation in the body. It identifies sources of inflammation and helps the brain modulate the inflammatory response appropriately. The vagus nerve may play an anti-inflammatory role in conditions such as endotoxin-induced sepsis, arthritis, and acute kidney injury.
Electrical stimulation of the vagus nerve is the most common method to modulate the inflammatory response and other functions of the vagus nerve. Pharmacological methods are also being studied for that purpose. However, there are many simple and natural ways to stimulate the vagus nerve.
Natural ways to stimulate the vagus nerve
Stimulating the vagus nerve can improve the response to emotional and physiological stress, thereby improving kidney health. It can also help decrease inflammation. In fact, the FDA approved a surgically implanted device that stimulates the vagus nerve to treat resistant depression. However, there are many ways to stimulate it naturally. I will mention them briefly here.
Activate the Vagus Nerve Naturally to Improve Kidney Disease:
- Cold exposure such as cold showers.
- Deep and slow breathing can activate the parasympathetic system.
- Singing, humming, chanting, and gargling can also activate the vagus nerve because it is connected to the vocal cords.
- Probiotics, interestingly, can be beneficial in activating the gut-brain axis and stimulating the vagus nerve.
- Meditation has been found to increase vagal tone and positive emotions.
- Omega-3 fatty acids have yet another great benefit- increasing vagal activity.
- Exercise is of course linked to better mental health and stimulation of the vagus nerve.
- Massage therapy has also been found to be associated with improved vagal tone.
- Socializing and laughing can also be beneficial.
The Bottom Line
Emotional stress can be associated with faster progression of CKD. The kidneys have both sympathetic and parasympathetic innervation. Acute stress can worsen kidney function, while relaxation practices can promote kidney health. Stimulating the vagus nerve can turn on the parasympathetic nervous system, improving renal blood flow and modulating inflammation in CKD. Stress reduction and the natural stimulation of the vagus nerve can help delay the progression of kidney disease.
The post Natural Treatments to Improve Chronic Kidney Disease: Vagus Nerve Stimulation appeared first on Integrative Kidney.]]>
Knowledge Does Not Correspond to Adherence to Renal Diet Restrictions in Patients With Chronic Kidney Disease
Education or knowledge alone is not sufficient to make meaningful lifestyle changes. It is not enough for a patient to meet with a doctor, dietitian, or nutritionist once and be expected to be educated about diet and other modifications.
The researchers in this study proved and concluded that “instruction efforts should go beyond providing nutrient-based diet information, and instead emphasize healthy food patterns and incorporate counseling to promote behavior change.”
In another word, patients need to be educated and coached to achieve better lifestyles and outcomes.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Apixaban, compared with warfarin, was associated with a lower risk of CKD stage progression
In this study, researchers studied 12,816 patients who received either apixaban or warfarin for non-valvular atrial fibrillation. Apixaban was associated with slower progression of CKD than warfarin. We think that this underlines the role of vitamin K in vascular and kidney health.
Periodontal disease was associated with incident kidney disease
Researchers studied data from the Hispanic community health study to see if there is any association between periodontal disease and the development of kidney disease.
They found that the presence of ≥30% of sites with probing depth ≥4 mm and absence of functional dentition (<21 permanent teeth present) were each associated with increased risk for incident low eGFR.
Does treating periodontal disease prevent the development and progression of CKD? Research does not have the answer yet but we think so.
Download Your Copy!
Join here to receive FREE monthly updates on the latest research in Integrative Nephrology, and tips on managing kidney disease straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com

Metformin and CKD survey results
You can see the results of the survey in this earlier blog. It demonstrated that caution about the use of metformin in CKD is common among primary care physicians and other specialties. Nephrologists seem to be more comfortable with using it until the patient reaches stage 4 CKD. Most nephrologists, however, were not aware that metformin is associated with nutritional deficiencies. You can read the results of the survey in detail here.
What is metformin?
Metformin was first produced in 1922. However, since 1958 it has been used in the control of blood sugar in type 2 diabetes. It is a biguanide drug, a class of medications that inhibit the liver’s production of glucose, enhancing the body’s insulin sensitivity and lowering the amount of blood sugar that the intestines absorb. It is chemically developed from galegina, a natural guanidine compound found in Galega officinalis (French lilac, also known as goat’s rue).
In essence, metformin is an oral diabetes medication that helps control blood sugar levels. It is often used together with diet and exercise to improve blood sugar control in patients with type 2 diabetes mellitus. There are other off-label uses for metformin and it is currently being studied for the treatment of polycystic kidney disease.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Does metformin cause acute kidney injury?
For many years clinicians have been avoiding the use of metformin in kidney patients and specifically in those with diabetes. This is fueled by the FDA warning about the use of metformin in these patients due to concerns about lactic acidosis (discussed later). The original FDA warning was much more strict, but the more recent one issued in 2016 weighed in with more detail about metformin and kidney function. The new warning allows metformin to be used in patients who have worse kidney function. The newest warning indicates metformin can be used in patients who have an estimated glomerular filtration rate (eGFR) greater than 45 mL/min. Furthermore, metformin can continue to be used in existing patients whose eGFR remains over 30 mL/min.
As you saw from the survey, many primary care physicians and other specialists think that metformin can cause direct acute kidney injury. However, the literature shows that metformin can be protective in kidney disease. There is absolutely no evidence that metformin can lead to kidney injury. In fact, there is evidence to the contrary.
Download Your Copy!
Metformin clearance by the kidneys
Metformin does not undergo metabolism in the body, and it is eliminated unchanged in the urine. It is mainly cleared by tubular secretion in the kidneys. Metformin clearance is proportional to creatinine clearance. Therefore, although metformin can be used in patients with advanced kidney disease the dose of metformin should be decreased. The following table shows the recommended doses of metformin according to kidney function.
| Kidney function | Total daily metformin dose |
| Normal | 3000 mg |
| 45-59 ml/min | 2000 mg |
| 30-44 ml/min | 1000 mg |
| Less than 30 ml/min | Not recommended |
Metformin-induced diarrhea
The most common side effects of metformin are gastrointestinal. This is because one of the ways that metformin works is directly on the gut, altering glucose (sugar) absorption in the gut. Consequently, symptoms of nausea and diarrhea can occur especially in the first few weeks of taking it. That is why the patient should be started on a low dose to get used to it. Despite the statistic that 25% of patients taking metformin report diarrhea, only about 5% of those taking it had to stop it due to that side effect.
Metformin and lactic acidosis
The real reason why metformin was technically “banned” from use in CKD patients in previous years was due to the development of lactic acidosis. Lactic acidosis is a disease where lactic acid, a metabolic byproduct, accumulates in the blood. Lactic acid is mainly produced in small amounts by the muscles and red blood cells. It is formed when the body breaks down sugars to make energy under conditions of low oxygen (anaerobic glycolysis). This can occur during intense exercise or severe infection (called sepsis).
Metformin can induce lactic acidosis by shifting energy production in the cells toward anaerobic glycolysis. It also decreases the ability of the liver to consume lactic acid. Yet, in practice this is rare. In fact, it has been shown that lactic acidosis can occur in patients taking other oral blood-sugar-lowering medications at the same rate as metformin. Most case reports of metformin-induced lactic acidosis were in patients who have other conditions that lead to acute kidney injury (AKI) and lactic acidosis such as septic shock.
So, it is not that:

Instead, what happens is:
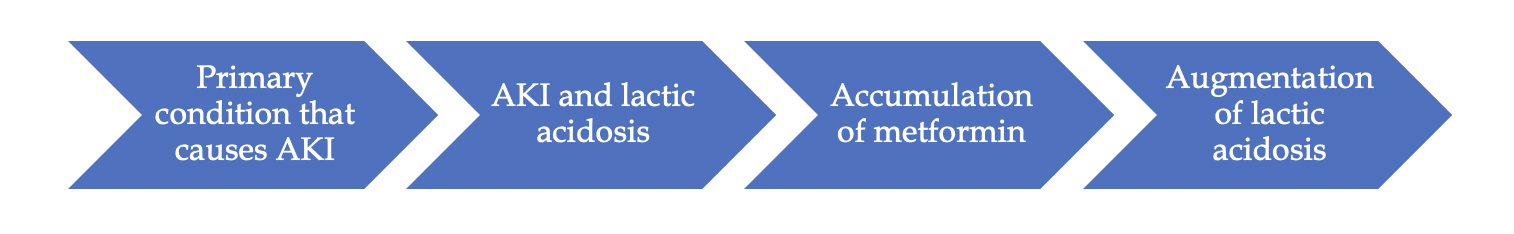
Finally, in a large retrospective study of patients with diabetes mellitus, metformin use was not significantly associated with lactic acidosis at kidney function levels higher than 30 ml/min/1.73 m2 (stage 3 or better).
Metformin and vitamin B12 deficiency
The association between long-term use of metformin and low vitamin B12 levels in type 2 diabetes mellitus patients is well-established. This is thought to be due to changes in the production of intrinsic factor in the stomach, which decreases vitamin B12 absorption in the intestine. Vitamin B12 deficiency is more pronounced if the patient is also taking a proton pump inhibitor such as omeprazole or histamine H2 receptor antagonist such as famotidine, which are medications used to treat acid reflux. Since diabetes can cause peripheral neuropathy, as well as vitamin B12 deficiency, can cause peripheral neuropathy, it can be confusing for diabetic patients on metformin and their practitioners.
Therefore, patients using metformin for a prolonged period should be screened initially and periodically for vitamin B12 deficiency. In addition, there is also growing evidence that other B vitamins, vitamin D, and magnesium can be impacted by metformin.
Bonus: Metformin’s impact on the gut microbiome
Because of metformin’s actions on the gut, it can also alter the intestinal microbiota for the better. Studies showed that metformin can enhance the growth of Akkermansia muciniphila, Butyrivibrio, Bifidobacterium, and Lactobacillus. In addition, it decreases the levels of some other bacteria like Intestinibacter. Metformin also promotes the production of the short-chain fatty acids that protect the intestinal barrier. In fact, some authors attribute some of metformin’s actions to its favorable impact on the gut microbiota and integrity.
The Bottom Line for Metformin and Kidney Disease
The answer to the question “Is metformin bad for the kidneys?” is generally no. Metformin can be used in patients with kidney disease. It actually leads to better outcomes in these patients. However, the risk of metformin-induced lactic acidosis increases in CKD stage 4 and above. It is, therefore, not recommended for use in these patients and patients with acute kidney injury. I also recommend avoiding it in patients with renovascular disease because their kidney function tends to fluctuate. Furthermore, metformin can be associated with vitamin B12 deficiency and patients should be screened initially and periodically for deficiency of this important vitamin.
The post To Metformin or Not To Metformin in CKD appeared first on Integrative Kidney.]]>While this is not official medical research, I aimed to identify current thoughts among nephrologists and primary care providers about its use in CKD patients. The survey results suggest that many practitioners avoid metformin in CKD based on outdated FDA guidelines. In fact, metformin has beneficial effects for kidney health and can safely be used in certain stages of CKD. In this blog, I will share the survey results. For the current evidence and recommendations for metformin use in kidney disease, read the second part of this blog, “To Metformin or Not to Metformin in CKD.”

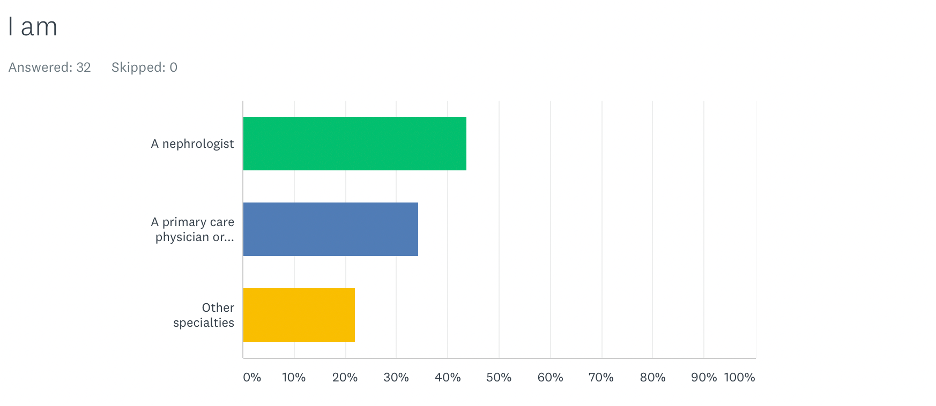
Participants in the Metformin and Kidney Disease Survey
We had 32 respondents: 44% of the respondents were nephrologists, 34% were primary care physicians, and 22% were from other specialties.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
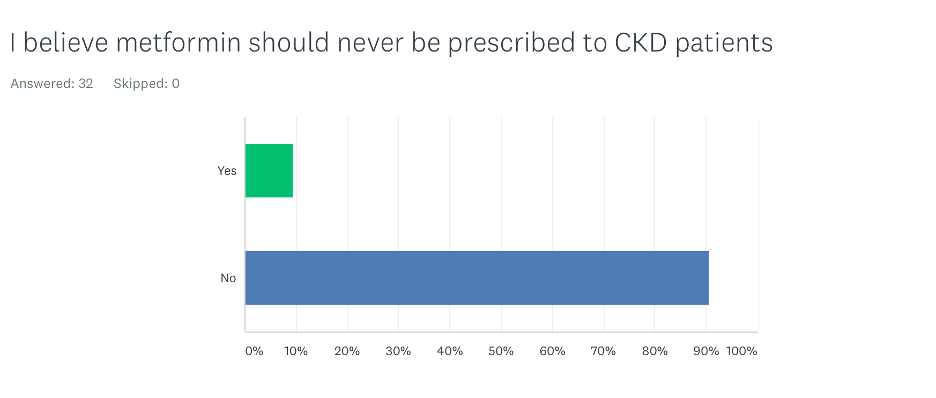
Should metformin be used in kidney disease patients?
Only 9% of participants thought that metformin should never be used in all CKD patients. All nephrologists believed that metformin can be used in patients with CKD. Among primary care providers, 27% believed that metformin should never be used in all CKD patients.
Does metformin cause acute kidney injury?
In the survey, 28% of participants believed that metformin causes acute kidney injury. The majority of those were primary care physicians. Only 7% of nephrologists believed that metformin causes acute kidney injury.
Block "fundamentals" not found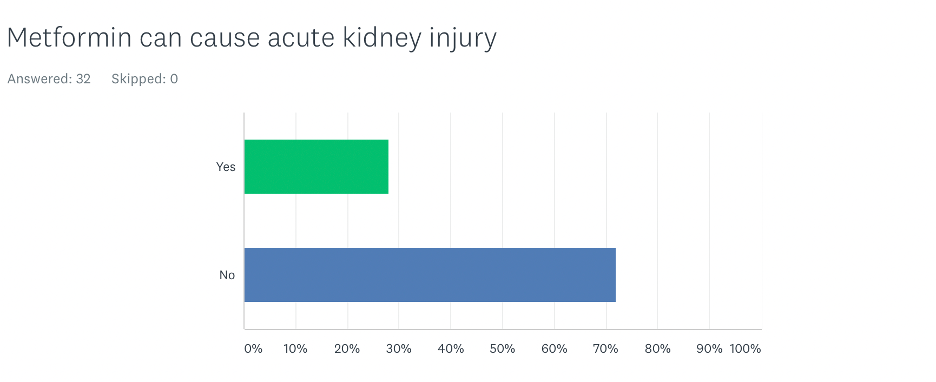
At what stage of CKD do you stop metformin?

The majority of participants (56%) believed that metformin should be discontinued when a patient reaches stage 4 CKD or above. Interestingly, 25% of the participants recommended discontinuing it if the patient reaches stage 3b CKD. Most nephrologists discontinued it when patients reached stage 4 CKD or above. Most physicians who discontinued it at earlier stages were primary care physicians.
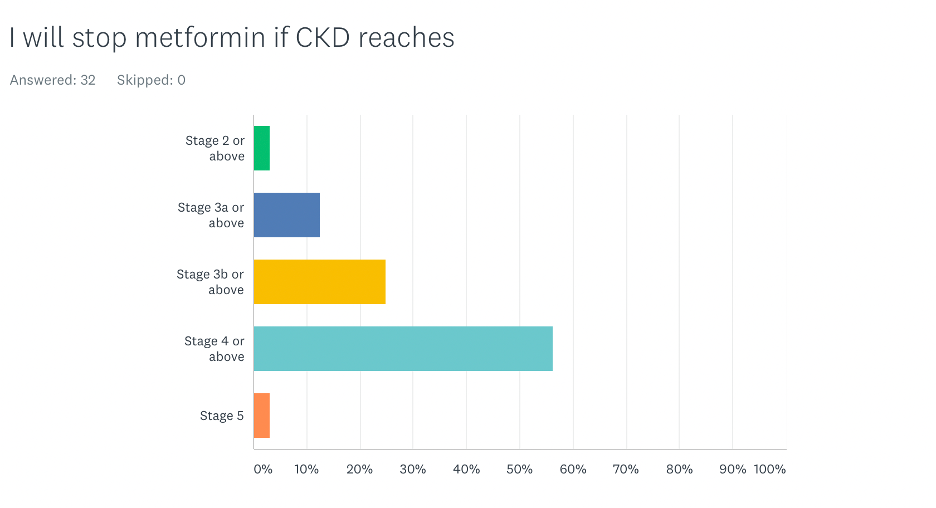
What is the problem with the use of metformin in advanced kidney disease?
This question may be suggestive, but about half of the participants believed that the risk of lactic acidosis is the only problem with the use of metformin in patients with advanced CKD. Interestingly, the majority of participants who believed there are other problems with the use of metformin in advanced kidney disease were non-nephrologists. One would think that nephrologists would be more aware of other risks beyond lactic acidosis.
When asked about specific reasons to avoid using metformin in advanced CKD, two rationales mainly emerged. Because metformin is removed from the body by way of the kidneys, one reason for its avoidance is the potential accumulation of excessively high levels of metformin in advanced kidney disease. The other reason was that metformin can be associated with diarrhea that can lead to volume depletion (loss of body fluids) and acute kidney injury.
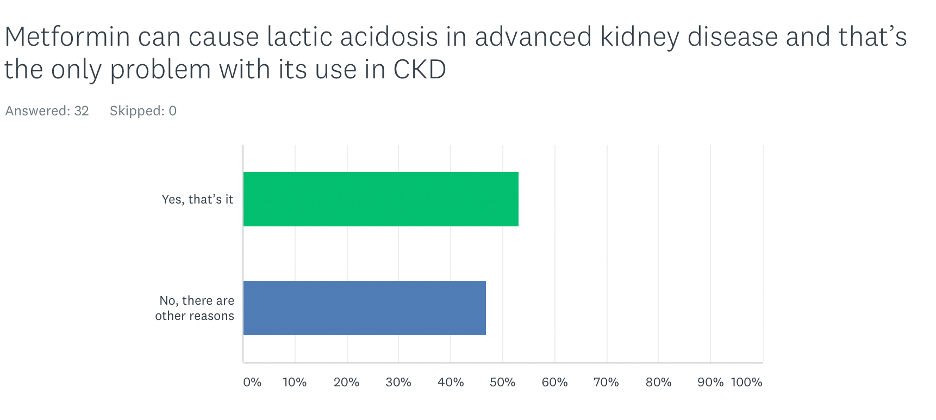
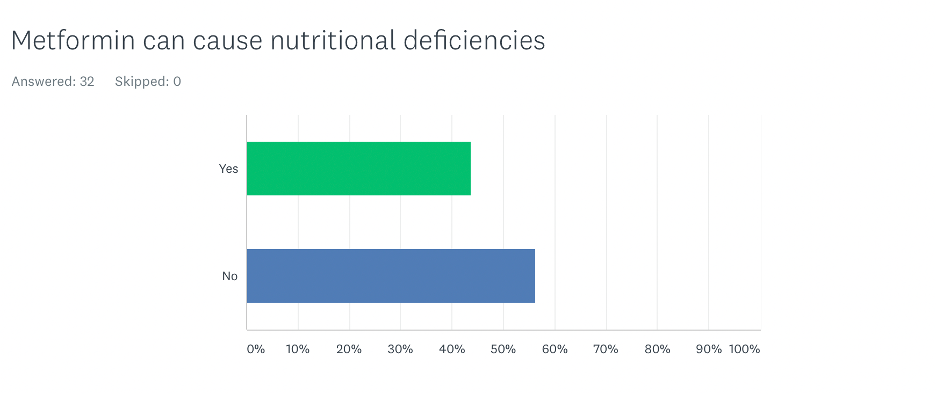
Can metformin cause vitamin B12 and other nutritional deficiencies?
Strikingly, 56% of participants were not aware of metformin and vitamin B12 deficiency or that it may cause depletion of other nutrients. Among nephrologists, this percentage was an amazing 79%.
The Bottom Line
Caution about the use of metformin in CKD is common among primary care physicians and other specialties. Nephrologists seem to be more comfortable with using it until the patient reaches stage 4 CKD. Most nephrologists, however, were not aware that metformin is associated with nutritional deficiencies. In the next blog, I will dig deeper into the research to find evidence-based answers to the questions presented in this survey.
Download Your Copy!

The Role of Dietary Fiber Supplementation in Regulating Uremic Toxins in Patients With Chronic Kidney Disease: A Meta-Analysis of Randomized Controlled Trials
Gut-derived uremic toxins, including indoxyl sulfate (IS) and p-cresyl sulfate (PCS), are waste products that accumulate in the body due to decreased clearance by the failing kidneys. These accumulated uremic toxins are a risk factor associated with cardiovascular disease, progression of kidney dysfunction, mineral bone disorders, and increased mortality in patients with CKD. This meta-analysis showed that dietary fiber supplementation could decrease IS and PCS levels in patients with CKD.
https://www.jrnjournal.org/article/S1051-2276(20)30291-0/fulltext
Causal effects of physical activity or sedentary behaviors on kidney function: an integrated population-scale observational analysis and Mendelian randomization study
The health benefits of exercise and regular physical activity are vast. Using Mendelian randomization (MR) to assess causality, this study found that “the genetic predisposition to a higher degree of physical activity was associated with a lower risk of CKD and a higher eGFR, while the genetically predicted television watching duration was associated with a higher risk of CKD and a lower eGFR.”
https://academic.oup.com/ndt/advance-article/doi/10.1093/ndt/gfab153/6214515?login=true
Prevalence of Inflammatory Bowel Disease and Celiac Disease in Patients with IgA Nephropathy over Time
Poor gut health and a lack of intestinal wall integrity is linked to many diseases, including IgA nephropathy (IgAN). The results of this Finnish study showed an increase in the prevalence of inflammatory bowel disease (IBD) in patients with newly diagnosed IgAN over a 36-year period.
The post June Research and News appeared first on Integrative Kidney.]]>





