
3 years of lifestyle interventions improved exercise capacity and decreased the losses in neuromuscular and cardiorespiratory fitness in CKD patients
In this study, researchers randomized 161 patients with stage 3-4 CKD to either get usual care or usual care plus lifestyle “intervention” for 3 years.
The lifestyle intervention comprised of care from a multidisciplinary team, including a nephrologist, nurse practitioner, exercise physiologist, dietitian, diabetes educator, psychologist, and social worker.
The patients were coached for 8 weeks and then followed for 34 months with a home-based program.
The study did not look at the progression of CKD but it found that a 3-year lifestyle intervention doubled the percentage of CKD patients meeting physical activity guidelines, improved exercise capacity, and decreased the losses in neuromuscular and cardiorespiratory fitness.
It appears that the study mainly focused on exercise. So imagine the benefit of a comprehensive lifestyle modification plan that includes nutrition, exercise, stress management, sleep improvement, and attention to toxin exposure and gut-kidney connection. That’s what we focus on.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Curcumin powder did not improve markers of vascular dysfunction in PKD
This is essentially a negative study.
It demonstrated that Curcumin powder did not improve markers of vascular dysfunction in children and young adults with PKD. The study was conducted for only one year using a dose of 25 mg/kg per day of curcumin.
This is a classic supplement or nutrient study that is usually underpowered or conducted for short periods of time for a disease that takes years or even decades to evolve. Nevertheless, the study proved that short-term use of curcumin is not beneficial for vascular health for young patients with polycystic kidney disease.
Low serum zinc levels were associated with infections in CKD patients
This did not really need research but it is now studied and it is official: Low zinc levels in patients with CKD lead to infection (..well among other things).
This retrospective study analyzed data from 299 CKD patients who had serum zinc levels checked to evaluate anemia. They used the level of 50 mcg/dl as the cutoff between low or “high” zinc values.
Low serum zinc values remained an independent risk factor for infection-related hospitalization. This was especially true for patients taking proton pump inhibitors (PPIs) medications.
Read about the effect of Zinc on kidney health in this blog.
Download Your Copy!
Join here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to here your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post January Research and News appeared first on Integrative Kidney.]]>
Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>Low magnesium levels have been associated with a number of adverse events, such as high risk for heart disease. However, little is understood about magnesium and kidney health. Here, we will discuss the potential benefits of magnesium on the kidneys. This is one of two articles on magnesium and kidneys. For more on how to test and treat kidney patients with magnesium deficiency, see part two, “Magnesium Deficiency: Assessment and Management for Better Kidney Health.”

Dietary sources of magnesium
A daily intake of 3.6 mg/kg is necessary to maintain magnesium balance in humans under normal conditions. This is estimated to be between 320 to 420 mg/day (13–17 mmol/day) for adults. Sadly, there has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is due to depletion of magnesium in soil over time. This, along with the rise of ultra-processed food, sodas, and taking medications such as proton pump inhibitors and diuretics that deplete magnesium levels (polypharmacy), has led to rising prevalence of magnesium deficiency.
Traditionally, the highest food sources of magnesium are:
- Leafy greens (78 mg/serving on average)
- Nuts (80 mg/serving on average)
- Pumpkin seeds have the highest level of magnesium per serving (156 mg).
- Whole grains (46 mg/serving on average)
A complete list of foods high in magnesium can be found here.
Can Magnesium Help Kidney Function?
There are many potential benefits of magnesium for kidney health including improving blood pressure control, insulin sensitivity, bone health, vascular health, and preventing kidney stones. Let’s explore the data.
Magnesium and blood pressure control
Magnesium supplementation may help reduce blood pressure (BP) by increasing the production of nitric oxide. Nitric oxide acts as a signaling molecule that helps relax blood vessels, which lowers BP. In fact, a review of 34 studies showed that supplementing magnesium with an average dose of 368 mg per day for 3 months can decrease systolic BP by 2.00 mmHg and diastolic BP by 1.78 mmHg. This supplementation was accompanied by 0.05 mmol/L increase in serum magnesium levels.
Download Your Copy!
Magnesium and insulin sensitivity
Diabetes is one of the major risk factors for kidney disease worldwide. Higher dietary intake of magnesium has been correlated with lower diabetes incidence. A review of 18 studies in people with diabetes showed that magnesium supplements reduced fasting plasma glucose levels. In people who are at high risk for diabetes, magnesium supplementation significantly improved plasma glucose levels after a 2-hour oral glucose tolerance test. These effects are thought to be due to the effects of magnesium on insulin receptors and signaling that allows for improvement in glucose transport and utilization.
Magnesium and vascular health
Magnesium levels have been associated with a lower incidence of cardiovascular disease. In fact, supplementing with magnesium was associated with improvement in vascular flow and endothelial function. Endothelial function refers to the lining of the blood vessels, which is involved in regulating blood vessel health and blood clotting.
Studies in patients receiving dialysis have shown that having a lower serum magnesium level is a significant risk for cardiovascular mortality. Laboratory data show that magnesium inhibits high phosphate-induced calcification of vascular smooth muscle cells. Calcification of arteries is a strong predictor of heart disease and heart-disease-related death.
Magnesium and vitamin D
Magnesium is essential to vitamin D metabolism. Vitamin D that we eat or make in our skin from sun exposure circulates in the blood and is bound to vitamin D binding protein (VDBP). VDBP binding activity depends on adequate magnesium levels. In addition, magnesium is an essential cofactor for the enzymes that activate vitamin D. Studies have demonstrated that magnesium deficiency is associated with impaired vitamin D metabolism.
On the other hand, taking large doses of vitamin D can induce severe depletion of magnesium. This is thought to be due to the overutilization of magnesium. Therefore, adequate magnesium supplementation should be an important part of vitamin D therapy.
Adequate magnesium supplementation should be an important part of vitamin D therapy.
Share on X
Magnesium and bone health
Besides magnesium’s effects on vitamin D metabolism, it is an essential component of hydroxyapatite, an essential component of bone and teeth. In fact, 60% of total Mg is stored in the bone. Low magnesium intake was found to be associated with lower bone mineral density in postmenopausal women. Magnesium deficiency contributes to osteoporosis directly by acting on crystal formation and on bone cells and indirectly by impacting the secretion and the activity of parathyroid hormone (PTH) and by promoting oxidative stress and inflammation.
In addition, a review of 8 studies looked at magnesium and chronic kidney disease (CKD). The study investigated magnesium supplementation on parameters of CKD-related mineral bone disease (CKD-MBD). Mg supplementation improved PTH levels and carotid intima-media thickness (CIMT). Low serum Mg levels were also found to impact PTH and worsen osteoporosis in CKD patients, particularly with diabetes.
Magnesium and kidney stones
Mg acts as an inhibitor of calcium oxalate crystallization and stone formation in the urine. It also decreases the absorption of dietary oxalate in the gut. Mg supplementation in patients with kidney stones was found to decrease the incidence of stone formation even in patients without signs of Mg deficiency.
Magnesium as a phosphate binder
Hyperphosphatemia (high phosphate level) is common in advanced kidney disease. Many kidney patients with stage 4 and above use binders that bind phosphate (or “phosphorus,” as it is commonly known) in the food and prevent it from getting absorbed. High phosphate levels have been associated with poor bone and vascular health in kidney patients. In fact, higher dietary phosphate load can be seen in earlier stages of CKD, and it can do harm even before it is detected.
Magnesium carbonate has been successfully used as a phosphate binder. Magnesium based phosphate binders were also found to reduce vascular calcifications in rats with kidney disease. Iron-magnesium hydroxycarbonate was also studied and found to be well tolerated and can effectively lower phosphate levels in dialysis patients. It is essential to know that most of the magnesium used as a phosphorus binder will not be absorbed.
The bottom line on magnesium and kidneys
Magnesium is essential to many biological functions. It has many health benefits for kidney, bone, and vascular health. Optimizing magnesium status is, therefore, an important step in the integrative approach to kidney health. In part two of this blog, “Magnesium Deficiency: Assessment and Management for Better Kidney Health,” we will discuss practical steps for figuring out a person’s actual magnesium status, the best form of magnesium to take, and the dose I recommend for each condition.
The post Magnesium and kidneys appeared first on Integrative Kidney.]]>
By Megan Morrison, PharmD
Yoga and meditation are both considered mind-body interventions which the National Center for Complementary and Integrative Health defines as “a range of procedures or techniques focusing primarily on the connections between the mind, body and behavior and their resultant effects on one’s health”.
Yoga, which is said to have originated in India more than 5,000 years ago, literally means “union” and is a practice that aims to balance and harmonize the mind, body, and emotions. Various styles of yoga include regulated breathing techniques (pranayama), physical postures (asanas), hand gestures (mudras), yogic relaxation (yoga nidra), and more. Meditation encompasses several techniques that are used to influence the mind through mechanisms in the parasympathetic nervous system to bring about calm and relaxation.
Both yoga and meditation have been documented to have measurable positive impact on various chronic conditions, including type 2 diabetes and hypertension, two disease states that are major risk factors for the development of kidney disease. We’ll look at the research supporting the benefits of both for kidney health.
Block "fundamentals" not found
Benefits of Yoga for Kidney Health
One study looked at the effects of a 6-month yoga program on renal function and quality of life in patients with chronic kidney disease. After 6 months, the yoga group saw a significant reduction in systolic and diastolic blood pressure, blood urea and serum creatinine levels, and significant improvement in quality of life. Furthermore, subjects in the yoga group had reduced need for dialysis than the control group although not statistically significant.
Another study looked at the effects of a 12-week yoga-based exercise program in hemodialysis patients. Significant improvements were seen for many of the variables measured including pain, fatigue, sleep disturbance, along with significant reductions in creatinine, blood urea, alkaline phosphatase, cholesterol, erythrocyte and hematocrit count.
Although the exact mechanism for many of the benefits of yoga have not been completely understood, they include:
- Improved blood pressure control
- Improved glycemic control
- More regulated eating patterns
- Improvement in markers for kidney function
- Significant reduction of the waste products associated with kidney damage, urea and serum creatinine
- Improved physical and psychological quality of life for kidney patients on dialysis
- Improved blood supply to muscles and increased muscle activity, strength, endurance, flexibility, and balance
- Favorable effects on body weight, adiposity, dyslipidemia, and insulin resistance
- Significant reduction in oxidative stress and increased antioxidant activity
- Improved parasympathetic tone, helping offset fight-or-flight response often associated with chronic disease
- Reduced need for dialysis
Benefits of Meditation for Kidney Health
In a study of 15 African American males with CKD and high blood pressure, there was a significantly greater reduction in systolic and diastolic blood pressures, mean arterial pressure, heart rate, and muscle sympathetic nerve activity during mindfulness meditation compared with the control.
Those with end-stage renal disease as well as mild CKD have been shown in multiple studies to have chronic activation and elevation of the sympathetic nervous system (SNS). Therefore, using a non-invasive, safe therapeutic intervention such as meditation to lower SNS activity in those with renal disease may mitigate long-term consequences.
One type of mindfulness meditation technique studied in kidney patients is called Benson’s relaxation technique which consists of “eye closing, muscle relaxation, breathing awareness, breathing out stating a word, and returning to the “word” when participants’ thoughts are distracted”. This technique has been shown to reduce levels of anxiety, depression, and improve sleep quality as well as improve quality of life in patients undergoing hemodialysis.
Benefits of meditation for those with kidney disease include:
- Improved quality of life for hemodialysis patients
- Reduced anxiety and depression
- Reduced sympathetic overactivity
- Better sleep
- Reduced blood pressure
- Less prone to various forms of stress
The Bottom Line
Research has demonstrated that yoga and meditation greatly improve measurable health outcomes and quality of life in those with kidney disease or those looking to protect their kidney health. Both modalities are generally safe for most people, but it’s best to consult your physician before initiating any exercise program including a yoga routine. When working with a clinician trained in Integrative or Functional Medicine, they will often suggest incorporating some type of mind-body intervention as part of your holistic therapeutic plan.
The post Yoga and Meditation for Kidney Health appeared first on Integrative Kidney.]]>In this series we’re focusing on the integrative approach to preventing kidney stone formation.
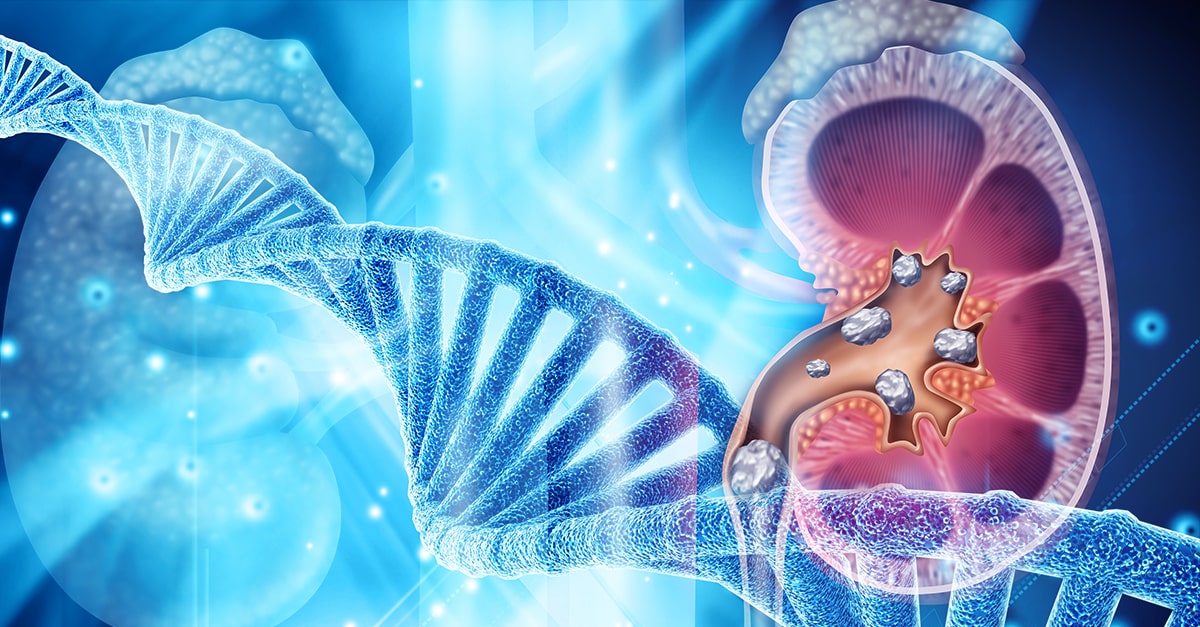
Conventional approaches to kidney stones tend to focus on medications, surgical removal, and using ultrasonic waves to break up stone. It rarely approaches the root cause including risk factors to prevent stone formation.
We covered the impact of diet, the microbiome and gut health, and electrolyte imbalances on kidney stones in previous blogs. Here we will discuss the role genetics play in kidney stone formation.
The Genetics of Kidney Stones
There seems to be a familial link when it comes to the development of kidney stones. In fact, two thirds of patients with calcium-containing kidney stones have a relative with kidney stones. Recently, genome-wide association studies uncovered several genetic sequence variants (SNPs) that lead to increased risk of kidney stone development. Although we are still scratching the surface in understanding the contribution of genetic factors to stone formation, we do know that we can modulate these risks through environmental and dietary modifications.
Genetic Variations in Calcium Handling
In a previous blog, we discussed the role of the kidney filtration units (specifically the nephrons). The kidneys are responsible for filtering large volumes of blood daily. This function is crucial, and, the unique design of the nephrons make it able to adjust this filtrate and prevent dehydration. This intricate design also makes the kidneys crucial in the balance of water and many electrolytes.
Calcium is one of the electrolytes filtered and reabsorbed in the kidneys. Calcium sensing receptors (CaSR) are present in kidney cells and are essential for the reabsorption of calcium. These receptors increase and decrease the amount of calcium reabsorbed based on the calcium level in the blood. In other words, activating these receptors increases the amount of calcium lost in the urine.
Single nucleotide polymorphisms (SNPs) in CaSR have been found to alter the function of these receptors leading to increased urinary excretion of calcium. Of particular interest, SNPs in rs7652589 and rs1501899 were associated with kidney stones in patients with normal citrate excretion. SNPS in rs1801725, rs1042636 of the CaSR gene were also associated with kidney stones in various populations.
Another gene that has been associated with increased calcium in the urine is the gene coding for the protein Claudin-14, responsible for forming tight junctions. This protein helps connect adjacent cells to form a barrier which acts like a gate, separating blood from urine. Tight junctions are also responsible for ensuring that minerals don’t pass between the cells. SNPs in the genes that code Claudin-14 (CLDN14) alters the integrity of these “gates” and allows for calcium to “sneak” between cells into the urine, increasing the risk for kidney stone formation. Specifically, SNPs at the locations rs219778, and rs219780 of the CLDN14 gene were significantly associated with kidney stones.
Genetic Variations in Vitamin D Receptors (VDR)
Vitamin D plays a crucial role in calcium balance. Studies have shown that vitamin D increases the absorption of calcium from the gut and also increases calcium excretion in the urine. Vitamin D receptors are essential for vitamin D to exert its action on calcium balance.
Some mutations or SNPs in the VDR are associated with increased absorption and excretion of calcium, significantly increasing the risk of kidney stones. It is worth mentioning here that there is some controversy about the link between vitamin D supplementation and kidney stone formation. Vitamin D deficiency appears to be common among kidney stone formers. This is likely because low vitamin D causes calcium loss from the bone in order to maintain normal calcium range in the blood for cardiovascular protection. Even though vitamin D3 supplementation may increase calcium excretion in the urine, it has not been conclusively found to increase the risk of kidney stone formation.
Therefore, genetic assessment may be a key to identify patients who are at risk of kidney stone formation from taking vitamin D supplements.
Genetic Variations in the Handling of Other Minerals
The kidneys are crucial for balancing many minerals in our bodies such as magnesium, phosphate, oxalate and others. Genetic mutation or SNPs affecting the genes that code for the channels or receptors that regulate these minerals can also impact the risk of kidney stones. Some SNPs on the other hand can be protective against kidney stones such as SNPs in the UMOD gene. Discussion of this long list of SNPs requires details beyond the scope of this blog, but we summarized most of the genes that have been associated with kidney stones in the table below.
| Gene symbol | Gene name | Phenotype |
| ADCT10 | Adenylate cyclase 10 | Increased calcium excretion |
| AGXT | Alanine-glyoxylate aminotransferase | Increased oxalate excretion |
| CA2 | Carbonic anhydrase II | Osteoporosis + decreased acid excretion |
| CASR | Calcium-sensing receptor | Increased calcium excretion |
| CLCN5 | Chloride channel, voltage-sensitive 5 | Dent disease |
| CLCNKB | Chloride channel, voltage-sensitive Kb | Bartter Syndrome, type 3 |
| CLDN14 | Claudin 14 | Increased calcium excretion |
| CLDN16 | Claudin 16 | Increased calcium and magnesium excretion |
| CLDN19 | Claudin 19 | Increased calcium and magnesium excretion |
| CYP24A1 | Cytochrome P450 | Decreased breakdown of vitamin D3 |
| GRHPR | Glyoxylate reductase | Increased oxalate excretion |
| HOGA1 | 4-Hydroxy-2-oxoglutarate aldolase 1 | Increased oxalate excretion |
| HPRT1 | Hypoxanthine phosphoribosyltransferase 1 | Increased uric acid excretion |
| SLC12A1 | Solute carrier family 12, member 1 | Bartter syndrome, type 1 |
| SLC26A1 | Solute carrier family 26, member 1 | Calcium oxalate kidney stones |
| SLC22A12 | Solute carrier family 22, member 12 | Decrease uric acid excretion |
| SLC2A9 | Solute carrier family 2, member 9 | Decreased uric acid excretion |
| SLC34A1 | Solute carrier family 34, member 1 | Calcium phosphate kidney stones |
| SLC34A3 | Solute carrier family 34, member 3 | Calcium phosphate kidney stones |
| SCL3A1 | Solute carrier family 3, member 1 | Increased Cystine excretion |
| SLC4A1 | Solute carrier family 4, member 1 | Decrease acid excretion (dRTA) |
| SLC7A9 | Solute carrier family 7, member 9 | Increased Cystine excretion |
| SLC9A3R1 | Solute carrier family 9, subfamily A, member 3, regulator 1 | Calcium phosphate kidney stones |
| UMOD | Uromodulin (most common urine protein) | Protective against kidney stones |
| VDR | Vitamin D (1,25-dihydroxy D3) receptor | Increased calcium excretion |
| XDH | Xanthine dehydrogenase | Increased xanthine excretion |
The Bottom Line
There are many factors that impact the risk of kidney stone development. Although there are pure genetic diseases that are associated with kidney stones, often the increased risk is subtle or offset by other factors. Increased risk, when combined with other factors including nutrient depletion, dysbiosis, electrolyte imbalances and dehydration, may lead to the development of kidney stones in some. Assessing the genetic profile of kidney stone patients can help identify the root cause of the disease to tailor appropriate, personalized management. Practitioners working with individuals to prevent kidney stone formation should formulate a comprehensive and individualized intervention that modifies all relevant components in their integrative approach.
The post Genetics of Kidney Stones appeared first on Integrative Kidney.]]>
As discussed in a previous blog, there are five major types of kidney stones: calcium oxalate, calcium phosphate, uric acid, struvite (magnesium ammonium phosphate), and cystine. Calcium oxalate is by far the most common, comprising approximately 75% of kidney stones. Therefore, the first step in evaluating a kidney stone patient is identifying the type of kidney stone they are forming. This can be done by stone analysis and by obtaining a 24-hour urine analysis for various metabolites and electrolytes.
In this series, we’re focusing on the integrative approach to preventing kidney stone formation.
Conventional approaches to kidney stones typically focus on medications, surgical removal, and using ultrasonic waves to break up a stone. It rarely addresses the root cause including risk factors to prevent stone formation.
We covered dietary approach and microbiome and gut health in previous blogs. In this blog, we will discuss the impact of electrolyte imbalances on kidney stone formation and prevention.
Calcium imbalances
Since most kidney stones are composed of calcium oxalate, oftentimes patients ask if restriction of dietary calcium is necessary. Dietary calcium intake has many benefits including maintaining adequate bone health and decreasing oxalate absorption from the gut. In fact, evidence suggests that restricting calcium intake leads to increased oxalate absorption, in turn increasing the risk for kidney stone formation. Furthermore, there is a significant link between kidney stones and metabolic bone diseases such as osteoporosis, although it is not yet clear if this link is related to calcium imbalances or other causes.
It’s the concentration of calcium in the urine that is important to track. The higher the urinary concentration of calcium, the higher the risk of oxalate binding to calcium (or other minerals) and forming kidney stones.
There are several factors that lead to increased calcium concentrations in the urine, including increased calcium excretion by the kidneys and decreased water intake. Factors increasing urinary calcium excretion include high sodium (salt) intake, genetic factors, certain medications, and certain medical conditions such as disorders of the parathyroid gland.
This explains why restricting sodium intake and increasing water intake are useful for decreasing the risk of stone formation. It is generally recommended that kidney stone formers drink at least 2 liters of filtered free water every day (some require more, especially if they are overweight/obese or losing fluid from physical activity or sweating). In general, we recommend that patients maintain a balanced diet providing1000 mg per day of calcium. . When taken with meals, calcium supplements bind oxalate in the food and prevent it from getting absorbed; however, calcium supplementation is controversial and should be monitored closely by a provider.
Oxalate imbalances
In a previous blog, we mentioned that oxalates play a role in the formation of kidney stones with 75% of kidney stones a result of calcium oxalate formation. As we discussed above, there is a significant connection between calcium consumption and the level of absorbed oxalate. Low oxalate diets have been proposed as a way to minimize stone formation, however, these are incredibly challenging to maintain. Oxalate restriction is challenging because oxalates are present in a large number of many healthy foods, risking nutrient deficiencies. Furthermore, the gut microbiome plays an important role in the absorption of oxalate.
Additionally, there are a few genetic variations in the oxalate gatekeepers in the gut and red blood cell transporters that impact/increase oxalates in the urine. Problems with fat digestion and absorption as seen in small bowel disease, gastric bypass, or pancreatic insufficiency can lead to increased oxalate absorption in the colon. That is why it is important to address gut integrity with a comprehensive gut restoration protocol in patients with kidney stones.
Citrate imbalances
Urinary citrate may be the most significant factor in formation of calcium oxalate stones. Citrate helps increase urinary pH (making it more alkaline or basic), preventing the binding of calcium and oxalate. It also binds calcium in the urine, further preventing kidney stone formation. The concentration of citrate in the urine is dependent on blood acidity, the higher the dietary acid load, the higher the risk of developing kidney stones. This phenomenon may explain why diets high in animal protein have been associated with the development of kidney stones (more about that in another blog here).
In addition, potassium depletion is often associated with pH changes that lead to decreased urinary citrate levels. Potassium depletion can also increase calcium excretion in the urine. Therefore, it is important to correct any potassium depletion in patients with kidney stones. This is why most citrate supplements that are used for kidney stone prevention are usually in the form of potassium citrate.
Magnesium imbalances
Magnesium plays an important role in the prevention of kidney stones. Although the way magnesium decreases the risk of kidney stones is not completely clear, evidence suggests that magnesium lowers oxalate absorption in the gut. Magnesium also increases the level of citrate in the urine which, as we saw above, decreases the risk of kidney stones.
Finally, magnesium can bind oxalate in the urine making it less available for binding with calcium and forming kidney stones. Magnesium oxalate compounds are 100 times more soluble than calcium oxalate. In fact, supplementing magnesium was found to be effective in preventing kidney stones even with patients with normal magnesium.
Uric acid imbalances
Uric acid stones are common in patients with metabolic syndrome and type 2 Diabetes. Elevated uric acid levels in the urine can lead not only to uric acid stones but also to calcium oxalate stone formation. In many instances, calcium oxalate stones have a center (nidus) composed of uric acid. Uric acid is the end product of the breakdown of purine, a product of digestion of nucleic acid in food (meat, poultry and fish) or tissue breakdown. Therefore, limiting the intake of animal protein may be a useful therapeutic approach for prevention of kidney stone formation.
Uric acid forms crystals in highly acidic urine. This is another contributing factor linking a high (animal) protein diet to increased acid load and increased uric crystal stone formation, compounding the impact of uric acid release
The Bottom Line
Many factors impact kidney stone formation, including electrolyte imbalances. Stone formation is usually not caused by a single electrolyte but rather it is likely a combination of factors impacting electrolyte balance, including nutrient depletion and dehydration. Diets low in plant-based foods and high animal protein tend to increase risk of stone formation due to an interplay of factors. Risk of kidney stone formation should be evaluated by weighing the impact of poor dietary choices combined with environmental factors, genetics, gut integrity and microbiome balance.
This is the foundation of our integrative approach to managing and prevention of kidney stones. Practitioners working with individuals to prevent kidney stone formation should formulate a personalized approach that modifies all relevant components in their comprehensive approach for best outcomes.
The post Electrolyte Imbalances and Kidney Stone Formation appeared first on Integrative Kidney.]]>Kidney stone formation (urolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. They are painful, inconvenient, and when left untreated, they may contribute to more serious conditions including obstruction and kidney damage.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Read more about the etiology and prevalence of kidney stones here.
In this series we’re building a case for a more integrative approach to preventing kidney stone formation.
Conventionally, the treatment approach does address kidney stones via a multi-pronged approach that may include medication, dietary and lifestyle, surgical removal, and using ultrasonic waves to break up stone.
However, these guidelines tend to focus too far downstream, on stone composition instead of on the underlying pathology upstream. Instead, we advocate for a more comprehensive approach that focuses on risk factors to prevent formation. Those factors include:
· Type of stone
· Socioeconomic factors
· Diet
· Hydration and electrolyte balance
· Microbiome and gut health
· Genetics
We covered individual dietary components in detail in a previous blog. Today we’ll look at the gut-kidney stone connection and the impact of the microbiome.
Gut Integrity and Kidney Stones: Leaky Gut
A normal and healthy GI tract has a natural barrier. This barrier serves to protect the GI and has three major jobs: 1. ensure proper digestion and absorption of nutrients and 2. ensure elimination of toxins and 3. protect the integrity of the microbiome – the “good” bacteria that lives in our GI tract and works with our body to maintain health.
Leaky gut describes a state when the cells that make up the lining of the GI tract separate enough to allow the contents of the gut to leak out. This is also sometimes called intestinal permeability or IP for short. This is a problem because it reduces absorption of nutrients, causes toxins to build up, alters the balance of the gut microbiome, and results in systemic inflammation.
One of the major contributors to leaky gut is the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
· Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
· Increased intake of processed/refined foods like cereals, crackers, baked goods, etc…
· Processed, fried, conventionally raised, high-nitrate animal protein
· Low intake of fiber and fresh produce in general
· A “beige” diet (low in phytonutrients and antioxidants) from consuming a variety of colorful fruits and vegetables
· Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
We have already established that eating more fresh produce, is protective from kidney stone formation, and we’ve done a deeper dive on specific nutrition impact on kidney stone risk in another blog if you’d like to learn more.
There are several factors that may contribute to development of leaky gut:
· “Proinflammatory” SAD: too much processed and high-sugar foods, not enough fiber and the wrong inflammatory fats
· Food sensitivities: consuming food that are cause reactivity
· Overconsumption of caffeine and alcohol – irritants to gut lining
· Use of certain medications, including NSIADs, steroids, antibiotics
· Stress and poor-quality sleep
We address risk factors for intestinal permeability in more detail in a previous blog here, as well as dive into a comprehensive gut restoration strategy here in this 5-part series.
The Microbiome and Kidney Stones
Balance of the gut bacteria also play an important role in causing or preventing kidney stones. The most studied organism is Oxalobacter formigenes, which has been found to be protective when present in adequate quantities as part of the GI microflora. This bacterium degrades oxalate in the gut decreasing its absorption and excretion in the urine.
When Oxalobacter was discovered, scientists thought they had pinpointed the key to curing kidney stones. They concluded that simply supplementing this missing species should reduce risk of stone formation in susceptible individuals. It would turn out that the connection wasn’t that simple.
More recent evidence points to a more complex picture in the connection between microbiome diversity and kidney stone pathology. The emerging research shows increased risk in kidney stone formation in certain susceptible individuals also presented with alterations in normal microbiome and metabolome (metabolic byproducts from microflora) – also termed dysbiosis.
In other words, it’s likely that genetic factors might be “turned on” by dysbiosis leading to increased risk of kidney stone formation in certain individuals. The good news is that means they should be “turned off” when the microbiome balance is restored.
Studies that looked at the use of targeted probiotics have failed to show enough significant improvement of risk of urolithiasis. Although there’s been some limited and temporary reduction in oxalate excretion and kidney stone formation with the use of a combination of Lactobacillus, Bifidobacterium, Enterococcus, it’s been shown to be temporary and limited in benefit. This is because dysbiosis cannot be addressed by simply applying a band aid of a probiotic.
We recommend instead a more comprehensive approach to gut restoration and microbiome balance. You can read more about the 5R protocol in our comprehensive 5-part series on gut restoration.
The Bottom Line
Although initial findings about the impact of the microflora that looked at Oxalobacter in isolation have not demonstrated significance in reducing incidence of kidney stone formation, more recent evidence pointing to an interplay of factors on microbiome diversity is promising. Furthermore, factors that impact kidney stone formation include dietary factors, including food quality, nutrient composition, and dehydration. Along with environmental factors, lifestyle, genetics, and gut integrity and microbiome balance should be addressed through a comprehensive and personalized approach. Practitioners working with individuals to prevent kidney stone formation should formulate a patient care plan that modifies all relevant components in their integrative approach to maximize effectiveness in preventing urolithiasis.
The post The Microbiome and Kidney Stone Formation appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
Recent studies have focused on the significance of a relationship between gastrointestinal (GI) integrity and microbiome diversity with various chronic diseases including kidney disease.
In previous blogs, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance leading to intestinal permeability (IP or “leaky gut”). The impact of leaky gut on kidney health and progression of chronic kidney disease (CKD) has been referred to as the gut-kidney connection is the result of complex biochemical and immune mechanisms.

A comprehensive approach to CKD includes addressing the health of the GI at the root of it. Furthermore, this means not simply supplementing with probiotics, but instead addressing all the mechanisms that underlie probiotic need. This includes modification of microbiota balance, integrity of the mucosa and epithelium of the GI tract, improving GI motility, absorption and digestion, and modulation of the immune system.
Below we will explore the fifth step, Rebalance. But first, let’s quickly review the first four steps of the comprehensive gut restoration protocol. A reminder that the 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD.
The 5 steps of the 5R protocol for healing leaky gut are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Rebalance
The goal of the fifth step is maintenance and prevention of recurrence of IP or leaky gut. There are various factors that lead to IP that involve dietary and lifestyle influences*, including:
· Standard American Diet (SAD) which is low in fiber, high in processed foods, and highly inflammatory
· Poor eating habits (for example, multitasking and not chewing adequately)
· Inadequate hydration and/or electrolyte imbalance
· Motility issues leading to constipation or unfavorable formation/frequency of stool
· Stress and poor sleep
· Not enough exercise
To reduce risk of CKD, we must work towards improving diet and lifestyle habits that support continued GI health.
*Read more about medications that impact gut health and ultimately increase risk for KD progression here.
Where to start?
Address lifestyle factors that impact gut health
Long term dietary goals focus on a plant-based diet that is high in fiber and a wide range of key nutrients. Organic sources of animal protein can beneficial when eaten in moderation. However, the key is to load up on naturally antiinflammatory, low carbohydrate vegetables to maximize vitamins, minerals, phytonutrients and, of course, fiber. Reduce intake of starchy vegetables and eat more**:
· Dark leafy greens (like spinach, arugula, and romaine)
· Cruciferous veggies (like broccoli, cauliflower, and kale)
· Fresh whole fruit (preferably lower sugar berries and avocados, and limit to two servings per day)
· Some colorful starchy veggies can be OK (for example, carrots, sweet potatoes, beets, and squashes)
Those veggies listed above promote a healthy microbiome and improve alkalinity associated with improved kidney health. In addition to those sources of prebiotic fiber, include probiotic sources like fermented vegetables and drinks like raw sauerkraut, kimchi, pickles, apple cider vinegar, and kombucha. It’s recommended these are eaten raw because pasteurization process will destroy the bacterial content of these foods.
In general, reduce processed carbs, like bread, cereals, and high sugar foods, desserts and pastries. These types of foods are usually low in fiber and nutrients and help contribute to starving of beneficial gut bacteria. Furthermore,high carb diets have been associated with increased risk of cardiovascular (CV) disease and diabetes. Instead, moderately consume whole grains like brown rice, quinoa, oatmeal, and legumes**.
Include antiinflammatory fats focusing primarily on omega 3 sources from fish, nuts, seeds, and mono- and polyunsaturated (MUFA and PUFA) like avocados and olive oil. Even though the topic of saturated fats is more controversial, recent evidence suggest that moderate intake of certain saturated fats like that found in beef, organ meat, or ghee (clarified butter) derived from grass-fed cows and virgin coconut oil might have health benefits. Everyone agrees, however, that trans fats(aka hydrogenated oils and artificial products like margarine) or excessive intake of processed and fried fats contributes to inflammation and increased risk of disease.
Ensuring adequate hydration, drinking at least half your body weight (pounds) in ounces of water, not only helps to maintain good kidney health, it also helps support daily regular bowel movements.
It’s well established that regular exercise can be beneficial for many reasons, including improved blood pressure, blood sugar, hypertension, stress relief, and even improved digestion and GI motility. In fact, exercise has been associated with improved microbiome balance as well as beneficial modulation of the immune system.
Poor sleep quality and stress are also deeply tied to many underlying factors impacting of GI health. Reduced sleep duration and quality has been associated with increased inflammatory markers (including TNF, IL-1, and IL-6) associated with GI disease like GERD and Irritable Bowel Disease/Syndrome (IBD/IBS) disrupting digestion and nutrient absorption. Furthermore, sleep has been shown to affect kidney health directly and indirectly, including associated risk of CV disease, diabetes, obesity, and hypertension(read more about the relationship between sleep and kidney health here).
**NOTE: Due to individual variations and progress of disease, work with a nutritionist to assess if you need to maintain any specific restrictions due to your unique case and needs.
The role of supplements
Because of the unique needs of kidney disease patients, many need to rely on supplements to help obtain adequate amounts of key nutrients to maintain GI and kidney health.
This may include GI and motility support including but not limited to digestive enzymes, bitters, probiotics, and magnesium citrate and triphala for motility). Furthermore certain individuals may benefit from supplementation of certain vitamins and important minerals, high potency antioxidants, and/or support of certain key underlying cellular mechanisms impacting mitochondrial health, detoxification and nitric oxide production.
That said, many factors must be taken into consideration when choosing appropriate supplements for each patient. Supplement quality and contamination are a common concern, as are potential interactions with medication or contraindications in certain commonly associated chronic disease. We suggest working under the care and guidance of a practitioner or team of providers who are trained in integrative and functional medicine and understand the unique needs of kidney patients.
Next steps
Unfortunately, addressing gut health is only the beginning. As mentioned above, kidney disease is often associated with multiple chronic diseases including CV disease, diabetes, high blood pressure, and obesity.
The comprehensive approach to kidney care means addressing the underlying causes of this constellation of diseases which is best accomplished by a Functional Medicine approach. The goal is to identify and then rebalance the biochemical and pathophysiological dysfunction at the root of chronic disease, we can stop the progression of kidney damage and preserve kidney function.
Bottom Line
In the final step in the 5R individualized gut restoration protocol, we Rebalance the foundational factors that impact the gut-kidney axis.
Although this might be the last step in the 5R protocol, within the broader context of kidney disease, it might signal the transition to a comprehensive therapeutic protocol that includes management of the underlying dysfunction associated with related conditions such as CV disease, diabetes, obesity, and hypertension.
Working with an integrative or functional medicine provider is essential to help you navigate the comprehensive program successfully, but can help you stabilize blood sugar, lower blood pressure, lose weight, and reduce the risk of CV and ultimately, KD.
More from InKidney on the gut-kidney connection:
· Comprehensive Gut Restoration Protocol https://old.inkidney.com/2019/07/05/comprehensive-gut-restoration-protocol-ckd/
· Feeding Gut Bacteria in Kidney Disease https://old.inkidney.com/2019/02/20/feeding-gut-bacteria-patients-kidney-disease/
· Kidney-Gut Axis: Nutrition can slow the progress of kidney disease https://old.inkidney.com/2018/09/20/kidney-gut-axis-nutrition-slow-kidney-disease/
· Inflammation, Leaky Gut And Kidney Disease https://old.inkidney.com/2018/10/05/inflammation-leaky-gut-kidney-disease/
· Leaky Gut And Kidney Disease: 6 Classes Of Medication That Might Be Contributing https://old.inkidney.com/2018/10/05/leaky-gut-kidney-disease-medications/
The post The 5R Protocol Part 5: Rebalance appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Kidney stone formation (also called urolithiasis or nephrolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. Anyone who’s experienced them can attest that stones are often very painful. When left untreated, they may contribute to more serious conditions including urinary tract obstruction and permanent kidney damage or CKD.
Read more about the etiology and prevalence of kidney stones here.
Conventionally, the approach to treatment is a multi-pronged approach and may include medication, dietary and lifestyle interventions, surgical removal, and using ultrasonic waves to break up stone. However, dietary guidelines tend to focus on stone composition instead of on the underlying pathology. In this series, we’ve been discussing the combination of upstream risk factors that impact risk of stone formation.
Factors that impact the integrative approach to kidney stones include:
- Type of stone
- Environment
- Diet
- Hydration and electrolyte balance
- Microbiome and gut health
- Genetics
Diet is a topic that deserves a deeper dive! We’ll focus on it here today.
Dietary Approach to Kidney Stone Prevention
The Diet Controversy and Kidney Stones
Dietary interventions are common in addressing kidney stones. The reality is, we need a more comprehensive approach when it comes to preventing kidney stone formation and kidney damage. Dietary approach to kidney stone prevention is one aspect of this comprehensive integrative approach.
First, let’s start by outlining the impact of the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
- Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
- Increased intake of processed/refined foods like cereals, crackers, baked goods, etc.
- Processed, fried, conventionally raised, high-nitrate animal protein
- Low intake of fiber and fresh produce in general
- A “beige” diet (low in phytonutrients and antioxidants) lacking color and low in enough variety of colorful fruits and vegetables
- Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
Interestingly, studies have shown that eating more fresh produce is protective from kidney stone formation. When we consider that the SAD is low in protective foods and the key nutrients found to be helpful in preventing urolithiasis. Let’s examine these factors that impact kidney stones in more detail.
Macronutrient Balance for Kidney Stones (Protein, Fat & Carbs)
Protein
For a long time, it was assumed that stone formation was at least in large part due to excessive protein intake. As a result, those at risk for stone formation were instructed to eat a low protein diet to prevent urolithiasis. However, there are underlying factors that cause a domino effect impacting how the products of protein breakdown lead to stone formation. This includes micronutrient and electrolyte balance, metabolic disorders, digestive abnormalities, and of course dysbiosis (we’ll discuss these in detail in another blog).
It’s true that excessive protein intake can lead to more acidic urine and increased uric acid production (a risk factor for uric stone formation). Elevated protein metabolism alone isn’t a problem in isolation, there’re other upstream factors to consider that make certain individuals more susceptible to stone formation. Furthermore, the source of protein seems to be significant. Animal protein sources seem to play a bigger significance in risk of stone recurrence, while plant-proteins might be protective.
Carbohydrates and Fiber
Though excessive consumption of carbohydrates isn’t recommended, excessive restriction of carbs in people prone to kidney stones should be avoided. We suggest avoiding sources of simple sugars, fructose and high fructose corn syrup (that includes sugar sweetened beverages, pastries, sodas, and even the so-called healthy sweetener agave). Fructose consumption in particular seems to increase production of excess uric acid and simultaneously reduces its excretion in the urine.
Instead, we want to opt for more nutrient-dense, high-fiber, low-glycemic carbohydrates, including root vegetables, berries, whole grains and legumes. Increased consumption of fiber helps to promote improved microbiome health and diversity, which may play a significant role in kidney stone formation risk. Furthermore, there is interesting research that indicates that individuals who aren’t getting enough dietary vitamin C to meet their needs while on a low-carb diet may increase their risk for developing certain types of kidney stones.
This begs the question, what other nutrient deficiencies might contribute to stone formation when macronutrients might not be optimized?
Fats and Essential Fatty Acids
In order to balance a moderate protein and carb intake, some individuals might benefit from increasing calories from fat. Healthy fats include omega 3 sources like fish and fish oil, nuts, seeds, olive oil, and avocado. Grass-fed ghee might be especially beneficial due to a combination of anti-inflammatory profile, vitamin A, and gut healing benefits of the butyric acid component.
Independently, increased fat intake has not been associated with kidney stone formation. However, when coupled with a SAD diet full of simple carbohydrates and fried and processed foods, and excessive protein intake that seems, at least in part, to be a contributing factor. At the same time, when eating a high-fat diet as part of a ketogenic approach, excessive restriction of carbohydrates seems to contribute to stone formation in individuals with existing risk factors. In those cases, it’s recommended to work with a nutritionist to ensure that you’re balancing your macronutrients effectively to avoid kidney stones.
Micronutrient Balance
Fat-soluble vitamins
We can’t talk about dietary approach to kidney stone prevention without discussion fat soluble vitamins. Fat soluble vitamins include vitamins A, D, E, and K. Deficiencies in D, A and K in particular have been associated with kidney stone formation. This might at least in part be due to the fact that these vitamins play a major role in calcium metabolism and bone mineralization. When D and K are deficient, calcium from diet is deposited in arteries instead of in bones, leading to calcification and stone formation.
In fact, vitamin K depletion might be an independent risk factor for kidney stone formation. When individuals form kidney stones, a vitamin-K dependent protein (matrix Gla protein) is secreted in inactive form. This protein when activated with vitamin K can inhibit the growth of calcium oxalate crystals. When vitamin K isn’t available, more crystals may form.
To improve dietary intake of these nutrients, foods like eggs (especially the yolks), organ meats, ghee, natto (fermented soybeans), and full-fat cheeses (assuming you’re able to tolerate dairy) can be helpful to include in your diet. Daily sun exposure (at least 20 minutes a day) can also help promote increased vitamin D levels. However, supplementation is often indicated. Issues with absorbing fats and fat-soluble vitamins due to digestive issues are common, a nutritionist or a clinician trained in Integrative and Functional Medicine can help you determine if supplementation is needed.
Oxalates
Oxalates are front and center in the dietary approach to kidney stone. In fact, calcium oxalate stones comprise most kidney stones (75%). There’s some evidence that limiting consumption of oxalates can help reduce stone formation. Food containing high levels of oxalates tend to be generally considered healthy, including spinach, beets, nuts and seeds, to name a few examples.
What makes these foods healthy for some people, and contribute to stone formation in others?
One reason might be inadequate calcium intake from the diet. Food sources like broccoli, sardines and canned salmon, and leafy greens like kale and collard greens can be good low-oxalate, calcium-rich food sources. When consumed in adequate amounts, calcium binds to oxalates in the intestines, preventing it from being absorbed and reducing formation of stones.
Another important consideration is the composition of the microbiome. When oxalate-degrading bacteria in the gut is inadequate, it can increase risk of developing kidney stone disease. More about that in a future article.
Vitamin C
Ascorbic acid has been associated with formation of kidney stones. Excessive intake of vitamin C might increase oxalate production. Some studies have shown higher intake of vitamin C is associated with increased risk of kidney stones, but only in people predisposed to forming oxalate-stones. Regardless, caution should be used in supplementing vitamin C within the context of the larger, integrative picture.
Phytate
Phytate, sometimes called phytic acid, is an antioxidant compound found in certain foods like beans, lentils, nuts, and seeds. Some have claim that phytate intake might prevent the absorption of certain nutrients, but actually it might be protective for kidney stone! One study found that women who consumed more phytate had reduced risk of kidney stone formation. Other studies have come to the same conclusion. This might account to the benefit of eating these particular foods, which happen to be good plant-based protein sources, in place of animal protein.
Salt and Other Electrolytes
Calcium is a controversial topic when it comes to risk of kidney stone formation. Surprisingly, higher intake of dietary calcium (from food sources like leafy greens, salmon, and legumes, for example) is associated with lower risk of stone formation. On the other hand, taking calcium supplements may be linked with increasing risk of kidney stone formation (this might be gender specific to men).
Furthermore, the conversation often fails to address the nuance of calcium consumption in the form of dairy, which might not be superior over plant-based forms of calcium, or to account for the impact of vitamin D deficiency.
Magnesium is another electrolyte that is often overlooked. Magnesium is part of hundreds of the body’s biochemical reactions, so adequate intake is essential. Furthermore, ensuring adequate intake of magnesium can help prevent the formation of calcium oxalate stones by destabilizing the bond between the two compounds causing them to split.
Food sources of magnesium include swiss chard, spinach, almonds, and pumpkin seeds. Many of those foods might be contraindicated in a low-oxalate diet, so supplementation might be indicated before gradually adding those foods back as part of a comprehensive approach. Supplementing with chelated forms of magnesium (like magnesium citrate, malate, or bisglycinate) or making time for an epsom salt bath (magnesium sulfate in the salts absorbs through the skin).
But the most important electrolyte in the dietary approach to kidney stone is sodium. Excessive sodium (salt) consumption has been associated with stone formation, particularly when fluid intake is inadequate. High sodium excretion increases calcium deficiency by increasing excretion through the urine. Reducing excessive intake of sodium, especially from processed foods like cured meats, can be helpful, along with ensuring adequate intake of potassium-rich foods like broccoli, leafy greens, squash, potatoes, mushrooms, bananas, cantaloupe, and grapefruit to name a few.
More about the impact of electrolyte balance on kidney stone formation in a future blog.
Water (the forgotten macronutrients)
Inadequate water intake is one of the most significant contributing factors to kidney stone formation. The process of excretion of oxalate consumes salt and water, as a result dehydration is more likely.
Taking mineral compounds containing citrate (like magnesium citrate or potassium citrate) can prevent the formation calcium oxalate and uric acid stones while balancing mineral needs. Adding lemon juice or apple cider vinegar (4oz per day) to water might also help prevent urolithiasis through the same mechanism. Your Integrative or Functional Medicine clinician can help create a plan that best fits your needs.
The Bottom Line
Multiple factors impact kidney stone formation, including dietary factors. It seems there’s no single food or food component that causes stone formation. Rather, there are likely a combination of factors including nutrient depletion, dehydration, and poor food choices combined with environmental factors, genetics, and gut integrity and microbiome balance which combined play a role.
Therefore, the integrative approach to addressing kidney stones must account for the constellation of these factors. Practitioners working with individuals to prevent kidney stone formation should formulate a personalized approach that modifies all relevant components in their integrative approach. We’ll explore these connections in future blogs in this series.
The post Dietary Approach to Kidney Stone Prevention appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms. Here we discuss the Replace step of the 5R protocol for kidney health.
In our previous blog, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance. Intestinal permeability (aka “leaky gut”) is the result of inflammatory assault to the gut lining that results in breaks in the integrity of the wall. Imagine it like breaks in a “fence” that let undigested food, bacteria, and metabolites “leak” through the holes.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The physiologic changes associated with this leaky state include a combination of factors including hypochlorhydria (insufficient stomach acid to break down food adequately), reduced production of digestive enzymes, altered bowel motility (often leading to constipation, but not always), and dysbiosis.
As a result of these pathologies, leaky gut leads to increased local and systemic inflammation, poor digestion, reduced nutrient absorption, and reduced ability to remove toxins and digestive byproducts.
Though we address step two, Replace in more detail below, let’s review the five steps of the comprehensive gut restoration protocol. The 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD. The five areas of GI mucosal integrity:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate to provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Replace
Due to diet quality, food sensitivities, pathogenic microbes, epithelial inflammation leads to damage to the gut mucosa integrity. This causes damage to the structure of the villi and reduction in the intestinal surface area leading to reduced absorption of nutrients. This is referred to as malabsorption, or poor absorption of nutrients across the gut wall into circulation to be used by the body.
A second pathology is maldigestion, or ineffective breakdown of nutrients from food. This might be one or more of the major macronutrients, fat, protein, and/or carbohydrates. In these cases, you’ll notice GI distress after eating, bloating, distention, and or excessive flatulence. This not only reduces the effective absorption of essential nutrients necessary for healthy metabolism, but also creates a backup that contributes to increased inflammation, buildup of endotoxins, and further contributing to dysbiosis.
Furthermore, poor motility can contribute to chronic constipation. It’s been estimated that 20% of the adult population suffers from chronic constipation – that’s 1 in 5 adults! Ideally bowel movements should be daily, easy to pass and as characterized by the Bristol Stool Scale below, smooth soft and formed.
Constipation is not only uncomfortable, but leads to inefficient removal of toxins, recirculation of endogenous hormones, and alteration of the microbiome balance. Furthermore, stool consistency and frequency can be used as clues for clinicians signaling the need for a gut restoration protocol. From there, integrative and functionally trained practitioners can use advanced stool testing that employs a technique called PCR technology as discussed in Part 1 Remove blog.
Where to start?
Assess Nutrient status
Often, we begin by assessing the baseline nutrition status for any micro- or macronutrient deficiencies. These tend to be more significant with long-term maldigestion and malabsorption, depriving the body of necessary vitamins, minerals, amino acids, and essential fats needed for healthy function. Supplementation is often necessary, but dietary adjustments can be made to optimize absorption from food as long as digestion and motility are also addressed (see below).
Assessment can be done by physical exam to detect signs and symptoms of nutrient deficiency – for example dry hair and skin, brittle nails, bleeding gums, and difficulty digesting animal protein or fat can all be indications of various nutrient malabsorption/depletion or maldigestion.
Furthermore, laboratory assessment of nutrient status can confirm specific deficiency and help us target key nutrients strategically to customize the therapeutic approach.
Address Maldigestion and Malabsorption
To enhance digestion, use of broad-spectrum digestive enzymes such as lipase and bile salts, proteases, and cellulases that break down fats, proteins and carbohydrates, respectively is necessary because the digestive tract isn’t producing sufficient enzymes to breakdown and absorb nutrients form food. In addition, betaine HCL, a digestive aid that address low hydrochloric acid (hypochlorhydria) in the stomach may also be necessary to improve gastric pH.
Herbs and foods can also be useful for improving nutrient absorption and motility. For example, pineapple, papaya, and bitter foods like dandelion greens can be helpful for aiding digestion, stimulating motility and digestive enzyme production. Eating beets and staying hydrated promotes bile salt flow. Fermented and fiber-rich foods can also help promote motility and healthy microbial balance (though sometimes we don’t introduce these until step 3: Reinoculate). Last but not least, traditional concoctions of herbal bitters and teas can be very helpful in promoting this process.
Address Motility
We cannot emphasize enough how important it is to have at least one daily bowel movement for overall health. That is why integrative and functional practitioners will often prioritize resolving constipation. Bulk laxatives, stool softeners, and stimulants can be useful as a short-term strategy but are only a band-aid. It’s important to understand the underlying causes and address it: slow motility and dysbiosis.
Restoring stool frequency and consistency to ideal can be a challenging and time-consuming process with involves multiple aspects of the 5R protocol. Useful tools include supplements like magnesium citrate, triphala, Swedish bitters, electrolyte replacement and hydration, increasing fiber (dietary and supplemental) as well as stimulating the migrating motor complex and balancing the HPA axis (stress response) through lifestyle modifications including exercise, massage, gargling and stress management.
Bottom Line
The second step in an individualized comprehensive gut restoration protocol involves promoting improved nutrient status and motility by exogenously replacing the necessary enzymes and nutrients needed. This is often done simultaneously to the Remove step, but in some cases, it’s addressed in sequence. It’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this successfully.
Next, we will tackle the third “R” in the gut restoration protocol: Reinoculate.
The post The 5R Protocol Part 2: Replace appeared first on Integrative Kidney.]]>





