Colchicine for CKD!
In a multi-center, nested, case-control study in three Korean hospitals, patients with CKD stage 3 and 4 who are using drugs including colchicine, allopurinol, and febuxostat for high uric acid or chronic gout were studied over a period of 10 years. The progression of CKD was compared between 3085 compared to 11715 control patients.
Colchicine use was associated with a lower risk of adverse kidney outcomes in CKD patients with hyperuricemia, or chronic gout.
Unlike a study published two years ago in NEJM which excluded patients with advanced CKD, this study included patients with kidney function as low as 15 ml/min. Colchicine is known to anti-inflammatory. It also protects against kidney fibrosis.
There are concerns about myopathy and neuropathy with the intake of colchicine. It is, therefore, important to adjust the dose with advanced kidney disease and to be cautious when using it with patients who are on other myopathy-inducing drugs such as statin drugs.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Long sleep duration is associated with decline in kidney function
This study is retrospective, longitudinal cohort study included 82,001 participants who visited a primary care center in Japan. Patients were categorized into CKD risk groups and sleep duration categories according to their self-reported average nightly sleep duration. The relationship between average nightly sleep duration and the incidence of composite renal outcome was studied.
Researchers found that an average sleep durations ≥8 h/night were associated with an increased risk of kidney function decline over time.
There are many reasons that connect sleep problems with poor kidney function. We summarized these in this blog.
Risk of environmental heavy metal toxicity is higher in CKD
In a study of 5,638 NHANES participants, lead and cadmium levels were higher in patients with CKD than those without it. This was also associated with decreased urinary lead excretion. Each decrease in estimated GFR by 10 ml/min/1.73m2 was associated with 0.05 mcg/dL increase in lead levels and 0.02 mcg/dL of cadmium levels. This association was even stronger among black participants.
The study concluded that CKD increases the susceptibility to heavy metal environmental exposure by reducing its elimination.
Download Your Copy!
Join here to receive FREE monthly updates on the latest research in Integrative Nephrology, and tips on managing kidney disease straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post May Research And News appeared first on Integrative Kidney.]]>

Root Causes of Itching in Kidney Disease
Uremic toxin-induced neurologic changes
Several studies showed that patients with CKD have an imbalance of opioid receptors in the central and peripheral nervous system. Interestingly, itching in CKD correlates better with the level of uremic toxins than with glomerular filtration rate (GFR, a measurement of how well the kidneys are working). These toxins play an important role in these neurologic changes that are common in CKD. Most of these toxins are generated by the gut microbiome and are excreted by the renal tubules and not by glomerular filtration (or GFR). Measuring these toxins can be helpful. This can be done by a metabolomic test such as Genova’s Metabolomix+.
Download Your Copy!
Inflammation
CKD is an inflammatory disorder. Elevated inflammatory markers have been found to be associated with itching in kidney disease. Measuring inflammatory markers such as hsCRP, Th-1 cells, and IL-6 can be helpful in the assessment of the root cause of itching in CKD.
Dehydration
Dehydration can also lead to dry skin and worsens itching in CKD patients. Correcting the dehydration can be important in the management of itching.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Drug-induced itching
Many medications that are used in CKD have been associated with itching. These include calcium channel blockers, hydrochlorothiazide, and ACE inhibitors. In addition, other medications such as opioids, anticoagulants, and antibiotics can cause itching.
The itch-scratch-itch cycle
One of the major problems with itching is that it leads to scratching, and this can cause skin damage. The latter can cause further itching and the cycle continues.
Natural Treatments for itching in kidney disease
There are many steps you can take to get rid of itching in kidney disease and a kidney disease rash. The first step is to visit a healthcare provider to help you determine the root cause. You can download this two-page handout to guide you in the natural treatment of itching. There is hope that you’ll relieve this frustrating problem and have comfortable skin once again.
The post Root Causes of Itching in Kidney Disease appeared first on Integrative Kidney.]]>
This routine should take no longer than an hour. Your kidneys and health deserve an hour from you every morning. I understand many of us have hectic lives, but if you are busy, try to go to bed early and wake up one hour earlier the next morning. Trust me, it is worth it. An hour morning routine beats dialysis any time. This could be the first step in how to heal your kidneys naturally.

Start with water
At night the body loses a lot of water due to sweat and breathing. In fact, in a small study presented at an Associated Professional Sleep Societies meeting in 2009, healthy young men were found to lose ¼ lbs. per hour while asleep. That is about 2 lbs. in eight hours of sleep.
This is why you should start your day with water. The rest of the routine can be done according to your judgment and comfort. Start with drinking two glasses of filtered water if you are not on any water restrictions. This is even more important for patients with polycystic kidney disease and kidney stones. If you are wondering how much water to drink in PKD, check out our calculator here.
Exercise
Exercise for about 20 minutes. The type and duration of exercise depend on an individual’s level of functioning, endurance, and preference. Studies on exercise in chronic kidney disease (CKD) have included resistance training and aerobic activities that use large muscle groups continuously such as walking, cycling, and jogging.
High-intensity interval training (HIIT) was shown to offer superior benefits in individuals with metabolic disease. In the few studies of HIIT in CKD, it was found to be a safe and feasible option for individuals with CKD.
If you are new to exercising, start slow and try to gradually get to 20 minutes of strenuous exercise five to six days a week. It is best to alternate between aerobic exercise and resistance training. If you absolutely can’t fit exercise into your morning routine, you can move it to whatever time that works for you. The benefits are all the same. Consistency is the key here.
Download Your Copy!
Meditation
Meditate for at least 10 minutes. Meditation is a great way to start your day, manage stress, and stimulate your vagus nerve, which connects the brain with many vital organs including the kidneys. There are many ways to meditate and many types of meditations. Mindfulness meditation is the most tested in kidney patients. It has been shown to improve the quality of life, reduce anxiety and depression, reduce sympathetic overactivity, improve sleep, and improve blood pressure.
To practice mindfulness meditation such as Benson’s relaxation technique, sit in a comfortable position or on a meditation cushion, close your eyes, relax your shoulders and muscles, and focus on your breathing. Say a word with every exhalation. Pick a positive word like “gratitude.” As your mind starts racing to distract you from your breath, try to return to the word you chose. Practicing this every day will make you better at it, and you will be able to reap all the benefits with consistency.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Journaling
Journaling is a great way to write your thoughts and plan your day. It has similar benefits to meditation. In fact, journaling was found to reduce stress and depression, boost the immune system, and improve memory among many other benefits. You can write whatever comes to mind. Think of it as a thought download. This should also not take more than 10 minutes.
Meditation and journaling can also be moved to a different time of the day for busy individuals with similar benefits. If you are very busy, consider meditation in the morning and journaling in the evening.
Cold shower
Taking a cold shower has many benefits for kidney patients. It increases endorphins and fights off depression, and it improves metabolism and circulation. Most importantly, it stimulates the vagus nerve and improves renal circulation. It may also soothe itchy skin, which is common in CKD patients. Cold showers help with post-workout muscle soreness. The benefits of a cold shower begin when the water temperature dips to 60 degrees Fahrenheit. This is 40 degrees lower than the typical steamy shower.
Now, I know it will be hard to turn that shower knob from hot to cold in the morning. The way to do it is to start with your hot shower. Then gradually lower the temperature at the end of the shower every day by turning the knob to cold. Give your body time to adjust. You can start with 30 seconds under the cold and gradually increase it to a maximum of three minutes. A quick shower for kidney health takes 10 minutes.
Plant-based breakfast
A good plant-based breakfast can be a super healthy way to start your day. A plant-based or plant-dominant diet has been linked to better kidney health and to slow the progression of kidney disease. There are many options that you can prepare in less than 10 minutes or plan the day before. You can find a few recipes in this e-book.
The bottom line
Your kidneys and health deserve an hour from you every morning. This morning routine can improve kidney health and help slow the progression of early kidney disease. If you’re wondering how to heal kidneys, this routine could serve as the first step, or it could be all you need for heathier kidneys. You can modify it to fit your schedule so that you’re at least doing some of the suggested morning actions every day. If you’ve been wondering how to improve kidney function, this morning routine is an important step in the right direction.
The post How To Improve Kidney Function with a Morning Routine appeared first on Integrative Kidney.]]>
3 years of lifestyle interventions improved exercise capacity and decreased the losses in neuromuscular and cardiorespiratory fitness in CKD patients
In this study, researchers randomized 161 patients with stage 3-4 CKD to either get usual care or usual care plus lifestyle “intervention” for 3 years.
The lifestyle intervention comprised of care from a multidisciplinary team, including a nephrologist, nurse practitioner, exercise physiologist, dietitian, diabetes educator, psychologist, and social worker.
The patients were coached for 8 weeks and then followed for 34 months with a home-based program.
The study did not look at the progression of CKD but it found that a 3-year lifestyle intervention doubled the percentage of CKD patients meeting physical activity guidelines, improved exercise capacity, and decreased the losses in neuromuscular and cardiorespiratory fitness.
It appears that the study mainly focused on exercise. So imagine the benefit of a comprehensive lifestyle modification plan that includes nutrition, exercise, stress management, sleep improvement, and attention to toxin exposure and gut-kidney connection. That’s what we focus on.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Curcumin powder did not improve markers of vascular dysfunction in PKD
This is essentially a negative study.
It demonstrated that Curcumin powder did not improve markers of vascular dysfunction in children and young adults with PKD. The study was conducted for only one year using a dose of 25 mg/kg per day of curcumin.
This is a classic supplement or nutrient study that is usually underpowered or conducted for short periods of time for a disease that takes years or even decades to evolve. Nevertheless, the study proved that short-term use of curcumin is not beneficial for vascular health for young patients with polycystic kidney disease.
Low serum zinc levels were associated with infections in CKD patients
This did not really need research but it is now studied and it is official: Low zinc levels in patients with CKD lead to infection (..well among other things).
This retrospective study analyzed data from 299 CKD patients who had serum zinc levels checked to evaluate anemia. They used the level of 50 mcg/dl as the cutoff between low or “high” zinc values.
Low serum zinc values remained an independent risk factor for infection-related hospitalization. This was especially true for patients taking proton pump inhibitors (PPIs) medications.
Read about the effect of Zinc on kidney health in this blog.
Download Your Copy!
Join here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to here your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post January Research and News appeared first on Integrative Kidney.]]>
Altered vitamin K biodistribution may decrease the benefit of vitamin K2 supplementation in advance CKD
In this study, investigators sought to determine if there are other causes for vitamin K deficiency in advanced CKD beyond decreased dietary intake. They compared vitamin K uptake and distribution into circulating lipoproteins after a single administration of vitamin K1 plus K2 (MK-4 and MK-7) between patients on dialysis and healthy individuals.
They found that patients with uremia and advanced kidney disease don’t incorporate MK-7 well into HDL and LDL particles compared to healthy individuals. In addition, the combination of a statin and PPI was associated with signs of functional vitamin K2 deficiency in these patients.
In essence, patients with advanced kidney disease may not benefit as well from vitamin K2 supplementation. This highlights the importance of optimizing vitamin K2 status at earlier stages in CKD.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Higher levels of deoxycholic acid were associated with a higher risk of progression in CKD
Deoxycholic acid is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. Intestinal bacteria metabolize the primary bile acid, cholic acid, into deoxycholic acid (DCA).
Researchers studied 3,147 CRIC study participants who had fasting DCA levels. DCA levels above the median were independently associated with higher risks of ESKD and all-cause mortality.
This study highlights the importance of the microbiome and dysbiosis in the progression of kidney disease as we discussed in our blog.
Download Your Copy!
The higher number of medications a kidney patient takes the faster her kidney disease progresses
In a study performed in Japan of 1117 CKD patients under nephrological care, the use of a higher number of medications was associated with an increased risk of kidney failure, cardiovascular events, and all-cause mortality in patients with CKD. This is one of the major reasons we advocate for lifestyle modifications and coaching as the first and major step in the management of kidney disease.
Subscribe here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post December Research and News appeared first on Integrative Kidney.]]>
Low magnesium levels have been associated with a number of adverse events, such as high risk for heart disease. However, little is understood about magnesium and kidney health. Here, we will discuss the potential benefits of magnesium on the kidneys. This is one of two articles on magnesium and kidneys. For more on how to test and treat kidney patients with magnesium deficiency, see part two, “Magnesium Deficiency: Assessment and Management for Better Kidney Health.”

Dietary sources of magnesium
A daily intake of 3.6 mg/kg is necessary to maintain magnesium balance in humans under normal conditions. This is estimated to be between 320 to 420 mg/day (13–17 mmol/day) for adults. Sadly, there has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is due to depletion of magnesium in soil over time. This, along with the rise of ultra-processed food, sodas, and taking medications such as proton pump inhibitors and diuretics that deplete magnesium levels (polypharmacy), has led to rising prevalence of magnesium deficiency.
Traditionally, the highest food sources of magnesium are:
- Leafy greens (78 mg/serving on average)
- Nuts (80 mg/serving on average)
- Pumpkin seeds have the highest level of magnesium per serving (156 mg).
- Whole grains (46 mg/serving on average)
A complete list of foods high in magnesium can be found here.
Can Magnesium Help Kidney Function?
There are many potential benefits of magnesium for kidney health including improving blood pressure control, insulin sensitivity, bone health, vascular health, and preventing kidney stones. Let’s explore the data.
Magnesium and blood pressure control
Magnesium supplementation may help reduce blood pressure (BP) by increasing the production of nitric oxide. Nitric oxide acts as a signaling molecule that helps relax blood vessels, which lowers BP. In fact, a review of 34 studies showed that supplementing magnesium with an average dose of 368 mg per day for 3 months can decrease systolic BP by 2.00 mmHg and diastolic BP by 1.78 mmHg. This supplementation was accompanied by 0.05 mmol/L increase in serum magnesium levels.
Download Your Copy!
Magnesium and insulin sensitivity
Diabetes is one of the major risk factors for kidney disease worldwide. Higher dietary intake of magnesium has been correlated with lower diabetes incidence. A review of 18 studies in people with diabetes showed that magnesium supplements reduced fasting plasma glucose levels. In people who are at high risk for diabetes, magnesium supplementation significantly improved plasma glucose levels after a 2-hour oral glucose tolerance test. These effects are thought to be due to the effects of magnesium on insulin receptors and signaling that allows for improvement in glucose transport and utilization.
Magnesium and vascular health
Magnesium levels have been associated with a lower incidence of cardiovascular disease. In fact, supplementing with magnesium was associated with improvement in vascular flow and endothelial function. Endothelial function refers to the lining of the blood vessels, which is involved in regulating blood vessel health and blood clotting.
Studies in patients receiving dialysis have shown that having a lower serum magnesium level is a significant risk for cardiovascular mortality. Laboratory data show that magnesium inhibits high phosphate-induced calcification of vascular smooth muscle cells. Calcification of arteries is a strong predictor of heart disease and heart-disease-related death.
Magnesium and vitamin D
Magnesium is essential to vitamin D metabolism. Vitamin D that we eat or make in our skin from sun exposure circulates in the blood and is bound to vitamin D binding protein (VDBP). VDBP binding activity depends on adequate magnesium levels. In addition, magnesium is an essential cofactor for the enzymes that activate vitamin D. Studies have demonstrated that magnesium deficiency is associated with impaired vitamin D metabolism.
On the other hand, taking large doses of vitamin D can induce severe depletion of magnesium. This is thought to be due to the overutilization of magnesium. Therefore, adequate magnesium supplementation should be an important part of vitamin D therapy.
Adequate magnesium supplementation should be an important part of vitamin D therapy.
Share on X
Magnesium and bone health
Besides magnesium’s effects on vitamin D metabolism, it is an essential component of hydroxyapatite, an essential component of bone and teeth. In fact, 60% of total Mg is stored in the bone. Low magnesium intake was found to be associated with lower bone mineral density in postmenopausal women. Magnesium deficiency contributes to osteoporosis directly by acting on crystal formation and on bone cells and indirectly by impacting the secretion and the activity of parathyroid hormone (PTH) and by promoting oxidative stress and inflammation.
In addition, a review of 8 studies looked at magnesium and chronic kidney disease (CKD). The study investigated magnesium supplementation on parameters of CKD-related mineral bone disease (CKD-MBD). Mg supplementation improved PTH levels and carotid intima-media thickness (CIMT). Low serum Mg levels were also found to impact PTH and worsen osteoporosis in CKD patients, particularly with diabetes.
Magnesium and kidney stones
Mg acts as an inhibitor of calcium oxalate crystallization and stone formation in the urine. It also decreases the absorption of dietary oxalate in the gut. Mg supplementation in patients with kidney stones was found to decrease the incidence of stone formation even in patients without signs of Mg deficiency.
Magnesium as a phosphate binder
Hyperphosphatemia (high phosphate level) is common in advanced kidney disease. Many kidney patients with stage 4 and above use binders that bind phosphate (or “phosphorus,” as it is commonly known) in the food and prevent it from getting absorbed. High phosphate levels have been associated with poor bone and vascular health in kidney patients. In fact, higher dietary phosphate load can be seen in earlier stages of CKD, and it can do harm even before it is detected.
Magnesium carbonate has been successfully used as a phosphate binder. Magnesium based phosphate binders were also found to reduce vascular calcifications in rats with kidney disease. Iron-magnesium hydroxycarbonate was also studied and found to be well tolerated and can effectively lower phosphate levels in dialysis patients. It is essential to know that most of the magnesium used as a phosphorus binder will not be absorbed.
The bottom line on magnesium and kidneys
Magnesium is essential to many biological functions. It has many health benefits for kidney, bone, and vascular health. Optimizing magnesium status is, therefore, an important step in the integrative approach to kidney health. In part two of this blog, “Magnesium Deficiency: Assessment and Management for Better Kidney Health,” we will discuss practical steps for figuring out a person’s actual magnesium status, the best form of magnesium to take, and the dose I recommend for each condition.
The post Magnesium and kidneys appeared first on Integrative Kidney.]]>
Lifestyle modifications do indeed prevent CKD and delay its progression
This study is coming from Japan. Researchers found that two years of healthy lifestyle choices prevent against the development of kidney disease and protein in the urine. Lifestyle modifications also slowed the decline in kidney function.
They studied 451,534 people (277,494 men and 174,040 women) aged 20–79 years. Healthy lifestyle choices in this study were defined as: 1. No smoking 2. Healthy eating habits, and 3. Low BMI
Now, we all know that there is more to lifestyle modifications than this but this is definitely a start.
Is Urea by itself toxic?
The authors in this study published in NDT definitely think so. They noted that the accumulation of uremic toxins is a risk factor for mortality and accelerated atherogenesis in patients with CKD. The researchers found that a post-translational protein modification called carbamylation is a predictor of CKD progression. They also noted that urea by itself promotes the carbamylation of proteins.
So urea is not just a benign marker and should be classified as a uremic toxin. The level of protein carbamylation may be used as a marker for risk stratification of CKD outcomes.
Download Your Copy!
Magnesium, bones, and kidneys… What?!
This study is coming from the dialysis world. It’s a meta-analysis that examined the effects of magnesium supplementation on chronic kidney disease mineral bone disease (CKD-MBD). As the researchers mentioned, magnesium supplementation in dialysis patients is controversial. However, this study showed that it helps by regulating calcium and parathyroid hormone and it decreases carotid intima-media thickness.
This is in the dialysis population. So paying attention to magnesium balance and supplementation may have a much better role in bone and vascular health in earlier stages of CKD. Optimizing vitamin D, vitamin K2, vitamin A, calcium, phosphorus , and magnesium is very important to improve bone and vascular health in CKD patients.
Subscribe here to receive a monthly update on the latest research in Integrative Nephrology.
The post October Research and News appeared first on Integrative Kidney.]]>
We discussed the effect high intake of animal protein has on kidney health in a previous blog. According to the National Institute of Health, the Dietary Reference Intake (DRI) of protein for the average person is 0.8-1.6 g/kg/day (or 0.36-0.75 g/lb/day). However, according to the Kidney Disease Improving Global Outcomes (KDIGO) guidelines, kidney patients should limit their protein intake. A low protein diet (LPD) by the KDIGO guidelines is 0.8 g/kg/day (or 0.36 g/lb/day). That means a kidney patient who weighs 150 lbs (68 kg) has a dietary protein target of approximately 54 grams of protein per day (0.8g/kg x 68kg). Furthermore, it’s suggested that at least half of that comes from plant-based sources. Protein intake, along with other related nutrients, should be tracked closely through nutrient intake analysis, ideally by a nutritionist.
Different dietary protein regimens have been proposed in the literature for patients with Chronic Kidney Disease (CKD):
- A low-protein diet (LPD), with protein target of 0.6-0.8 g/kg/day;
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with essential amino acids, or
- A very low-protein diet (VLPD) (0.3 g/kg/day) supplemented with nitrogen-free keto-analogues of essential amino acids.
The controversy in the literature, however, has always been whether or not lowering the targeted protein intake puts patients at risk for nutritional deficiency, including essential amino acids and other nutrients often found in high protein foods like folate, B12, carnitine, and zinc. And in that case, is supplementation among these patients necessary and useful to maintain adequate nutrition?
Let’s focus on amino acids, but first let’s review.
What are essential amino acids?
Amino acids (AA) are the basic organic building blocks of protein. There are a total of twenty-two AA, each composed of a core structure made of nitrogen, carbon, hydrogen and oxygen, and variable side chain groups unique to each AA.
Of the twenty-two AA, there are nine essential amino acids (EAA). They’re called “essential” because the human body cannot make them, and therefore we must get from our diet. These EAA are:
- Histidine
- Leucine
- Isoleucine
- Valine
- Lysine
- Threonine
- Methionine
- Phenylalanine
- Tryptophan
The remaining thirteen AA are termed “unessential” not because they’re not important, but because the body can make them through various biochemical processes as long as the necessary building blocks are available from the diet.
When restricting protein intake in kidney patients, it is important that the patient receive adequate intake of EAA since adequate levels of these essential nutrients can only be maintained through food. This can be challenging to keep balanced, which is why we suggest working with a nutritionist to track and support adequate intake.
Using ketoacid analogues with very low protein diet for kidney health
To understand the relevance of ketoacid analogues in CKD, we need a biochemistry lesson in AA metabolism. Biochemistry isn’t everyone’s thing, so we summarized it here.
One thing to remember about AA metabolism is that it requires cofactors which are nutrients that facilitate the reactions. Vitamin B6 is a major cofactor here. Other cofactors needed in the amino acid breakdown (catabolism) include biotin, folate, and B12.
How do ketoacid-analogue supplements help in kidney disease?
Theoretically, the process described above can be used in reverse to reduce circulating nitrogen and the formation of nitrogen waste products (i.e., like urea), and, therefore, decrease the burden on the diseased kidneys. Supplementing ketoacid analogues of EAA in kidney patients on a LPD and VLPD, might simultaneously achieve two goals: 1. It closes the gap on adequate EAA nutritional need 2. It decreases the toxic nitrogen waste products contributing to disease complications. There is also some evidence that certain analogues can decrease protein breakdown in the body.
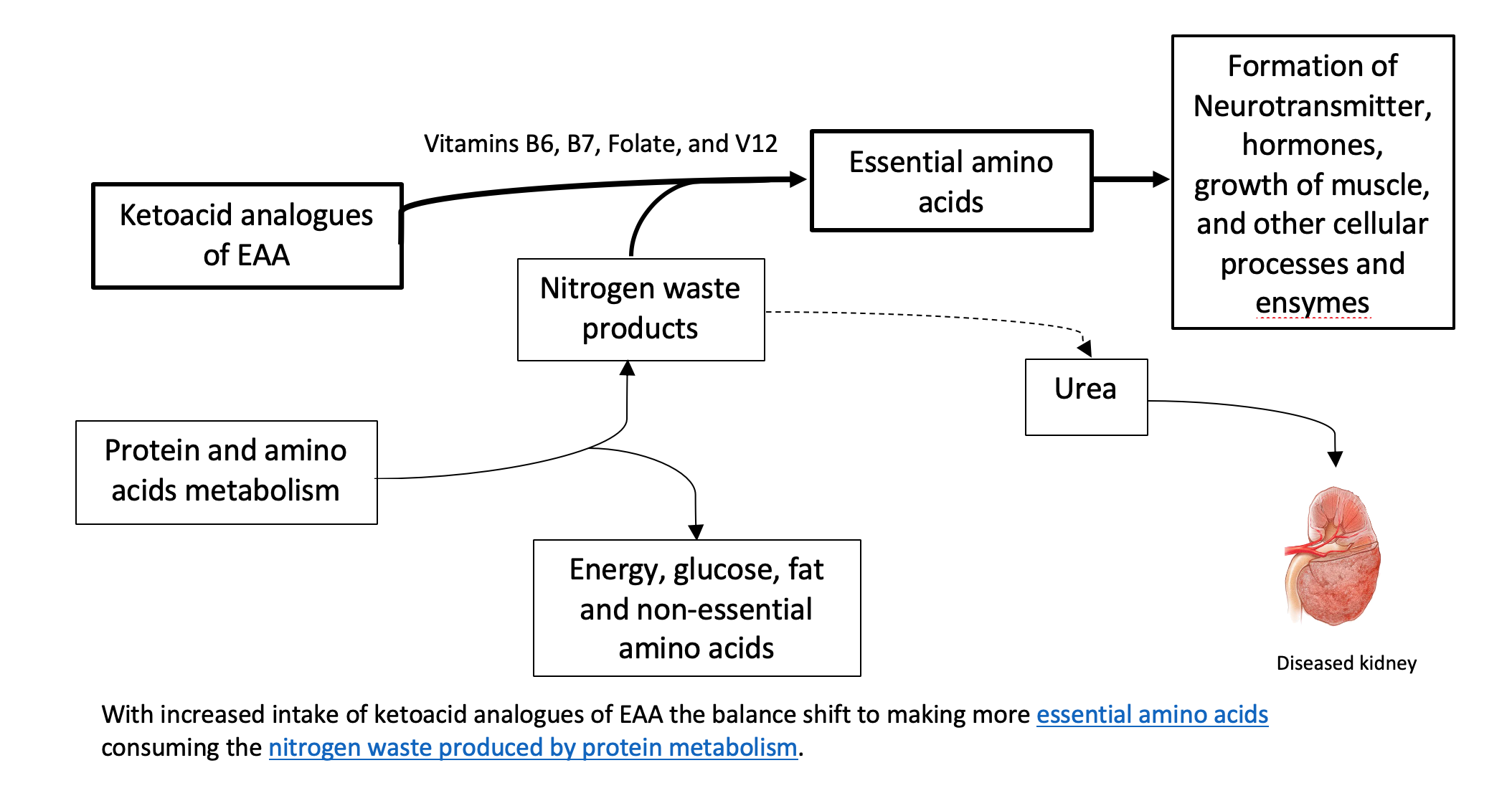
Some studies have demonstrated that very low protein diets supplemented with ketoacid analogues decrease waste product generation in kidney disease and prevent protein-energy decline. Supplementation of keto-analogues in kidney patients was found to decrease protein in the urine as well as improve bone health, insulin sensitivity, and blood pressure control. In addition, these dietary protein restrictions with keto-analogue supplements have been found to delay symptoms of kidney failure and the need for dialysis.
However, one of the major concerns of supplementing ketoacids in kidney patients on VLPD is that it does not address the other micronutrients and cofactors that need to be supplemented. it also does not take into account the carbohydrate and healthy fat content of a healthy diet.
Ketoacid analogue preparations
There are a few different preparations available in Europe, but only one available in the US. If one looks at the content of these preparations, it will become obvious that the kidney patient will need several pills of ketoacid daily to achieve an adequate daily dose. This makes this therapy very expensive and impracticable.
Luckily, there’s a way to use diet to balance macronutrients to induce the same effect. A low protein diet, with the majority of protein coming from whole food plant sources. When consuming animal protein, we recommend organic and grass-fed sources. Strictly vegetarian patients can still get adequate sources of EAA if they plan their diet well with the help of a nutritionist. If AA supplements are determined to be needed, one should aim to achieve the recommended daily intake from a trusted source (with good quality control). According to the World Health Organization, the recommended daily dose of each of the EAA is as follows:
| Essential Amino Acid | Recommended daily intake |
| Lysine | 30 mg/kg/day |
| Leucine | 39 mg/kg/day |
| Isoleucine | 20 mg/kg/day |
| Valine | 26 mg/kg/day |
| Threonine | 15 mg/kg/day |
| Phenylalanine | 25 mg/kg/day |
| Tryptophan | 4 mg/kg/day |
| Histidine | 10 mg/kg/day |
| Methionine | 10 mg/kg/day |
The bottom line on very low protein diet for kidney health
High animal protein intake is associated with faster progression of CKD, likely due to excess production of nitrogen byproducts from protein breakdown. Following a whole food plant-dominant diet can be helpful to delay the progression of kidney disease. The use of ketoacid analogues as supplements may not be practical. In order to assure that the patient is getting adequate sources of EAA and other necessary nutrients, dietary restrictions should be implemented with the help of an experienced nutritionist, preferably one trained in a personalized approach to kidney health.
The post Very Low Protein Diet for Kidney Health appeared first on Integrative Kidney.]]>Kidney stone formation (urolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. They are painful, inconvenient, and when left untreated, they may contribute to more serious conditions including obstruction and kidney damage.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Read more about the etiology and prevalence of kidney stones here.
In this series we’re building a case for a more integrative approach to preventing kidney stone formation.
Conventionally, the treatment approach does address kidney stones via a multi-pronged approach that may include medication, dietary and lifestyle, surgical removal, and using ultrasonic waves to break up stone.
However, these guidelines tend to focus too far downstream, on stone composition instead of on the underlying pathology upstream. Instead, we advocate for a more comprehensive approach that focuses on risk factors to prevent formation. Those factors include:
· Type of stone
· Socioeconomic factors
· Diet
· Hydration and electrolyte balance
· Microbiome and gut health
· Genetics
We covered individual dietary components in detail in a previous blog. Today we’ll look at the gut-kidney stone connection and the impact of the microbiome.
Gut Integrity and Kidney Stones: Leaky Gut
A normal and healthy GI tract has a natural barrier. This barrier serves to protect the GI and has three major jobs: 1. ensure proper digestion and absorption of nutrients and 2. ensure elimination of toxins and 3. protect the integrity of the microbiome – the “good” bacteria that lives in our GI tract and works with our body to maintain health.
Leaky gut describes a state when the cells that make up the lining of the GI tract separate enough to allow the contents of the gut to leak out. This is also sometimes called intestinal permeability or IP for short. This is a problem because it reduces absorption of nutrients, causes toxins to build up, alters the balance of the gut microbiome, and results in systemic inflammation.
One of the major contributors to leaky gut is the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
· Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
· Increased intake of processed/refined foods like cereals, crackers, baked goods, etc…
· Processed, fried, conventionally raised, high-nitrate animal protein
· Low intake of fiber and fresh produce in general
· A “beige” diet (low in phytonutrients and antioxidants) from consuming a variety of colorful fruits and vegetables
· Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
We have already established that eating more fresh produce, is protective from kidney stone formation, and we’ve done a deeper dive on specific nutrition impact on kidney stone risk in another blog if you’d like to learn more.
There are several factors that may contribute to development of leaky gut:
· “Proinflammatory” SAD: too much processed and high-sugar foods, not enough fiber and the wrong inflammatory fats
· Food sensitivities: consuming food that are cause reactivity
· Overconsumption of caffeine and alcohol – irritants to gut lining
· Use of certain medications, including NSIADs, steroids, antibiotics
· Stress and poor-quality sleep
We address risk factors for intestinal permeability in more detail in a previous blog here, as well as dive into a comprehensive gut restoration strategy here in this 5-part series.
The Microbiome and Kidney Stones
Balance of the gut bacteria also play an important role in causing or preventing kidney stones. The most studied organism is Oxalobacter formigenes, which has been found to be protective when present in adequate quantities as part of the GI microflora. This bacterium degrades oxalate in the gut decreasing its absorption and excretion in the urine.
When Oxalobacter was discovered, scientists thought they had pinpointed the key to curing kidney stones. They concluded that simply supplementing this missing species should reduce risk of stone formation in susceptible individuals. It would turn out that the connection wasn’t that simple.
More recent evidence points to a more complex picture in the connection between microbiome diversity and kidney stone pathology. The emerging research shows increased risk in kidney stone formation in certain susceptible individuals also presented with alterations in normal microbiome and metabolome (metabolic byproducts from microflora) – also termed dysbiosis.
In other words, it’s likely that genetic factors might be “turned on” by dysbiosis leading to increased risk of kidney stone formation in certain individuals. The good news is that means they should be “turned off” when the microbiome balance is restored.
Studies that looked at the use of targeted probiotics have failed to show enough significant improvement of risk of urolithiasis. Although there’s been some limited and temporary reduction in oxalate excretion and kidney stone formation with the use of a combination of Lactobacillus, Bifidobacterium, Enterococcus, it’s been shown to be temporary and limited in benefit. This is because dysbiosis cannot be addressed by simply applying a band aid of a probiotic.
We recommend instead a more comprehensive approach to gut restoration and microbiome balance. You can read more about the 5R protocol in our comprehensive 5-part series on gut restoration.
The Bottom Line
Although initial findings about the impact of the microflora that looked at Oxalobacter in isolation have not demonstrated significance in reducing incidence of kidney stone formation, more recent evidence pointing to an interplay of factors on microbiome diversity is promising. Furthermore, factors that impact kidney stone formation include dietary factors, including food quality, nutrient composition, and dehydration. Along with environmental factors, lifestyle, genetics, and gut integrity and microbiome balance should be addressed through a comprehensive and personalized approach. Practitioners working with individuals to prevent kidney stone formation should formulate a patient care plan that modifies all relevant components in their integrative approach to maximize effectiveness in preventing urolithiasis.
The post The Microbiome and Kidney Stone Formation appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
Recent studies have focused on the significance of a relationship between gastrointestinal (GI) integrity and microbiome diversity with various chronic diseases including kidney disease.
In previous blogs, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance leading to intestinal permeability (IP or “leaky gut”). The impact of leaky gut on kidney health and progression of chronic kidney disease (CKD) has been referred to as the gut-kidney connection is the result of complex biochemical and immune mechanisms.

A comprehensive approach to CKD includes addressing the health of the GI at the root of it. Furthermore, this means not simply supplementing with probiotics, but instead addressing all the mechanisms that underlie probiotic need. This includes modification of microbiota balance, integrity of the mucosa and epithelium of the GI tract, improving GI motility, absorption and digestion, and modulation of the immune system.
Below we will explore the fifth step, Rebalance. But first, let’s quickly review the first four steps of the comprehensive gut restoration protocol. A reminder that the 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD.
The 5 steps of the 5R protocol for healing leaky gut are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Rebalance
The goal of the fifth step is maintenance and prevention of recurrence of IP or leaky gut. There are various factors that lead to IP that involve dietary and lifestyle influences*, including:
· Standard American Diet (SAD) which is low in fiber, high in processed foods, and highly inflammatory
· Poor eating habits (for example, multitasking and not chewing adequately)
· Inadequate hydration and/or electrolyte imbalance
· Motility issues leading to constipation or unfavorable formation/frequency of stool
· Stress and poor sleep
· Not enough exercise
To reduce risk of CKD, we must work towards improving diet and lifestyle habits that support continued GI health.
*Read more about medications that impact gut health and ultimately increase risk for KD progression here.
Where to start?
Address lifestyle factors that impact gut health
Long term dietary goals focus on a plant-based diet that is high in fiber and a wide range of key nutrients. Organic sources of animal protein can beneficial when eaten in moderation. However, the key is to load up on naturally antiinflammatory, low carbohydrate vegetables to maximize vitamins, minerals, phytonutrients and, of course, fiber. Reduce intake of starchy vegetables and eat more**:
· Dark leafy greens (like spinach, arugula, and romaine)
· Cruciferous veggies (like broccoli, cauliflower, and kale)
· Fresh whole fruit (preferably lower sugar berries and avocados, and limit to two servings per day)
· Some colorful starchy veggies can be OK (for example, carrots, sweet potatoes, beets, and squashes)
Those veggies listed above promote a healthy microbiome and improve alkalinity associated with improved kidney health. In addition to those sources of prebiotic fiber, include probiotic sources like fermented vegetables and drinks like raw sauerkraut, kimchi, pickles, apple cider vinegar, and kombucha. It’s recommended these are eaten raw because pasteurization process will destroy the bacterial content of these foods.
In general, reduce processed carbs, like bread, cereals, and high sugar foods, desserts and pastries. These types of foods are usually low in fiber and nutrients and help contribute to starving of beneficial gut bacteria. Furthermore,high carb diets have been associated with increased risk of cardiovascular (CV) disease and diabetes. Instead, moderately consume whole grains like brown rice, quinoa, oatmeal, and legumes**.
Include antiinflammatory fats focusing primarily on omega 3 sources from fish, nuts, seeds, and mono- and polyunsaturated (MUFA and PUFA) like avocados and olive oil. Even though the topic of saturated fats is more controversial, recent evidence suggest that moderate intake of certain saturated fats like that found in beef, organ meat, or ghee (clarified butter) derived from grass-fed cows and virgin coconut oil might have health benefits. Everyone agrees, however, that trans fats(aka hydrogenated oils and artificial products like margarine) or excessive intake of processed and fried fats contributes to inflammation and increased risk of disease.
Ensuring adequate hydration, drinking at least half your body weight (pounds) in ounces of water, not only helps to maintain good kidney health, it also helps support daily regular bowel movements.
It’s well established that regular exercise can be beneficial for many reasons, including improved blood pressure, blood sugar, hypertension, stress relief, and even improved digestion and GI motility. In fact, exercise has been associated with improved microbiome balance as well as beneficial modulation of the immune system.
Poor sleep quality and stress are also deeply tied to many underlying factors impacting of GI health. Reduced sleep duration and quality has been associated with increased inflammatory markers (including TNF, IL-1, and IL-6) associated with GI disease like GERD and Irritable Bowel Disease/Syndrome (IBD/IBS) disrupting digestion and nutrient absorption. Furthermore, sleep has been shown to affect kidney health directly and indirectly, including associated risk of CV disease, diabetes, obesity, and hypertension(read more about the relationship between sleep and kidney health here).
**NOTE: Due to individual variations and progress of disease, work with a nutritionist to assess if you need to maintain any specific restrictions due to your unique case and needs.
The role of supplements
Because of the unique needs of kidney disease patients, many need to rely on supplements to help obtain adequate amounts of key nutrients to maintain GI and kidney health.
This may include GI and motility support including but not limited to digestive enzymes, bitters, probiotics, and magnesium citrate and triphala for motility). Furthermore certain individuals may benefit from supplementation of certain vitamins and important minerals, high potency antioxidants, and/or support of certain key underlying cellular mechanisms impacting mitochondrial health, detoxification and nitric oxide production.
That said, many factors must be taken into consideration when choosing appropriate supplements for each patient. Supplement quality and contamination are a common concern, as are potential interactions with medication or contraindications in certain commonly associated chronic disease. We suggest working under the care and guidance of a practitioner or team of providers who are trained in integrative and functional medicine and understand the unique needs of kidney patients.
Next steps
Unfortunately, addressing gut health is only the beginning. As mentioned above, kidney disease is often associated with multiple chronic diseases including CV disease, diabetes, high blood pressure, and obesity.
The comprehensive approach to kidney care means addressing the underlying causes of this constellation of diseases which is best accomplished by a Functional Medicine approach. The goal is to identify and then rebalance the biochemical and pathophysiological dysfunction at the root of chronic disease, we can stop the progression of kidney damage and preserve kidney function.
Bottom Line
In the final step in the 5R individualized gut restoration protocol, we Rebalance the foundational factors that impact the gut-kidney axis.
Although this might be the last step in the 5R protocol, within the broader context of kidney disease, it might signal the transition to a comprehensive therapeutic protocol that includes management of the underlying dysfunction associated with related conditions such as CV disease, diabetes, obesity, and hypertension.
Working with an integrative or functional medicine provider is essential to help you navigate the comprehensive program successfully, but can help you stabilize blood sugar, lower blood pressure, lose weight, and reduce the risk of CV and ultimately, KD.
More from InKidney on the gut-kidney connection:
· Comprehensive Gut Restoration Protocol https://old.inkidney.com/2019/07/05/comprehensive-gut-restoration-protocol-ckd/
· Feeding Gut Bacteria in Kidney Disease https://old.inkidney.com/2019/02/20/feeding-gut-bacteria-patients-kidney-disease/
· Kidney-Gut Axis: Nutrition can slow the progress of kidney disease https://old.inkidney.com/2018/09/20/kidney-gut-axis-nutrition-slow-kidney-disease/
· Inflammation, Leaky Gut And Kidney Disease https://old.inkidney.com/2018/10/05/inflammation-leaky-gut-kidney-disease/
· Leaky Gut And Kidney Disease: 6 Classes Of Medication That Might Be Contributing https://old.inkidney.com/2018/10/05/leaky-gut-kidney-disease-medications/
The post The 5R Protocol Part 5: Rebalance appeared first on Integrative Kidney.]]>





