
Metformin and CKD survey results
You can see the results of the survey in this earlier blog. It demonstrated that caution about the use of metformin in CKD is common among primary care physicians and other specialties. Nephrologists seem to be more comfortable with using it until the patient reaches stage 4 CKD. Most nephrologists, however, were not aware that metformin is associated with nutritional deficiencies. You can read the results of the survey in detail here.
What is metformin?
Metformin was first produced in 1922. However, since 1958 it has been used in the control of blood sugar in type 2 diabetes. It is a biguanide drug, a class of medications that inhibit the liver’s production of glucose, enhancing the body’s insulin sensitivity and lowering the amount of blood sugar that the intestines absorb. It is chemically developed from galegina, a natural guanidine compound found in Galega officinalis (French lilac, also known as goat’s rue).
In essence, metformin is an oral diabetes medication that helps control blood sugar levels. It is often used together with diet and exercise to improve blood sugar control in patients with type 2 diabetes mellitus. There are other off-label uses for metformin and it is currently being studied for the treatment of polycystic kidney disease.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
Does metformin cause acute kidney injury?
For many years clinicians have been avoiding the use of metformin in kidney patients and specifically in those with diabetes. This is fueled by the FDA warning about the use of metformin in these patients due to concerns about lactic acidosis (discussed later). The original FDA warning was much more strict, but the more recent one issued in 2016 weighed in with more detail about metformin and kidney function. The new warning allows metformin to be used in patients who have worse kidney function. The newest warning indicates metformin can be used in patients who have an estimated glomerular filtration rate (eGFR) greater than 45 mL/min. Furthermore, metformin can continue to be used in existing patients whose eGFR remains over 30 mL/min.
As you saw from the survey, many primary care physicians and other specialists think that metformin can cause direct acute kidney injury. However, the literature shows that metformin can be protective in kidney disease. There is absolutely no evidence that metformin can lead to kidney injury. In fact, there is evidence to the contrary.
Download Your Copy!
Metformin clearance by the kidneys
Metformin does not undergo metabolism in the body, and it is eliminated unchanged in the urine. It is mainly cleared by tubular secretion in the kidneys. Metformin clearance is proportional to creatinine clearance. Therefore, although metformin can be used in patients with advanced kidney disease the dose of metformin should be decreased. The following table shows the recommended doses of metformin according to kidney function.
| Kidney function | Total daily metformin dose |
| Normal | 3000 mg |
| 45-59 ml/min | 2000 mg |
| 30-44 ml/min | 1000 mg |
| Less than 30 ml/min | Not recommended |
Metformin-induced diarrhea
The most common side effects of metformin are gastrointestinal. This is because one of the ways that metformin works is directly on the gut, altering glucose (sugar) absorption in the gut. Consequently, symptoms of nausea and diarrhea can occur especially in the first few weeks of taking it. That is why the patient should be started on a low dose to get used to it. Despite the statistic that 25% of patients taking metformin report diarrhea, only about 5% of those taking it had to stop it due to that side effect.
Metformin and lactic acidosis
The real reason why metformin was technically “banned” from use in CKD patients in previous years was due to the development of lactic acidosis. Lactic acidosis is a disease where lactic acid, a metabolic byproduct, accumulates in the blood. Lactic acid is mainly produced in small amounts by the muscles and red blood cells. It is formed when the body breaks down sugars to make energy under conditions of low oxygen (anaerobic glycolysis). This can occur during intense exercise or severe infection (called sepsis).
Metformin can induce lactic acidosis by shifting energy production in the cells toward anaerobic glycolysis. It also decreases the ability of the liver to consume lactic acid. Yet, in practice this is rare. In fact, it has been shown that lactic acidosis can occur in patients taking other oral blood-sugar-lowering medications at the same rate as metformin. Most case reports of metformin-induced lactic acidosis were in patients who have other conditions that lead to acute kidney injury (AKI) and lactic acidosis such as septic shock.
So, it is not that:

Instead, what happens is:
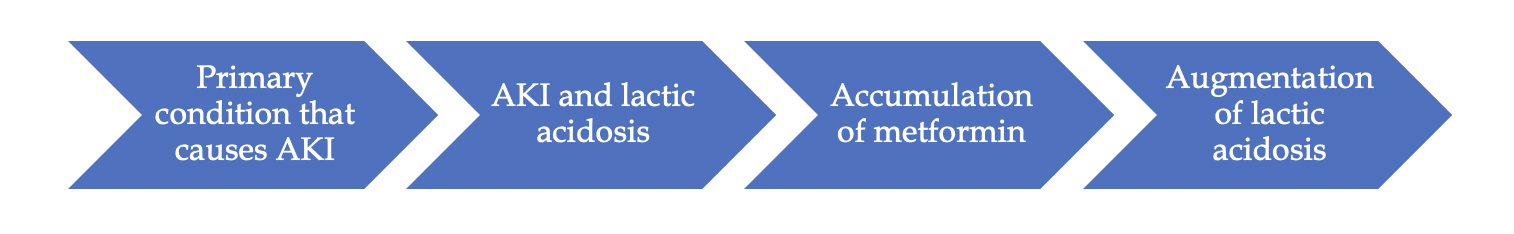
Finally, in a large retrospective study of patients with diabetes mellitus, metformin use was not significantly associated with lactic acidosis at kidney function levels higher than 30 ml/min/1.73 m2 (stage 3 or better).
Metformin and vitamin B12 deficiency
The association between long-term use of metformin and low vitamin B12 levels in type 2 diabetes mellitus patients is well-established. This is thought to be due to changes in the production of intrinsic factor in the stomach, which decreases vitamin B12 absorption in the intestine. Vitamin B12 deficiency is more pronounced if the patient is also taking a proton pump inhibitor such as omeprazole or histamine H2 receptor antagonist such as famotidine, which are medications used to treat acid reflux. Since diabetes can cause peripheral neuropathy, as well as vitamin B12 deficiency, can cause peripheral neuropathy, it can be confusing for diabetic patients on metformin and their practitioners.
Therefore, patients using metformin for a prolonged period should be screened initially and periodically for vitamin B12 deficiency. In addition, there is also growing evidence that other B vitamins, vitamin D, and magnesium can be impacted by metformin.
Bonus: Metformin’s impact on the gut microbiome
Because of metformin’s actions on the gut, it can also alter the intestinal microbiota for the better. Studies showed that metformin can enhance the growth of Akkermansia muciniphila, Butyrivibrio, Bifidobacterium, and Lactobacillus. In addition, it decreases the levels of some other bacteria like Intestinibacter. Metformin also promotes the production of the short-chain fatty acids that protect the intestinal barrier. In fact, some authors attribute some of metformin’s actions to its favorable impact on the gut microbiota and integrity.
The Bottom Line for Metformin and Kidney Disease
The answer to the question “Is metformin bad for the kidneys?” is generally no. Metformin can be used in patients with kidney disease. It actually leads to better outcomes in these patients. However, the risk of metformin-induced lactic acidosis increases in CKD stage 4 and above. It is, therefore, not recommended for use in these patients and patients with acute kidney injury. I also recommend avoiding it in patients with renovascular disease because their kidney function tends to fluctuate. Furthermore, metformin can be associated with vitamin B12 deficiency and patients should be screened initially and periodically for deficiency of this important vitamin.
The post To Metformin or Not To Metformin in CKD appeared first on Integrative Kidney.]]>While this is not official medical research, I aimed to identify current thoughts among nephrologists and primary care providers about its use in CKD patients. The survey results suggest that many practitioners avoid metformin in CKD based on outdated FDA guidelines. In fact, metformin has beneficial effects for kidney health and can safely be used in certain stages of CKD. In this blog, I will share the survey results. For the current evidence and recommendations for metformin use in kidney disease, read the second part of this blog, “To Metformin or Not to Metformin in CKD.”

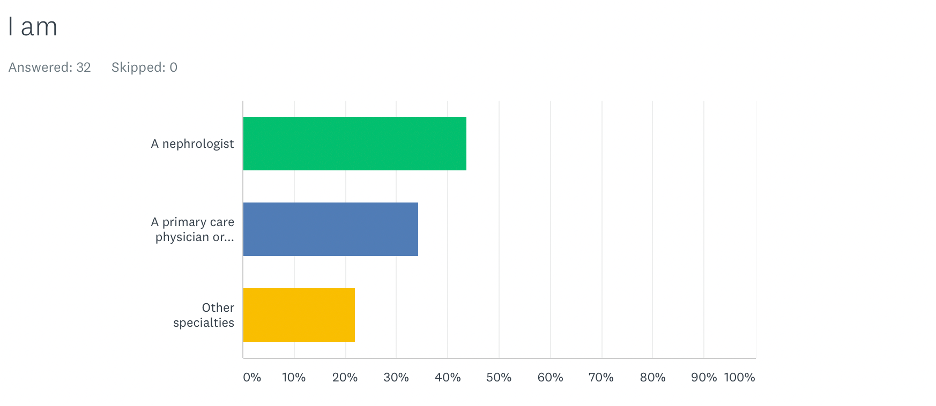
Participants in the Metformin and Kidney Disease Survey
We had 32 respondents: 44% of the respondents were nephrologists, 34% were primary care physicians, and 22% were from other specialties.
Join us in the fight against kidney disease and receive the FREE Report “5 Pitfalls to Avoid When Caring for Kidney Patients”
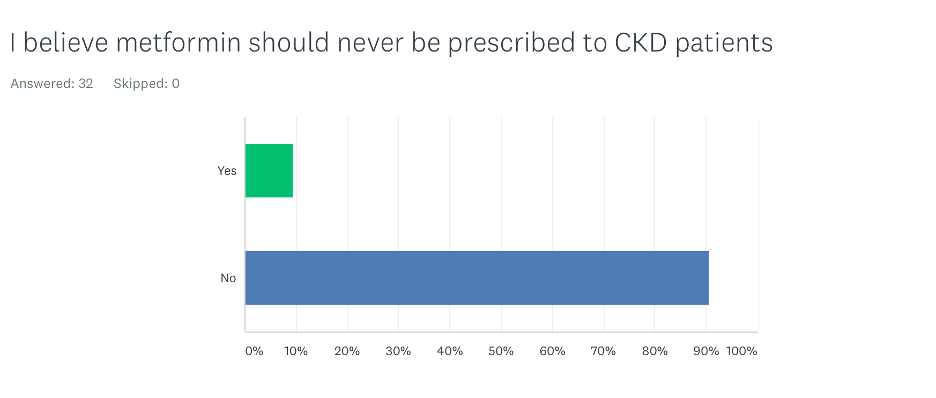
Should metformin be used in kidney disease patients?
Only 9% of participants thought that metformin should never be used in all CKD patients. All nephrologists believed that metformin can be used in patients with CKD. Among primary care providers, 27% believed that metformin should never be used in all CKD patients.
Does metformin cause acute kidney injury?
In the survey, 28% of participants believed that metformin causes acute kidney injury. The majority of those were primary care physicians. Only 7% of nephrologists believed that metformin causes acute kidney injury.
Block "fundamentals" not found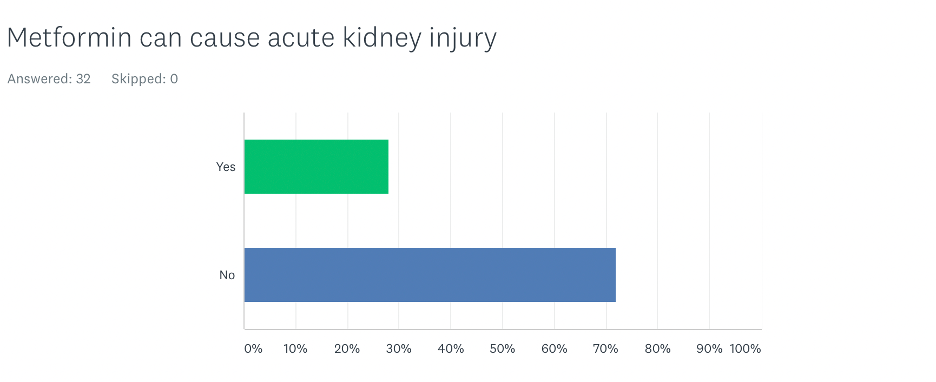
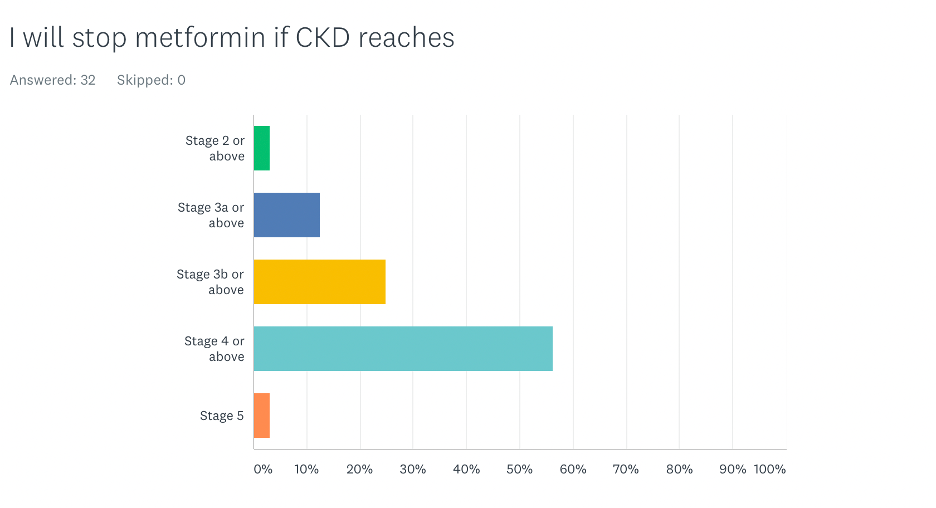
At what stage of CKD do you stop metformin?

The majority of participants (56%) believed that metformin should be discontinued when a patient reaches stage 4 CKD or above. Interestingly, 25% of the participants recommended discontinuing it if the patient reaches stage 3b CKD. Most nephrologists discontinued it when patients reached stage 4 CKD or above. Most physicians who discontinued it at earlier stages were primary care physicians.
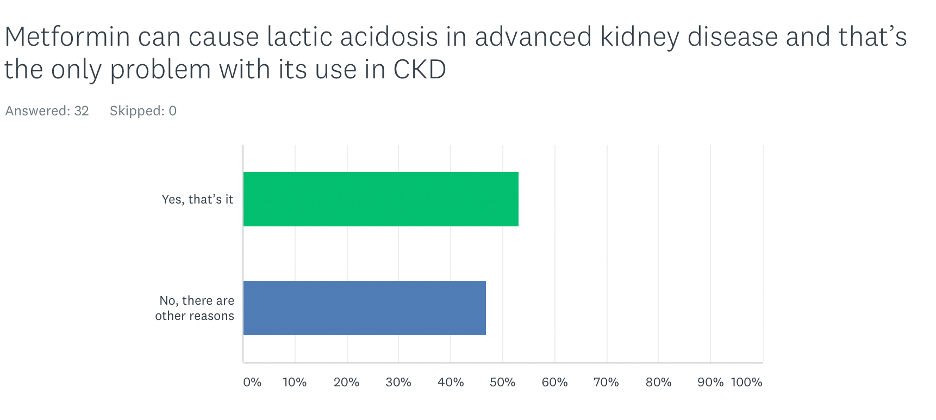
What is the problem with the use of metformin in advanced kidney disease?
This question may be suggestive, but about half of the participants believed that the risk of lactic acidosis is the only problem with the use of metformin in patients with advanced CKD. Interestingly, the majority of participants who believed there are other problems with the use of metformin in advanced kidney disease were non-nephrologists. One would think that nephrologists would be more aware of other risks beyond lactic acidosis.
When asked about specific reasons to avoid using metformin in advanced CKD, two rationales mainly emerged. Because metformin is removed from the body by way of the kidneys, one reason for its avoidance is the potential accumulation of excessively high levels of metformin in advanced kidney disease. The other reason was that metformin can be associated with diarrhea that can lead to volume depletion (loss of body fluids) and acute kidney injury.
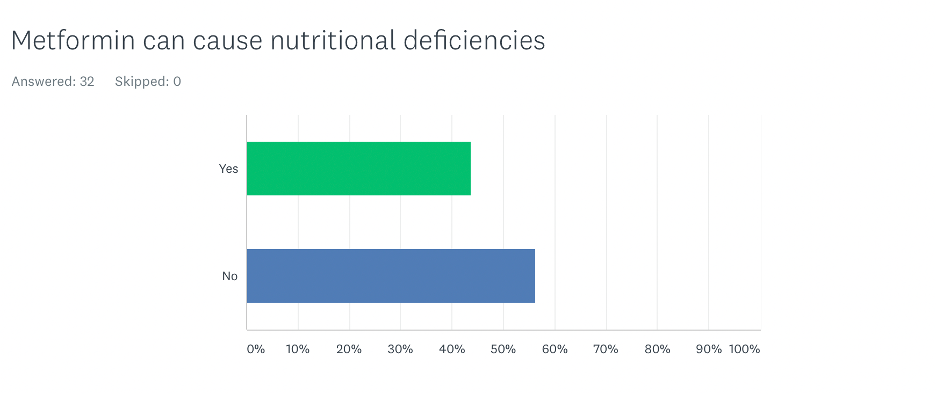
Can metformin cause vitamin B12 and other nutritional deficiencies?
Strikingly, 56% of participants were not aware of metformin and vitamin B12 deficiency or that it may cause depletion of other nutrients. Among nephrologists, this percentage was an amazing 79%.
The Bottom Line
Caution about the use of metformin in CKD is common among primary care physicians and other specialties. Nephrologists seem to be more comfortable with using it until the patient reaches stage 4 CKD. Most nephrologists, however, were not aware that metformin is associated with nutritional deficiencies. In the next blog, I will dig deeper into the research to find evidence-based answers to the questions presented in this survey.






