The root cause of IgA nephropathy
IgA nephropathy is a kidney disease that is defined by the pathologic appearance of glomerular deposition of IgA immune complexes. However, this definition does not address the root cause of the disease.
It has been increasingly recognized that IgA immune complex that deposit in the kidneys predominantly contain polymeric IgA1 lacking galactose within its O-glycosylated hinge region.
In this study, researchers found that patients with IgA nephropathy have elevated levels of certain B cells that are enriched for λ light chains. These cells are predestined for homing to upper respiratory and digestive tract mucosal tissues. In the mucosal tissues, these B cells mature and excrete abnormal IgA in the setting of upper respiratory or digestive infection. You can read more IgA nephropathy by reading our blog here.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Metformin: Some people love it and some people hate it… But it sure is getting a lot of attention lately
This study was done in rats with “non-diabetic kidney disease.” CKD was established in these rats by feeding them high adenine diet. Then they were randomized to receive either metformin or canagliflozin (an SGLT-2 inhibitor).
Metformin, but not canagliflozin, halted the decline in kidney function. Additionally, kidneys of metformin-treated animals showed less interstitial area and inflammation as compared to the vehicle group.
Metformin is increasingly being studied in humans for various kidney diseases. If used judiciously it may be a cheap alternative to preserving kidney function.
Download Your Copy!
Tremor + gum disease + nephrotic Syndrome = ?
In this study, investigators in Beijing looked into the manifestation of mercury poisoning in 172 patients. 26.74% of these patients had kidney injury (3/4 were women) and most of them had nephrotic syndrome. The most common finding on the biopsy was membranous nephropathy.
Other findings of chronic mercury poisoning were neurotoxicity and gingivitis. Chelation with DMPS alone was as effective as chelation and prednisone in reversing kidney injury.
The most common source of exposure without kidney disease was industrial exposure. Interestingly, the most common source of exposure leading to kidney disease was cosmetics containing ionic mercury (mercury concentration in one of the patients cosmetic was 4600 mg/kg – national standards are < 1 mg/kg).
Join here to receive FREE monthly updates on the latest research in Integrative Nephrology straight to your inbox.
We would love to here your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post April Research And News appeared first on Integrative Kidney.]]>

Altered vitamin K biodistribution may decrease the benefit of vitamin K2 supplementation in advance CKD
In this study, investigators sought to determine if there are other causes for vitamin K deficiency in advanced CKD beyond decreased dietary intake. They compared vitamin K uptake and distribution into circulating lipoproteins after a single administration of vitamin K1 plus K2 (MK-4 and MK-7) between patients on dialysis and healthy individuals.
They found that patients with uremia and advanced kidney disease don’t incorporate MK-7 well into HDL and LDL particles compared to healthy individuals. In addition, the combination of a statin and PPI was associated with signs of functional vitamin K2 deficiency in these patients.
In essence, patients with advanced kidney disease may not benefit as well from vitamin K2 supplementation. This highlights the importance of optimizing vitamin K2 status at earlier stages in CKD.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Higher levels of deoxycholic acid were associated with a higher risk of progression in CKD
Deoxycholic acid is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. Intestinal bacteria metabolize the primary bile acid, cholic acid, into deoxycholic acid (DCA).
Researchers studied 3,147 CRIC study participants who had fasting DCA levels. DCA levels above the median were independently associated with higher risks of ESKD and all-cause mortality.
This study highlights the importance of the microbiome and dysbiosis in the progression of kidney disease as we discussed in our blog.
Download Your Copy!
The higher number of medications a kidney patient takes the faster her kidney disease progresses
In a study performed in Japan of 1117 CKD patients under nephrological care, the use of a higher number of medications was associated with an increased risk of kidney failure, cardiovascular events, and all-cause mortality in patients with CKD. This is one of the major reasons we advocate for lifestyle modifications and coaching as the first and major step in the management of kidney disease.
Subscribe here to receive FREE monthly update on the latest research in Integrative Nephrology straight to your inbox.
We would love to hear your feedback. Let us know what you think of these educational materials and if you like us to focus on certain topics. Email us at info@inkidney.com
The post December Research and News appeared first on Integrative Kidney.]]>

The Role of Dietary Fiber Supplementation in Regulating Uremic Toxins in Patients With Chronic Kidney Disease: A Meta-Analysis of Randomized Controlled Trials
Gut-derived uremic toxins, including indoxyl sulfate (IS) and p-cresyl sulfate (PCS), are waste products that accumulate in the body due to decreased clearance by the failing kidneys. These accumulated uremic toxins are a risk factor associated with cardiovascular disease, progression of kidney dysfunction, mineral bone disorders, and increased mortality in patients with CKD. This meta-analysis showed that dietary fiber supplementation could decrease IS and PCS levels in patients with CKD.
https://www.jrnjournal.org/article/S1051-2276(20)30291-0/fulltext
Causal effects of physical activity or sedentary behaviors on kidney function: an integrated population-scale observational analysis and Mendelian randomization study
The health benefits of exercise and regular physical activity are vast. Using Mendelian randomization (MR) to assess causality, this study found that “the genetic predisposition to a higher degree of physical activity was associated with a lower risk of CKD and a higher eGFR, while the genetically predicted television watching duration was associated with a higher risk of CKD and a lower eGFR.”
https://academic.oup.com/ndt/advance-article/doi/10.1093/ndt/gfab153/6214515?login=true
Prevalence of Inflammatory Bowel Disease and Celiac Disease in Patients with IgA Nephropathy over Time
Poor gut health and a lack of intestinal wall integrity is linked to many diseases, including IgA nephropathy (IgAN). The results of this Finnish study showed an increase in the prevalence of inflammatory bowel disease (IBD) in patients with newly diagnosed IgAN over a 36-year period.
The post June Research and News appeared first on Integrative Kidney.]]>Kidney stone formation (urolithiasis) is a complex disease influenced by multiple factors including diet, genetics, and environment. They are painful, inconvenient, and when left untreated, they may contribute to more serious conditions including obstruction and kidney damage.
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Read more about the etiology and prevalence of kidney stones here.
In this series we’re building a case for a more integrative approach to preventing kidney stone formation.
Conventionally, the treatment approach does address kidney stones via a multi-pronged approach that may include medication, dietary and lifestyle, surgical removal, and using ultrasonic waves to break up stone.
However, these guidelines tend to focus too far downstream, on stone composition instead of on the underlying pathology upstream. Instead, we advocate for a more comprehensive approach that focuses on risk factors to prevent formation. Those factors include:
· Type of stone
· Socioeconomic factors
· Diet
· Hydration and electrolyte balance
· Microbiome and gut health
· Genetics
We covered individual dietary components in detail in a previous blog. Today we’ll look at the gut-kidney stone connection and the impact of the microbiome.
Gut Integrity and Kidney Stones: Leaky Gut
A normal and healthy GI tract has a natural barrier. This barrier serves to protect the GI and has three major jobs: 1. ensure proper digestion and absorption of nutrients and 2. ensure elimination of toxins and 3. protect the integrity of the microbiome – the “good” bacteria that lives in our GI tract and works with our body to maintain health.
Leaky gut describes a state when the cells that make up the lining of the GI tract separate enough to allow the contents of the gut to leak out. This is also sometimes called intestinal permeability or IP for short. This is a problem because it reduces absorption of nutrients, causes toxins to build up, alters the balance of the gut microbiome, and results in systemic inflammation.
One of the major contributors to leaky gut is the standard American diet (SAD), which seems to increase risk of kidney stone formation. When we use the term SAD, we are generally referring to a diet that includes:
· Consumption of sugary beverages and soda (and high carbohydrate consumption in general)
· Increased intake of processed/refined foods like cereals, crackers, baked goods, etc…
· Processed, fried, conventionally raised, high-nitrate animal protein
· Low intake of fiber and fresh produce in general
· A “beige” diet (low in phytonutrients and antioxidants) from consuming a variety of colorful fruits and vegetables
· Inadequate amounts of healthy, anti-inflammatory fats, and high amounts of refined unhealthy fats
We have already established that eating more fresh produce, is protective from kidney stone formation, and we’ve done a deeper dive on specific nutrition impact on kidney stone risk in another blog if you’d like to learn more.
There are several factors that may contribute to development of leaky gut:
· “Proinflammatory” SAD: too much processed and high-sugar foods, not enough fiber and the wrong inflammatory fats
· Food sensitivities: consuming food that are cause reactivity
· Overconsumption of caffeine and alcohol – irritants to gut lining
· Use of certain medications, including NSIADs, steroids, antibiotics
· Stress and poor-quality sleep
We address risk factors for intestinal permeability in more detail in a previous blog here, as well as dive into a comprehensive gut restoration strategy here in this 5-part series.
The Microbiome and Kidney Stones
Balance of the gut bacteria also play an important role in causing or preventing kidney stones. The most studied organism is Oxalobacter formigenes, which has been found to be protective when present in adequate quantities as part of the GI microflora. This bacterium degrades oxalate in the gut decreasing its absorption and excretion in the urine.
When Oxalobacter was discovered, scientists thought they had pinpointed the key to curing kidney stones. They concluded that simply supplementing this missing species should reduce risk of stone formation in susceptible individuals. It would turn out that the connection wasn’t that simple.
More recent evidence points to a more complex picture in the connection between microbiome diversity and kidney stone pathology. The emerging research shows increased risk in kidney stone formation in certain susceptible individuals also presented with alterations in normal microbiome and metabolome (metabolic byproducts from microflora) – also termed dysbiosis.
In other words, it’s likely that genetic factors might be “turned on” by dysbiosis leading to increased risk of kidney stone formation in certain individuals. The good news is that means they should be “turned off” when the microbiome balance is restored.
Studies that looked at the use of targeted probiotics have failed to show enough significant improvement of risk of urolithiasis. Although there’s been some limited and temporary reduction in oxalate excretion and kidney stone formation with the use of a combination of Lactobacillus, Bifidobacterium, Enterococcus, it’s been shown to be temporary and limited in benefit. This is because dysbiosis cannot be addressed by simply applying a band aid of a probiotic.
We recommend instead a more comprehensive approach to gut restoration and microbiome balance. You can read more about the 5R protocol in our comprehensive 5-part series on gut restoration.
The Bottom Line
Although initial findings about the impact of the microflora that looked at Oxalobacter in isolation have not demonstrated significance in reducing incidence of kidney stone formation, more recent evidence pointing to an interplay of factors on microbiome diversity is promising. Furthermore, factors that impact kidney stone formation include dietary factors, including food quality, nutrient composition, and dehydration. Along with environmental factors, lifestyle, genetics, and gut integrity and microbiome balance should be addressed through a comprehensive and personalized approach. Practitioners working with individuals to prevent kidney stone formation should formulate a patient care plan that modifies all relevant components in their integrative approach to maximize effectiveness in preventing urolithiasis.
The post The Microbiome and Kidney Stone Formation appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
The gut-kidney axis refers to the relationship between gut integrity and microbiome diversity with kidney disease. Excessive intestinal permeability, also known as hyperpermeability or more commonly as “leaky gut,” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
So far we looked at the first three steps of the 5R protocol, Remove , Replace, and Reinoculate. Applied sometimes sequentially and at times simultaneously, these steps are used to address the underlying factors associated with leaky gut. The idea is that comprehensive approach that reveres damage to the gut caused by exposure to food and environmental triggers, addresses the disruption of digestion and nutrient absorption, altered bowel motility, and dysbiosis, improves gut health and ultimately overall kidney health.
Read on below where we will explore step 4, Repair. But first, let’s first review the five steps of the comprehensive gut restoration protocol. A reminder that the 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD and in our upcoming blog on Rebalance, we will explore maintenance and next step in integrative kidney care.
The 5 steps of healing leaky gut are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Gut Repair
Up until this phase, we’ve focused on removing triggers that contribute to local and systemic inflammation. We’ve even taken steps to rebalance the microbiome. In the repair phase, we work to provide nutritional support that directly impacts the integrity of the gut mucosa and repairs hyperpermeability.
Recall from our previous blog, Part 1: Remove, that exposure to toxins, food sensitivity, and presence of pathogens leads to increased inflammation locally that triggers the immune system and leads to damage to the lining of the gut and mucosa.
The term Intestinal hyperpermeability (aka “leaky gut”) is the result of this inflammatory assault to the gut lining. These breaks in the integrity of the wall (imagine gaps in a fence on your lawn) let undigested food, bacteria, and metabolites “leak” through the holes.
The physiologic changes associated with this leaky state include a combination of factors that reduce your gut’s ability to absorb nutrients. These include hypochlorhydria (insufficient hydrochloric acid in the stomach to digest food), reduced production of digestive enzymes, altered bowel motility (often leading to constipation, but not always), and dysbiosis.
Now that we’ve addressed those aspects in steps 1-3, we can focus our energy on repair. This stage is often several weeks into the comprehensive gut restoration program because initiating it simultaneously may interfere with the efficacy of the prior steps. Working with an integrative or functional medicine practitioner can help guide you through your personalized program.
Where to start?
Address permeability with nutrients and herbs
To support intestinal mucosa regrowth and cell repair we focus on nutrients that have properties that promote rebuilding a healthy mucosal lining. They help improve the integrity of the intestinal wall by supporting building and formation of the intestinal epithelium, villi and cell connective tissue.
This includes a wide spectrum of micronutrients including vitamin A, D, E, and C. L-glutamine, butyric acid, and collagen supplements. These are useful to build collagen that forms the epithelium of the GI tract. Whey, colostrum or serum bovine immunoglobulins may be utilized to balance inflammatory mediators based in the gut. Zinc carnosine, melatonin, cabbage juice, aloe vera, and mucilaginous herbs like marshmallow root and slippery elm are also used therapeutically to support various aspects of the rebuilding process.
This is of course layered in on top of an anti-inflammatory, nutrient-dense, fiber-rich diet that includes healthy fats, moderate intake of animal protein, and a colorful variety of organic fruits and vegetables that provide added antioxidant and phytonutrient repair support.
Bottom Line
The fourth step in an individualized comprehensive gut restoration protocol involves leveraging food and herbs to promote the repair of the gut mucosa. This is often done after Remove, but might be simultaneous to Replace and Repair steps. However, every case is unique, and it’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this safely and successfully. Next, we will tackle the 5th “R” in the gut restoration protocol: Rebalance.
The post The 5R Protocol Part 4: Repair appeared first on Integrative Kidney.]]>By Lara Zakaria, PharmD, CNS, CDN, IFMCP
The Gut-Kidney Connection
In this series we examine the comprehensive gut restoration protocol used to as a foundational approach to heal gut integrity. Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
In our previous blogs, we discussed the impact of exposure to food and environmental triggers that impact the gut lining (or mucosa)integrity and microbiome balance leading to intestinal permeability (aka “leaky gut”).
So far we have looked at how the first and second steps of the 5R protocol, Remove and Replace, help to address the underlying factors associated with leaky gut that include a combination of factors like exposure to food and environmental triggers causing local and systemic inflammation, disruption of digestion and nutrient absorption, altered bowel motility, and dysbiosis.
Below, we will explore step 3, Reinoculate. Let’s first review the five steps of the comprehensive gut restoration protocol. The 5R Protocol addresses leaky gut as a foundational approach to reduce the risk of progression of CKD. The five areas of GI mucosal integrity are:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Reinoculate
The basic premise of the third stage of gut restoration is to foster an environment that allows beneficial microflora (aka good bacteria) to thrive in the gastrointestinal (GI) tract. This is achieved by leveraging diet, supplementation, and lifestyle modification.
In previous blogs, we have explored the connection between dysbiosis and kidney health. An increasing number of studies have demonstrated a significant relationship between the health of the microbiome and the progression of kidney disease, a relationship referred to as gut-kidney axis.
Those with a healthy microbiome, abundance of good bacteria and no overgrowth of bad bacteria, are less likely to develop chronic kidney disease. In fact, the presence of certain strains of bacteria in the gut can actually slow the progression of chronic kidney disease and even reduce the need for dialysis.
Where to start?
Prebiotics (aka Fiber)
Most Americans fall significantly short of the recommended fiber intake of 30+ g/day. High fiber intake is associated with reducing the risk of heart disease, obesity, diabetes, and even certain kinds of cancers like colorectal cancer. These benefits are linked to to improved bowel movements, neutralizing and removing toxins, and “feeding” gut bacteria contributing to a favorable microbiome balance.
Research even suggests that dietary fiber-intake may be one of the most significant predictors not only of gut health, but overall health and risk of disease!
Remember to always choose whole, fresh, fiber-rich fruits and veggies whenever possible to maximize nutrients and prebiotics simultaneously. Whole grains and legumes make good fiber sources as well, but always avoid processed foods that claim to have “added fiber”. They are usually packed full of fillers, sugars, grains, cereals and artificial ingredients.
High-sugar diets can be a major disrupter of your gut microbiome, primarily because it feeds bad bacteria and yeast overgrowth. This is one of the proposed mechanisms contributing to metabolic diseases like diabetes and heart disease, two conditions associated with KD.
Prebiotic supplementation might also be necessary in some cases. Supplemental powders and capsules of resistant starch, arabinogalactan, and mastic gum, among others, might be useful in many situations. Your integrative or functional medicine provider can help you determine which, if any, are appropriate for your unique situation.
Probiotics
Supplementation with high intensity probiotics may be very useful in the Reinoculation phase of the 5R protocol. There are a variety of strains of bacteria and even beneficial yeast that are used to help to “seed” the gut (though technically more recent studies suggest the benefit is transient, it can still be helpful). Depending on your individual needs, your integrative and functional medicine provider may choose a particular strain or opt for a broad-spectrum formulation. Either way, the quality and potency of the probiotic is important to consider (not all brands are created equal).
That said, research seems to support that the best long-term strategy is to increase the intake of probiotics coupled with prebiotic fiber intake through diet. Traditionally fermented foods are a great source of naturally found probiotics, these include non-pasteurized traditionally made kefir, sauerkraut, kimchi, miso, and pickled vegetables.
Other factors impacting the microbiome
Interestingly, exercise impacts microbiome balance and promote changes that improve gut health. Aim for at least 20 minutes of exercise daily at a level appropriate to your physical fitness and make it priority to get up and move – you and your kidneys are worth it!
Furthermore, stress has been found to negatively alter the balance of the microbiome by reducing the presence of friendly flora and promote the growth of bad bacteria. Although we know that stress cannot always be avoided, everyone can find a stress management practice to reduce its negative impact. Breathing exercises, meditation, long walks, listening to music, adult coloring books – the options are endless. Find what works best for you!
Bottom Line
The third step in an individualized comprehensive gut restoration protocol involves promoting microbiome balance. This is often done after the Remove step but might be simultaneous to the Replace step. Sequencing the steps of the protocol is case-by-case dependent. It’s important to work with an integrative or functional medicine provider trained in the comprehensive gut restoration protocol to help you navigate this successfully.
Next, we will tackle the fourth “R” in the gut restoration protocol: Repair.
The post The 5R Protocol Part 3: Reinoculate appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Researchers have established a relationship between gut integrity and microbiome diversity with various chronic diseases, including kidney disease. Increased intestinal permeability, also known colloquially as “leaky gut” has been shown to be at the root of this connection. This gut-kidney relationship is the result of complex biochemical and immune mechanisms.
Past studies have attempted to explore the impact of dietary changes shifting the gut microbiome to help restore the lining of the gut and reduce the resulting inflammation. However, many of the studies in the literature looking at the use of probiotics to reduce uremic load show some limited benefit in reducing the chronic kidney disease (CKD) burden.
But these studies failed to present a comprehensive approach that reveres damage to the gut while simultaneously inoculating the necessary bacteria. This single dimensional approach does not acknowledge all the different factors involved in the gut-kidney Axis.
To assure that the patient is getting a comprehensive gut restoration protocol, all the mechanisms that underlie probiotic use should be addressed. These include modification of microbiota, competitive adherence to the mucosa and epithelium, strengthening of gut barrier and modulation of the immune system.
The 5R gut restoration program addresses these gaps and help reduce the risk of progression of CKD.
This program is designed to address five areas of GI mucosal integrity repair:
1) Remove potential triggers, including polypharmacy, pathogenic organisms, food intolerances, sensitivities and allergies, or toxic exposure.
2) Replace digestive aid to support improved nutrient absorption and metabolism, including digestive enzymes, or agents that promote improved motility and regular bowel movements.
3) Reinoculate provide an environment where good bacteria can thrive and where bad ones cannot.
4) Repair support of the cellular repair process through the above, as well as by providing specific nutritional support for the regeneration of the GI protective barrier.
5) Rebalance lifestyle factors that influence the gut bacteria such as stress, sleep, exercise and relationships and assure ongoing gut health.
Remove
The first step of this protocol focuses on removing any exposures that may be contributing to increase inflammation in the gut. This includes food exposures, toxins, as well as screening for and treating pathogenic bacteria, fungi, parasites, or viruses that maybe disrupting the normal microbiota balance, which may require antimicrobial treatment to eradicate.
First, food known to cause sensitivities and allergies should be eliminated as part of an elimination diet. There are multiple categories of foods that contribute to inflammatory response that disrupts the lining of the gut. For example, gluten has been found to be associated with the development of leaky gut and IgA nephropathy. At the same time, we should be emphasizing the inclusion of key nutrient-dense foods that help to restore gut integrity and reduce inflammation, including antiinflammatory fats, organic fiber and phytonutrient-rich vegetables and fruit.
Typically, an elimination diet removes common food triggers like gluten, dairy, eggs, and soy. Depending on the root of the food reactivity, sometimes the removal or reduction of grains, legumes, FODMAPS, or night-shade vegetables is also necessary.
A nutritionist or clinician trained in implementing the elimination diet can help guide on which foods to eliminate or reduce. The decision may be done empirically, using symptom monitoring to guide the progress. Alternatively, the program can be personalized by using specialized testing for immune response against food can help guide this process.
Second, exposure to environmentally derived toxic substances should also be minimized, this includes mercury, arsenic as well as pesticides and other environmental pollutants whenever possible. We are exposed to toxic chemicals on a daily basis including pesticides in conventionally-farmed food, non-stick cookware, plastic use, and flame retardants and off-gassing released from furniture.
Addressing this aspect involves focusing on consuming organic produce and animal products and addressing environmental sources of toxicity. This topic is covered in more detail in this blog here.
Lastly, remove also entail identifying potential microbial triggers that might be contributing to inflammatory response. As mentioned above this may be “bad” bacteria, viruses, candida, and/or parasites. The presence of these offenders can be detected through symptoms as well as through advanced stool testing that employs a technique called PCR. Once identified, your practitioner can use antimicrobials to help eradicate the pathogenic organism, using prescriptive antibiotics or even herbal antimicrobials if appropriate.
Where to start?
Inflammatory foods
Some foods can be inflammatory or induce allergic reactivity. Furthermore, they may provide an environment that allows for the growth of pathogenic microbes, including low-fiber diets and high sugar diets. Furthermore, depending on your genetics, some foods can even lead to autoimmune conditions that may affect the kidneys. Eliminating food that is known to cause sensitivities varies dependent on the individual and their genetic predisposition.
An elimination diet can help with identifying what food an individual should avoid. This diet involves the removal of foods commonly associated with food sensitivities or immune reactivity. There are many variations of the elimination diet, and it’s very important to work with a nutritionist who can ensure you’re doing the protocol correctly and not missing any essential nutrients.
After a period of removal lasting at least 4 weeks, and assuming improvement in symptoms that suggests improved gut integrity, your integrative or functional medicine provider will work with you on gradual and careful reintroduction of foods to assess if tolerance has improved.
Decrease/Eliminate Exposure to Toxins
It is very hard to eliminate all sources of toxin exposure, but these steps can help minimize it:
1. Water filtration: we discussed options for water filtration in a previous blog.
2. Cookware: gradually eliminate all toxic non-stick cookware from your kitchen.
3. Avoid plastic containers or utensils: gradually switch from plastic to non-plastic containers and utensils.
4. Eat organic when possible: we know that choosing organic food can be expensive. If you are on a budget, there are certain fruits and vegetables that you should buy organic because of their high toxic burden. Visit the Environmental Working Group website to learn about their “Dirty dozen” T. This list gets updated yearly depending on their tests.
5. Be careful of processed drinks and juices. Not only do these contain excessive amounts of sugar, but they can also be a source of heavy metals and toxins because of poor regulations.
Eliminate Pathogens
As discussed above, laboratory analysis of the patient’s stool and serum can help give insight to the extent of intestinal permeability (leaky gut), as well as the status of microbiota. Establishment of baseline status is essential for measuring outcomes at designated milestones to give quantifiable outcomes. A comprehensive stool analysis can be a good start. In addition, a good dietary intake can help identify issues that could be related to food intake to guide this process.
Depending on the results of the individual’s comprehensive stool analysis, certain bacterial or yeast overgrowth can be detected. Also, parasites can sometimes be identified. Any of these should be treated by various medications, dietary changes, and/or herbal supplements as indicated and advised by an integrative or functional medicine provider.
Remove stress
Stress can have a bad influence on digestion and absorption. People who are stressed tend to eat too fast, make poor choices, and may eat too much at various intervals. This can lead to food choices that can lead to feeding the bad bacteria in the gut and poor digestion causing nutritional deficiencies. Furthermore, stress itself can influence epigenetic changes that impact dysbiosis as well as disease expression in general.
Bottom Line
The first step in an individualized comprehensive gut restoration protocol involves the removal of food sensitivities, environmental toxins, pathogenic organisms, stress. It’s important to work with an integrative or functional medicine provider trained to guide patients with elimination diets can help you navigate this successfully.
Next, we will tackle the second “R” in the gut restoration protocol: Replace.
The post The 5R Protocol Part 1: Remove appeared first on Integrative Kidney.]]>The Gut-Kidney Connection
Chronic kidney disease affects millions of American and is often associated with comorbid conditions such as cardiovascular disease, diabetes and hypertension. Dysbiosis and leaky gut are implicated in many systemic inflammatory and immune-related factors that lead to chronic disease. Dysbiosis is the term used to define alterations in the gut microbiota that includes overgrowth of bad bacteria, as well as underrepresentation of good bacteria. The normal integrity of the gut can be compromised resulting in systemic complaints, even in the absence of overt gut symptoms.
Research has established a relationship between gut microbes and different diseases, including kidney disease. This occurs through very complex biochemical and immune mechanisms. The presence of good bacteria has been associated with immune changes that inhibits inflammation. The use of probiotics and prebiotics that produce favorable changes in the microbiome have also been associated with gut changes that reduce advanced glycation end products (AGE), a uremic toxin associated with advancing kidney damage.
So, balancing the gut bacteria help slow kidney disease by directly inhibiting the immune response; and indirectly by reducing toxic burden known to progress CKD.
The Gut as a Potential Source of Inflammation
In the cases of dysbiosis, we see a rise in inflammatory markers that interact with the lining of the gut and result in damage and increased permeability or leaky gut. This causes shifts in the breakdown of nutrients, including amino acids. These shifts increase the levels of circulating substances involved in kidney disease and inflammation such as p-cresol, phenol, and indole. On top of that, these “pro-inflammatory” changes has also been associated with hypertension and increased risk of diabetes and cardiovascular disease; all of which are associated with CKD risk.
Yet, there have been inconsistencies in studies evaluating the benefits of targeting the gut-kidney axis with probiotics and prebiotics. Some suggest this may in part be due to the fact that research in this field is still in its infancy stage and conflicts in research may be due to statistical problems or inadequate interventions. But in an attempt to isolate the intervention, most studies utilized unilateral approach of inoculating the microbiome. In doing so, they usually fail to address other aspects of gut restoration and mucosal repair.
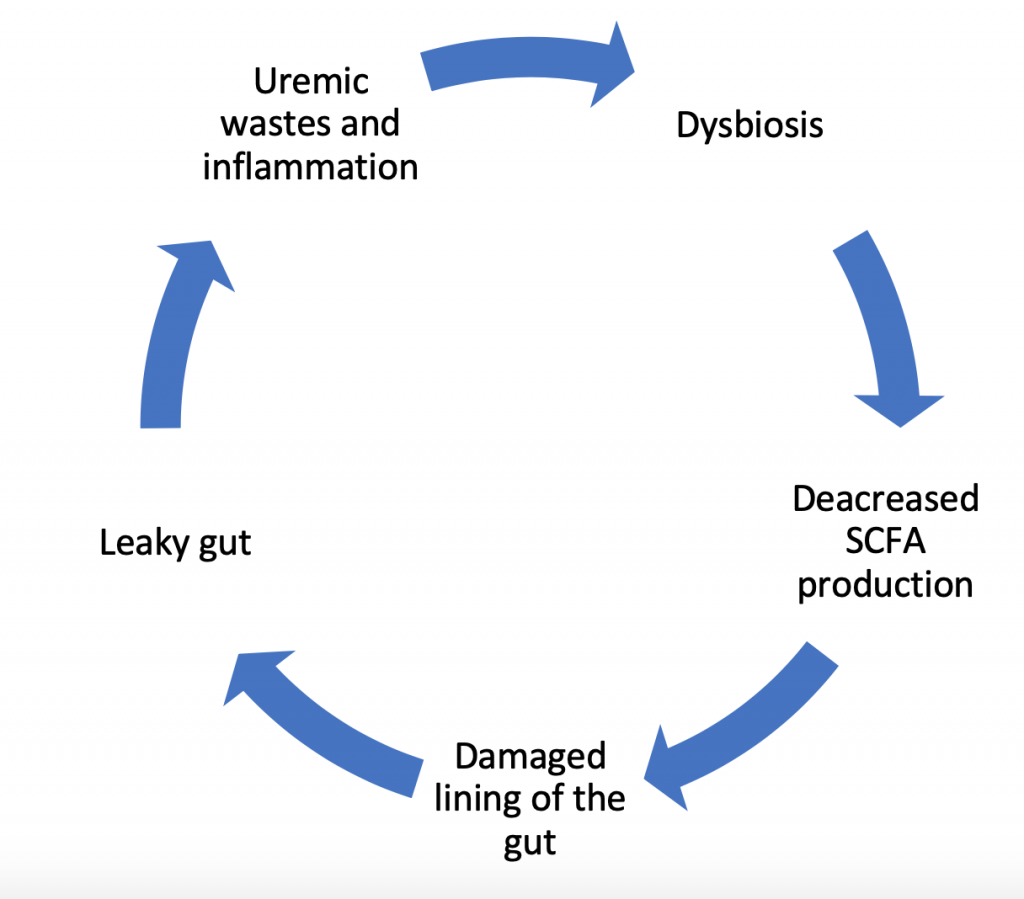
The Dysbiosis Cycle
In fact, changes in the gut bacteria do not operate as a one-way inflammatory force ending in damage to the lining of the gut. Instead it’s a dynamic process with a variety of contributing factors. Research confirms that it’s not only the dysbiosis that causes inflammatory damage, but also damage to the lining of the gut contributes to further dysbiosis and reduced production of short chain fatty acids (which feeds the lining of the gut). This, in-turn, increases the uremic waste products and further perpetuates growth of bad bacteria. This dysbiosis cycle damages the gut cell wall and promotes leakage of parts of the bad bacteria and toxins produced by these bacteria into the bloodstream. This leads to more inflammation, and the cycle repeats.
What is Missing in Current Research?
Individual Factors
As mentioned above, studies focusing on the use of probiotics to improve gut health and reduce inflammation and CKD risk have had mixed results. But these studies looked at a single change to see if it impacts kidney disease when in reality the process is more complex. Considerations for other factors that contribute to inflammation and CKD risk must also be accounted for. This includes environmental exposures, genetic risk factors, metabolic factors and changes in the body fluid volume that contribute to diabetes and hypertension, respectively, contributing to increased CKD risk.
Medications
On top of that, the effect of medication and polypharmacy on the gut and nutrient balance cannot be forgotten. Drugs commonly prescribed in kidney patients, like NSAIDs, Proton-pump inhibitors (PPI) or steroids, may also contribute to leaky gut. Changes in digestion due to some of these medications also contribute to altered nutrient absorption, including malabsorption of macronutrients, as well as micronutrients needed as cofactors in many biochemical reactions in the body.
Nutrients
The gut also can also serve as a source of increased oxidative stress, contributing to increased systemic inflammation that accelerates CKD and its comorbid conditions, including cardiovascular disease. Research suggests that one mechanism by which toxins generated by bacterial load speed up CKD progression is by altering metabolism and absorption of normally occurring nutrients. This may indicate that nutrient repletion maybe a necessary step for successful outcomes with probiotic re-inoculation.
This is why the use of an individualized comprehensive gut restoration protocol, that steps outside of the conventional linear model and takes into account the various layers described above can slow the progression of kidney disease. One such approach can be summarized by the 5R program which we will explain in details in the upcoming blogs.
The post Comprehensive Gut Restoration and Progression of CKD appeared first on Integrative Kidney.]]>You can think of your gut’s bacterial ecosystem (or microbiota) as a society. Just like any society, there are good guys and bad guys, and we can measure such attributes as population and diversity. So, the three questions we ask when evaluating the microbiome are:
- Are there any bad bacteria that need to be evicted?
- What’s the bacterial population? Do you have enough inhabitants living in the gut?
- Is there enough bacterial diversity? Different bacteria have different jobs and contributions, so a broader diversity is an indicator for better health.
There are multiple factors that affect the answers to those questions, and here we’ll focus on diet. Eating the right foods is possibly the most effective way to cultivate a robust microbiome and defend against “bad-guy overgrowth”.
Eating the right foods is possibly the most effective way to cultivate a robust microbiome and defend against “bad-guy overgrowth”
Share on X
Probiotics and Prebiotics: What’s the difference?
Prebiotics is a term used to describe “bacteria food” needed to cultivate a healthy microbiome. Bacteria primarily feed on fiber found in vegetables, fruit, and whole grains (not processed grains like bread and pasta). In fact, in people eating the Standard American Diet (SAD), which is high in animal protein and fat and low in dietary fiber, we see microbiome quantity and diversity significantly reduced. This imbalance is thought to be one of the major contributors to the chronic disease epidemic in the US.
Probiotics are live bacteria (and sometimes yeast) that are usually consumed through traditionally fermented foods, like kefir, kombucha, and sauerkraut and other fermented veggies, and are good for optimal gut function. They can also be taken as a supplement, which have grown in popularity as more research has suggested a benefit of supporting healthy gut bacteria.
As it turns out, focusing only on probiotics has yielded mixed results in the research. That’s because bacterial levels in the gut are transient unless supported with long-term dietary changes that incorporate regular intake of prebiotic fiber. Therefore, we need to look at both pre- and probiotic intake to optimize the microbiome.
It turns out, focusing only on probiotics has yielded mixed results in the research. That’s because bacterial levels in the gut are transient unless supported with long-term dietary changes that incorporate regular intake of prebiotic…
Share on X
Fiber, Fiber, and More Fiber
There are two kinds of fiber: soluble and insoluble.
Soluble fiber is the kind of food that suck up water and becomes like a gel. This helps to move digested food through the gut. Insoluble fiber does not break down during digestion. It passes through the gut intact, forming a sort of net, binding digested food byproducts together to create firm stool.
Fiber intake of at least 30 g/day is associated with reducing the risk of heart disease, obesity, diabetes, and even certain kinds of cancers like colorectal cancer. These benefits are likely due to improved bowel movements, removal of toxins, and support of a healthy microbiome.
To preserve kidney function, animal protein sources should be reduced or restricted (more about that here). The research suggests, that plant-based protein sources might be ideal for kidney disease patients since they also double as sources of diverse dietary fiber.
Whole grains, legumes, nuts, and seeds make excellent sources of both fiber and protein. In addition, many vegetables and fruits have surprisingly significant amount of protein including avocado*, broccoli, cauliflower, mushrooms (especially Portobello and shitake), and seaweed. Always keep in mind, though these foods may be generally healthy, in some patients with potassium restrictions you need to consult with your renal nutritionist to personalize your diet.
Remember to always choose whole, fresh foods whenever possible and avoid processed foods that claim to have “added fiber”. They are usually packed full of fillers, sugars, grains, cereals and artificial ingredients.
Remember to always choose whole, fresh foods whenever possible and avoid processed foods that claim to have “added fiber”. They are usually packed full of fillers, sugars, grains, cereals and artificial ingredients
Share on X
The recommended daily fiber intake is somewhere between 30-40 grams of both soluble and insoluble fiber. The following options allow you to mix and match to achieve your daily fiber. Every kidney patient is different and needs an individualized approach, we’ve highlighted the foods within this list that are high in potassium with this symbol (*) and food high in phosphorus with this symbol (‡) so you can further customize to fit your needs:
- 1 cup of cooked oatmeal = 4 g
- 1/2 cup of raspberries = 4 g
- 1 cup of avocado*= 10 g
- 2 cups of romaine lettuce = 2.4 g
- Medium-sized banana*= 3.1 g
- 1 cup of carrots = 3.6 g
- 1 cup of beets*= 3.8 g
- 1 artichoke*= 10.3 g
- 1 cup of Brussels sprouts = 6.4 g
- 1 cup of kidney beans‡= 11.3 g
- 1 cup of cooked split peas*= 16.3 g
- 1 cup of cooked chickpeas = 12.5 g
- 1 cup of cooked quinoa*= 5.2 g
- 1 ounce of dried chia seeds‡= 10.6 g
- 1 cup of blueberries = 3.5 g
- 1 cup of strawberries = 3.3 g
- 1 cup broccoli = 3 g
- 2 carrots = 6.4 g
- 1 cup of brown rice = 3.5 g
- Half a cup of lentils‡*= 15.6 g
- Medium-sized boiled sweet potato*= 3.8
- 1 oz of almonds = 3.3 g
- 1 apple = 5 g
- 1 pear = 6 g
- 1 ounce of dark chocolate‡= 3.1 g
- 3 cups of popped popcorn = 3 g
The recommended daily fiber intake is somewhere between 30-40 grams of both soluble and insoluble fiber
Share on X
Cut out the sugar, You Are Sweet Enough Already
High-sugar diets can be a major disrupter of your gut microbiome, primarily because it feeds bad bacteria and yeast overgrowth. This is one of the proposed mechanisms contributing to metabolic diseases like diabetes and heart disease, two conditions associated with KD.
Should you take probiotics supplement?
Though supplementation may be very useful in some cases, the best long-term strategy is to increase the intake of per- and probiotics naturally through your diet. Traditionally fermented foods are a great source of naturally found probiotics, these include non-pasteurized traditionally-made kefir, sauerkraut, kimchi, miso, and pickled vegetables. However, for various reasons they may be restricted in some patients, so work with a nutritionist to implement the best strategy for gut bacteria balance – which may include a combination of food-based intervention and supplementation.
Other lifestyle factors that affect the microbiome
Interestingly, exercise may change the composition of the microbes in the gut for the better. Aim for at least 20 minutes of exercise daily and make it priority (you and your kidneys are worth it). In addition, stress can deplete the friendly flora and promote the growth bad bacteria. Although stress cannot always be avoided, find way to manage stress like breathing exercises and meditation, and do your best to avoid triggers.
It’s important to work with a physician and nutritionist familiar with integrative approach to kidney disease. He/she can help you design a comprehensive plan that includes personalized dietary recommendations and supplements that promote healthy bacteria balance for optimal gut and kidney health.
The post Feeding Gut Bacteria in Patients with Kidney Disease appeared first on Integrative Kidney.]]>What is IgA?
Immunoglobulins are a large protein structure that is part of the adaptive immune system. There are various types, categorized by a letter and sometimes a number to identify the subclass, for example IgA1and IgA2. For the context of this discussion, we will the focus immunoglobulin A (IgA) without distinction to subclass.
IgA is found in the blood serum, lining of the respiratory (lungs) and digestive (gut) tracts. It is also found in saliva, tears, and breastmilk. Also called an antibody, the body makes IgA and other types of antibodies to help fight off invading pathogens (i.e. bacteria, viruses) and prevent disease. It is one of the first lines of defense against invading organisms from the environment, diet, and toxins.
What is IgA nephropathy?
IgA nephropathy is an autoimmune kidney disease. A trigger causes the immune system to produce abnormal IgA proteins that causes the immune defense to damage the kidney (nephrons). Abnormal IgA immune complex structures are formed, they circulate and filter through the kidney where they are “captured”. Unfortunately, these structures build-up in the kidneys, resulting in inflammation which damages kidney tissues.
What triggers an IgA immune response?
Though there’s some evidence that points to certain heavy metals (like cadmium) as a source of toxicity, the strongest evidence in the literature points to triggers can be classified into the following categories:
- Food sensitivities/allergies (i.e. gluten, soy, lectins/lipopolysaccharides)
- Microbial imbalance (known as dysbiosis) in the intestinal tract
Several studies have demonstrated an association between intake of gluten and lectins. One study demonstrated that patients with IgA nephropathy who followed a gluten-free diet for 6 months had a reduction in IgA levels. Studies on Mediterranean people eating a diet rich in gluten-containing foods published as far back as 1986 have suggested a dietary component to kidney disease. Soy is another example of a food antigen that has been associated with IgA nephropathy. However, heavy use of pesticides (glyphosate) in our food sources (especially gluten and soy) may play a role in food-related nephropathy.
Heavy use of pesticides (glyphosate) in our food sources (especially gluten and soy) may play a role in food-related nephropathy
Share on X
In susceptible individuals, the intake of a dietary trigger leads to intestinal permeability (IP), also known as leaky gut. In fact, research dating back decades has supported a connection between the gut and IgA nephropathy, called the gut-kidney connection. One of the proposed mechanisms, is the development of microbial imbalance (dysbiosis) which leads to bacteria and food proteins “leaking” through the gut barrier, triggering IgA immune response damaging the kidney.
The role of dysbiosis
In the case of IP, the compromised lining of the gut (mucosa) allows abnormal microbes, endotoxins (toxins produced by microbes), or food particles and metabolites to enter the bloodstream.
Furthermore, studies have shown that patients with progressive IgA nephropathy tend to have low number of naturally occurring healthy bacteria in the gut (likeBifidobacterium species) and a high amount of unwanted, or pathogenic, bacteria (likeSterptococceae). Our gut lining cells have complex mechanisms to detect the type of microbes that are present in our gut. There is a consistent link between these mechanisms and the production of an abnormal IgA.
The genetic link to an abnormal IgA response?
Genetics likely also play a role in the risk of developing kidney disease. Researchers have identified genetic variances (also referred to as single nucleotide polymorphisms, or SNPs) linked to IgA nephropathy in a multinational study.
Most of the genes involved in regulating the response and immunity against gut microbes. Interestingly, many of these SNPs associated with IgA nephropathy risk are related to inflammatory bowel diseases (IBD) or with the maintenance of the intestinal barrier. These SNPs lead to abnormal trafficking of IgA and the formation of an abnormal IgA that triggers the body’s immune response.
The autoimmune response, putting it all together
Recall, the presence of abnormal IgA in the blood leads to the formation of protein complexes in response to neutralize the threat. Unfortunately, these proteins end up forming structures that are difficult for our body to eliminate. These complex structures with abnormal IgA end up depositing in the kidney, leading to local inflammation and loss of kidney function.
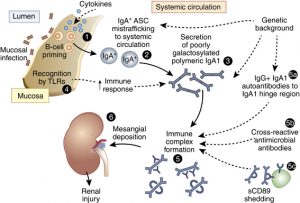
Now it should be clearer that IgA nephropathy is a classic example of how a combination of genetic, environmental factors, dietary and gut integrity interact to cause disease.
IgA nephropathy is a classic example of how a combination of genetic, environmental factors, dietary and gut integrity interact to cause disease
Share on X
Conventional medicine approach
The conventional approaches have failed to find a completely effective treatment for IgA nephropathy. Approaches include conservative control of blood pressure and blockage of the angiotensin-renin pathway, reduction of protein in the urine, and use of immunosuppressive therapy (including steroids and other medications), and fish oil.
More recently, a special kind of steroid that targets the terminal part of the gut (ileum and cecum) is being studied. The hope is that these special steroids will focus on one aspect of the process that leads to the formation of abnormal IgA which is decreasing the immune response in the gut.
Integrative medicine approach
A more integrative approach to IgA nephropathy would personalize treatment to take into account the impact of IP:
- Identifying the and eliminating the dietary or environmental triggers that contribute to leaky gut
- Assess and address dysbiosis
- Capitalize on epigenetic modifications to reduce the expression of SNPs contributing to IgA upregulation.
In combination with evidence-based conventional medicine, this approach targets various mechanisms and complications of the disease and improving outcomes.
The post IgA nephropathy: A target for Integrative approach appeared first on Integrative Kidney.]]>





