
Causes of magnesium deficiency
Poor dietary choices
There has been a steady decline in magnesium content in cultivated fruits and vegetables over the past 100 years. This is caused by the depletion of magnesium in soil over time. In addition, utilizing phosphate-based fertilizers leads to the formation of magnesium phosphate salts that are not soluble. This means the soil is deprived of both components: magnesium and phosphorus.
On top of that, the rise of ultra-processed food and drinks have also contributed to the depletion of magnesium in the modern standard American diet. Grain bleaching and vegetable cooking and adding preservatives can lead to a significant loss of magnesium content. Preservatives such as various forms of phosphate and oxalate can bind with magnesium and prevent its absorption. Phosphoric acid in soft drinks has similar effects.
The addition of fluoride to drinking water also prevents magnesium absorption by binding to it and forming insoluble complexes. Finally, drinking caffeine and alcohol can also lead to an increase in the excretion of magnesium by the kidneys, causing magnesium deficiency.
Drug-induced magnesium deficiency
Many medications can interfere with magnesium absorption or increase its excretion, leading to deficiency. Most of the medications leading to magnesium deficiency are summarized in the following table:
| Medication class | Example | Mechanism |
| Anti-diabetic medications | Insulin, insulin mimetics | Interferes with Na/Mg exchange leading to renal loss |
| Antimicrobial | Gentamicin, pentamidine, foscarnet, amphotericin B | Increased renal loss |
| Beta agonists | Salbutamol | Renal loss and cellular shifts |
| Bisphosphonate | Pamidronate | Renal loss |
| Cardiac glycoside | Digoxin | Increased renal loss |
| Chemotherapy agents | Cisplatin | Renal loss |
| Diuretics | Thiazide diuretics | Renal loss |
| Proton-pump inhibitors | Omeprazole | Decreased GI absorption |
Measuring magnesium status
Simply put, there is no ideal test for assessing magnesium status in the body. Mg blood levels are tightly controlled and represent only 0.8% of total body stores (0.5% in red blood cells and 0.3% in the serum). Red blood cell Mg levels have been used as an alternative method, but this too does not represent total body stores and is not well validated. Measuring urine Mg requires measuring a 24-hour urine specimen. This too has been found to be imperfect due to large variations from day to day.
The Mg retention test has been proposed as a more accurate way to assess Mg status. Here, the patient receives an intravenous Mg load (0.25 mmol magnesium/kg body weight at a rate of 2.5 mmol/hour), and a 24-hour urine specimen is collected before and after the load. The percentage of administered magnesium that is retained by the body (not excreted in urine) determines magnesium status. This test is not standardized yet, but retention of 25%-50% may indicate a moderate deficiency, and retention of more than that may indicate severe deficiency.
Ideally, measuring muscle or bone magnesium may be more reflective of accurate magnesium stores but this is obviously not practical. Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status.
Combining a serum Mg test, a 24-hour urinary Mg, and assessing dietary Mg intake is the most comprehensive and practical evaluation of a patient’s magnesium status
Share on X
Patients at high risk for magnesium deficiency include:
- Diabetics
- Heart disease patients
- Osteoporosis patients
- People who eat a diet high in processed food and soda
- People who suffer from leg cramps
- People with metabolic syndrome
- People who take certain medications
Those patients at risk of magnesium deficiency should be targeted for additional testing and supplementation.
Download Your Copy!
What type of magnesium should I take?
The type of magnesium supplement used depends on the exact indication. Magnesium supplements are available as oxide, hydroxide, gluconate, chloride, citrate, lactate, malate, taurate, L-threonate, sulfate, glycinate, orotate, and carbonate salts. In addition to magnesium citrate’s direct effects on kidney stones, magnesium benefits the person with kidney disease through its effects on blood pressure, insulin sensitivity, vascular health, and bone. The following indications are listed with the recommended types of magnesium supplements and doses. These doses are for prevention only. Patients who are deficient may need higher doses. Magnesium supplements should be discontinued or decreased in kidney patients if the serum magnesium level is higher than 2.6.
| Indication | Mg type | Dose |
| Prevention of kidney stones | Magnesium citrate | 400 mg daily |
| Bone health | Magnesium citrate or chloride | 400 mg daily |
| Improving blood pressure | Magnesium taurate | 400 mg once or twice daily |
| Improving insulin sensitivity | Magnesium taurate | 400 mg once or twice daily |
| Improving vascular health | Magnesium glycinate or orotate | 200-400 mg daily |
| Phosphate binder | Magnesium carbonate | 250 mg with meals |
We recommend using high-quality supplements. This article can be a useful guide.
The bottom line
Magnesium is essential to many biological functions, as I described in part one, “Magnesium and Kidneys.” It has many health benefits for kidney, bone, and vascular health. Assessing magnesium status is difficult but magnesium deficiency is very common and underrecognized. Supplementing magnesium may be important for patients with kidney disease. The type of supplement used depends on the indication. As always, it is recommended that you check with a Functional or Integrative Medicine provider and nephrologist before taking any new supplement.
The post Magnesium Deficiency: Assessment and Management for Better Kidney Health appeared first on Integrative Kidney.]]>
By Lara Zakaria, PharmD, CNS, CDN, IFMCP
Zinc: The Micromineral
Zinc is classified as a trace mineral because it’s typically needed in small doses to exert its biological effects. This nutritional mineral is involved in functions that include glycolysis (converting glucose into energy), digestion especially protein breakdown, bone formation, DNA metabolism, alcohol metabolism, angiotensin conversion, heme biosynthesis, and as an antioxidant protecting from reactive oxygen species (ROS) and the resulting cellular damage from inflammation. In fact, it’s so important, that it is the third most relevant intracellular ion (behind magnesium and potassium).
In addition, there are over 200 metal-containing enzymes, called metalloenzymes, responsible for catalyzing a variety of crucial reactions throughout the cells in our bodies. In fact, 20 distinct biological functions are associated with zinc metalloenzymes.
Zinc Absorption and Deficiency
Zinc is primarily absorbed in the small intestines via mechanisms called transcellular and paracellular transport. That means it can passively diffuse through tight junctions or by carrier mediated transporters. The latter means that absorption of zinc can be influenced by genetic SNPs which can result in decreased zinc transport and subsequently increased risk for deficiency.
Furthermore, absorption is reduced with aging and maldigestion/low stomach acidity. Drugs that inhibit stomach acid production, such as proton pump inhibitors (PPIs) and H2 antagonists (H2A) are thought to cause zinc depletion by reducing stomach acidity. This effect can be reversed by addressing the gut dysfunction through the 5R protocol.
Other drugs that may alter zinc status include angiotensin-converting enzyme inhibitors (ACEi), aspirin, thiazide and loop diuretics, long-term use of steroids, and fluoroquinolone, penicillamine, and tetracycline antibiotics. The degree of depletion is dependent on multiple factors, including individual differences, dose and length of therapy.
Assessment of zinc status through laboratory testing has been criticized as being unreliable. However, coupled with a comprehensive assessment of symptoms it can be very helpful in identifying mineral deficiency. Examples of symptoms of zinc deficiency include loss of/diminished taste and/or reduced sense of smell, frequent colds, slow wound healing, loss of appetite, presence of or worsening of acne/rosacea.
Zinc and kidney disease
The prevalence of zinc deficiency in chronic kidney disease (CKD) patients is well documented. Furthermore, zinc nutritional status is also altered in obesity. In fact, fasting plasma zinc concentrations are inversely correlated to BMI and plasma glucose levels.
Several inflammatory mechanisms are associated with zinc depletion, while adequate levels of zinc decreases formation of reactive oxygen species (ROS). ROS activate nuclear factor-κB (NF-κB) which induces the generation of inflammatory cytokines and adhesion molecules associated with various types of kidney diseases, including IgA nephropathy.
Superoxide dismutase (SOD), and enzyme which converts the oxidative superoxide into an inert product, hydrogen peroxide. The SOD enzyme requires zinc in order to work efficiently, and zinc deficiency reduces the activity of the enzyme and has been shown to increase inflammation-induced kidney damage, in part via this mechanism.
Another interesting associations include the impact of zinc deficiency on increased calcification in the development of kidney stones.
Zinc also plays an important role in protecting the kidneys from environmental toxins, especially heavy metal. Heavy metal toxicity, such as lead and cadmium, is significantly associated with kidney disease. Studies have shown that a deficiency in zinc, as well as other essential metals such as calcium or iron, can lead to increased absorption and toxicity of lead and cadmium. Therefore, providing yet another potential benefit of ensuring zinc repletion to protect against metal toxicity and its impact on kidney, as well as overall health.
Implications of zinc deficiency in cardiovascular and metabolic disease associated with KD
Zinc impacts kidney disease via indirect associations as well. Since cardiovascular disease (CVD) and diabetes are comorbid conditions associated with kidney disease, it makes sense that factors that impact them will also impact kidney disease.
It’s worth noting that various inflammatory markers that are triggered by deficiency of zinc, such as transcription of NF-κB, rise in IL-6, IL-2, and TNF-α, are associated with inflammatory/immune response that leads to systemic inflammation including atherosclerosis (vascular disease), a known complication of CKD.
Zinc has some interesting effects on blood lipids, including low density lipoproteins (LDL) and triglycerides (TG). Dyslipidemia is associated with increased risk of CVD. Zinc deficiency has also been associated with elevations in TG. Furthermore, maintaining adequate levels of zinc has a protective effect against atherosclerosis by inhibiting the oxidation of LDL by cells or transition metals.
In addition, there are multiple mechanism where zinc impacts metabolic disease. Interestingly, zinc is essential for insulin synthesis and release, and deficiency impairs the release of insulin contributing to insulin resistance and elevations in blood sugar.
Another hormone responsible for feeling of satiety (fullness after eating) is leptin, which is produced by fat cells. Leptin resistance is associated with overeating and obesity. Zinc depletion may impact leptin indirectly because leptin secretion is regulated by insulin. Furthermore, deficiencies in zinc impact the activity of peroxisome proliferator-activated receptors (PPARs). PPARs are associated with multiple metabolic pathways of lipid and glucose metabolism. They initiate cascade of mechanisms which activate atherogenesis, and are, therefore, central to metabolic disease, CVD, and kidney disease.
Food-based sources of zinc
Red meat, organ meats, and shellfish are considered the best sources of zinc, with levels significantly higher than vegetarian sources. Although not as high as animal sources, nuts and seeds are good sources of zinc. Although whole grains are good sources as well, refined grains are not because zinc is lost when the bran and germ coating is removed in processing. Unfortunately, although an essential part of a healthy diet, fruits and vegetables are not significant sources of this nutrient. Not surprisingly, vegetarians are at increased risk for zinc deficiency and should be screened routinely.
There are food interactions that reduce absorption of zinc. Phytic acid, naturally found in grains, may inhibit the absorption of zinc. Therefore it’s advisable to soak grains for 8-24 hours in filtered water and 1 tablespoon of lemon juice or apple cider vinegar prior to cooking to reduce levels of phytic acid and enhance absorption of zinc and other minerals. It’s also interesting to note that absorption of zinc is reduced by the intake of dairy, likely due to the high calcium content which is known to interfere with zinc uptake.
Supplementing zinc is a good option in cases of deficiency. There are multiple compounds available on the market. We recommend using zinc picolinate or zinc citrate. Zinc carnosine is useful as part of gut restoration protocol to address GERD and as part of a PPI tapering protocol. Note that the dosage and length of therapy should be customized to individual needs and tracked by your functional medicine provider or nutritionist. If you notice a metallic taste in your mouth, have your healthcare provider check your zinc levels to ensure they are not too high or toxic.
The Bottom Line
There are multiple suggested mechanisms contributing to the impact of zinc deficiency on kidney health. It’s no surprise considering the various mechanisms that involve zinc. It’s important to consider the impact of diet, medications, dysbiosis and intestinal permeability on zinc status and subsequently on protecting the kidneys from disease.
The post Zinc and Kidney Disease, Exploring the Links appeared first on Integrative Kidney.]]>
Vitamin D as a hormone
Vitamin D is a term used to describe a family of compounds derived from cholesterol. There are two major forms to keep in mind. Vitamin D2, or ergocalciferol, mostly found in plants, and D3, or cholecalciferol, the form found in animal sources and produced naturally in our skin when we’re exposed to the sun. It is a “conditionally essential vitamin” because we need cholesterol precursors to produce it but we can also consume it with food.
Vitamin D is not only a vitamin, but it’s also considered a hormone because of its direct interaction with cell receptors in the body. We rely on our a “factory” in the skin to make Vitamin D when we are exposed to the sun. The UV rays stimulate the conversion of precursors derived from cholesterol into active vitamin D3. Like other hormones derived from cholesterol, such as estrogen and testosterone, not eating or producing enough cholesterol can contribute to a deficiency of vitamin D.
Foods that contain vitamin D
We typically think of vitamin D as “the sunshine vitamin.” However, various factors impact the ability of the body to produce adequate active vitamin D3, including average daily sun exposure, geographic location, skin color, and genetic variations that impact “the vitamin D factory.” Luckily, there are other options for meeting our needs, including consuming vitamin D-rich foods as well as taking a high-quality supplement.
Foods that naturally contain the active form of vitamin D3 include wild salmon, herring, sardines, cod liver oil, tuna, oysters, shrimp, and egg yolks. Surprisingly, mushrooms are the only plant-based source of natural vitamin D2. However, you can actually significantly increase the naturally occurring amount of D3 by laying mushrooms in the sun (learn more here). Since Vitamin D is a fat-soluble vitamin, absorption is best when eaten with a healthy fat or food naturally containing fat.
Many people associate milk with food sources of Vitamin D. However, milk is not naturally a good source of vitamin D. Beverage companies fortify milk and other drinks (like orange juice, and nut milk products) and market them as “a great sources of vitamin D” – in other words vitamin D is artificially added in during the manufacturing process.
Milk is not naturally a good source of vitamin D. Beverage companies fortify milk and other drinks (like orange juice, and nut milk products) and market them as “a great sources of vitamin D”
Share on X
Vitamin D receptors
Vitamin D is most commonly associated with bone health, because it’s necessary in bone formation process along with vitamins K, calcium, magnesium, and phosphorous. However, it’s vital beyond bone health. Receptors for vitamin D have been identified in almost all organs in the body. These genes are responsible for the calcium and phosphate balance, immune response, and cell growth and differentiation. The presence of vitamin D receptors in the blood vessels also indicates that vitamin D plays an important role in maintaining heart health.
The presence of vitamin D receptors in the blood vessels also indicates that vitamin D plays an important role in maintaining heart health
Share on X
Activation of Vitamin D by the kidneys (and the liver)
Vitamin D that is produced by our skin or consumed is transported to the liver as a “prohormone” by a protein called Vitamin D Binding Protein (DBP). In the liver, this precursory form is converted by an enzyme called 25-hydroxylase (or CYP2R1). The end-product is called 25-hydroxyvitamin D, abbreviated 25-(OH)D, and is the main circulating form of vitamin D.
Once produced, 25-(OH)D is eventually transported to the kidneys where another (-OH) group is added. The result is the active hormone called 1,25-Dihydroxyvitamin D (or 1,25-Dihydroxycalceferol, abbreviated 1,25-(OH)2D).
Vitamin D receptors are mainly activated by 1,25 (OH)2D, however, some cells may have a modest capacity to activate vitamin D locally. In addition, at a very high concentration the less active 25 (OH)D can bind with vitamin D receptors,
Genetic variations in the activating enzymes involved in this multi-step process are well documented and influence individual ability to activate the prohormone into the needed active product. This is why checking both the 25-(OH)D and the 1,25 (OH)2D levels may give us a better idea of the true level of bioavailable vitamin D.
Genetic variations in the activating enzymes involved in this process are well documented and influence individual ability to activate the prohormone. This is why checking both the 25-(OH)D and the 1,25 (OH)2D levels may give us a better…
Share on X
Vitamin D in Kidney Disease
In KD, the gradual loss of functional kidney tissue responsible for activation of vitamin D contributes to the deficiency of the active form 1,25 (OH)2D. Interestingly, more than 80% of KD patients also have a low level of the precursor form 25(OH)D when measured in the serum. Several factors have been implicated in the cause of this deficiency including inadequate outdoor physical activity, inadequate dietary intake, genetic variations, and impaired retention of the filtered 25(OH)D by the kidneys. There is also evidence that the accumulation of waste product, a common effect of KD, can decrease the production of 25(OH)D by the liver.
Remember, vitamin D circulates in the blood bound to DBP. Without this protein to provide transportation, vitamin D precursors will not reach the liver for the step of activation to 25(OH)D. Since some KD causes urinary protein loss, low levels of DBP may contribute to low 25(OH)D in kidney patients, however there’s conflicting evidence of the significance of this factor. There may be more to the story worth continued research especially surrounding the genetic variations in the gene that codes for DBP and its subsequent effect on production and binding capacity.
We can’t talk about vitamin D and KD without talking about the parathyroid glad (not to be confused with the thyroid gland). The main function of the parathyroid gland is to maintain blood levels of circulating calcium, a mineral very important in heart and bone health, as well as normal muscle function.
As kidney function declines, phosphate accumulation indirectly contributes to further reduction in vitamin D activation. These compounding factors promote production of parathyroid hormone (PTH) by the parathyroid gland. KD). PTH maintain calcium by influencing absorption from the gut as well as increasing its reabsorption during kidney filtration.
When calcium levels in the blood drop too low and endanger cardiac function, it triggers PTH to also mobilize calcium from the bone storage into the blood to normalize circulating levels. This is the contributing mechanism that leads to a high bone turnover, weakened bones, and increase the risk of fracture in kidney patients. In addition, vitamin D deficiency in kidney patients has been associated with muscle weakness, falls, insulin resistance, enlargement of heart muscles, blood vessel disease and calcifications.
vitamin D deficiency in kidney patients has been associated with muscle weakness, falls, insulin resistance, enlargement of heart muscles, blood vessel disease and calcifications
Share on X
The target level of vitamin D in kidney disease patients
There is controversy surrounding the ideal target goal of 25 (OH)D and 1,25(OH2)D for kidney patients. Studies showed that maximum benefit to decrease muscle weakness and fall risk in kidney patients is in the range between 24-44 ng/mL and that levels less than 15 ng/mL have been associated with increased risk for mortality and progression to dialysis in kidney patients. Conventionally, a target level of ~40 ng/mL but has been the standard of care, however, some practitioners argue that some patients may benefit from higher circulating levels.
Conventionally, a target level of ~40 ng/mL but has been the standard of care, however, some practitioners argue that some patients may benefit from higher circulating levels
Share on X
Conventional and Integrative approach to bone health in kidney disease
The conventional medicine approach to bone health in KD has been focused on correcting 1,25(OH)2D levels and decreasing PTH levels (though target levels in KD are unclear). Utilizing active vitamin D analogues has been linked to improved outcomes in KD and dialysis patients.
When addressing vitamin D’s impact on KD risk, we need to pay close attention to dietary factors that impact nutrient status (including calcium, magnesium, vitamin K and other relevant nutrients), as well as digestive issues that may reduce nutrient absorption from food. In addition, the role of genetic and epigenetic modifications in coding for factors that impact vitamin D activation and vitamin D receptors might mean that simple recommendations to “get more sun” or supplement may not be adequate for some individuals and warrant a more personalized approach to optimize kidney health.
References:
1. Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281.
2. Townsend, K.; Evans, K.N.; Campbell, M.J.; Colston, K.W.; Adams, J.S.; Hewison, M. Biological actions of extra-renal 25-hydroxyvitamin D-1alpha-hydroxylase and implications for chemoprevention and treatment.
3. J. Steroid Biochem. Mol. Biol. 2005, 97, 103–109. Souberbielle, J.C.; Body, J.J.; Lappe, J.M.; Plebani, M.; Shoenfeld, Y.; Wang, T.J.; Bischoff-Ferrari, H.A.; Cavalier, E.; Ebeling, P.R.; Fardellone, P.; et al. Vitamin D and musculoskeletal health, cardiovascular disease, autoimmunity and cancer: Recommendations for clinical practice. Autoimmun Rev. 2010, 9, 709–715.
4. Eknoyan, G.; Levin, A.; Levin, N.W. Bone metabolism and disease in chronic kidney disease. Am. J. Kidney Dis. 2003, 42, S1–S201.
5. Ishimura, E.; Nishizawa, Y.; Inaba, M.; Matsumoto, N.; Emoto, M.; Kawagishi, T.; Shoji, S.; Okuno, S.; Kim, M.; Miki, T.; et al. Serum levels of 1,25-dihydroxyvitamin D, 24,25 dihydroxyvitamin D, and 25-hydroxyvitamin D in nondialyzed patients with chronic renal failure. Kidney Int. 1999, 55, 1019–1027.
6. Nguyen-Yamamoto, L.; Karaplis, A.C.; St-Arnaud, R.; Goltzman, D. Fibroblast Growth Factor 23 Regulation by Systemic and Local Osteoblast-Synthesized 1,25 Dihydroxyvitamin D. J. Am. Soc. Nephrol. 2017, 28, 586–597.
7. Slatopolsky, E.;Weerts, C.; Thielan, J.; Horst, R.; Harter, H.; Martin, K.J. Marked suppression of secondary hyperparathyroidism by intravenous administration of 1,25-dihydroxy-cholecalciferol in uremic patients. J. Clin. Investig. 1984, 74, 2136–2143.
8. Kim, S.M.; Choi, H.J.; Lee, J.P.; Kim, D.K.; Oh, Y.K.; Kim, Y.S.; Lim, C.S. Prevalence of vitamin D deficiency and effects of supplementation with cholecalciferol in patients with chronic kidney disease. J. Ren. Nutr. 2014, 24, 20–25.
9. Cankaya, E.; Bilen, Y.; Keles, M.; Uyanik, A.; Akbas, M.; Gungor, A.; Arslan, S.; Aydinli, B. Comparison of Serum Vitamin D Levels Among Patients With Chronic Kidney Disease, Patients in Dialysis, and Renal Transplant Patients. Transpl. Proc. 2015, 47, 1405–1407.
10. Kalousova, M.; Dusilova-Sulkova, S.; Zakiyanov, O.; Kostirova, M.; Safranek, R.; Tesar, V.; Zima, T. Vitamin D Binding Protein Is Not Involved in Vitamin D Deficiency in Patients with Chronic Kidney Disease. Biomed. Res. Int. 2015, 2015, 492365.
11. Garcia-Canton, C.; Bosch, E.; Ramirez, A.; Gonzalez, Y.; Auyanet, I.; Guerra, R.; Perez, M.A.; Fernandez, E.; Toledo, A.; Lago, M. Vascular calcification and 25-hydroxyvitamin D levels in non-dialysis patients with chronic kidney disease stages 4 and 5. Nephrol. Dial. Transpl. 2010, 26, 2250–2256.
12. Namir, Y.; Cohen, M.J.; Haviv, Y.S.; Slotki, I.; Shavit, L. Vitamin D levels, vitamin D supplementation, and prognosis in patients with chronic kidney disease. Clin. Nephrol. 2016, 86, 165–174.
13. Kendrick, J.; Cheung, A.K.; Kaufman, J.S.; Greene, T.; Roberts,W.L.; Smits, G.; Chonchol, M. Associations of plasma 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D concentrations with death and progression to maintenance dialysis in patients with advanced kidney disease. Am. J. Kidney Dis. 2012, 60, 567–575.
14. Yousefzadeh P, Shapses SA, Wang X. Vitamin D Binding Protein Impact on 25-Hydroxyvitamin D Levels under Different Physiologic and Pathologic Conditions. Int J Endocrinol. 2014;2014:981581.
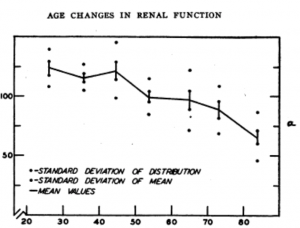
Each of us are born with a specific number of nephrons, the functional kidney units which does the job of filtering toxins out of our system. Depending on factors like diet, lifestyle factors, and genetics/epigenetics (to name a few), the rate of decline varies and results in different outcomes to each patient. So, is there anything we can do to reduce our risk of age-related kidney function decline to avoid dialysis?
What causes the Age-related kidney decline?
- Blood flow
Recent studies demonstrate that the blood flow to the kidneys decreases by approximately 10% every 10 years after age 40, affecting each nephron. In addition, the arteries of each nephron become less responsive to changes in blood pressure, increased pressure leads to damage and accelerated loss of kidney mass.
- Inflammation
Furthermore, as the lining of the filters inside the nephron become more permeable protein begin to seep through, contributing to increased inflammation. As time passes, inflammation leads to scarring of the filtration units. In fact, on average by the time an individual reaches the end of their 80’s, 30% of the nephrons are too damaged to be functional.
Inflammation leads to scarring of the filtration units. In fact, on average by the time an individual reaches the end of their 80’s, 30% of the nephrons are too damaged to be functional
Share on X
- Renin-Angiotensin System (RAS)
Factors that affects the blood flow to each nephron includes a system called the Renin-Angiotensin System (RAS). So that if the blood flow to the nephron declines, the nephron produces a chemical called Renin which promotes the production of another chemical called Angiotensin (Angiotensin I then Angiotensin II).
Angiotensin II increase the pressure inside the nephron and maintain adequate function by promoting “clamping” of some of the vessels in the nephron. With aging, production of Renin decreases. This results in a subsequent decline in the kidney’s ability to maintain good flow in response to changing blood pressure.
- Nitric Oxide production
Blood vessels are also controlled by Nitric oxide (NO2), another system that functions by expanding the blood vessels inside the nephron to help maintain good blood flow. This system also serves to protect the kidney from the formation of scar tissue.
Many factors help improving the production of NO2 while others lead to its breakdown. One example, are factors like genetics or availability of precursors that can influence the action of the enzyme responsible for making NO2, Nitric Oxide Synthetase (i.e. eNOS, iNOS, and nNOS). Another factor affecting the breakdown of Nitric Oxide are sex hormones. Estrogen, the dominant sex hormone in females, prevents the breakdown of NO2keeping its level high. Meanwhile, testosterone, the male dominant sex hormone, has the opposite effect of increasing its breakdown. This may in part explain why hypertension and kidney function decline is more pronounced in men than in women.
With aging, NO2 production declines and leads to the closing of the arteries in the kidney, sodium retention, and scar tissue formation. These factors make the aging kidneys more vulnerable to even minor changes in blood pressure, changes in hydration, and medication – in turn, making them more susceptible to acute kidney injury.
With aging, NO2 production declines and leads to the closing of the arteries in the kidney, sodium retention, and scar tissue formation
Share on X
What can be done to slow the rate of age-related kidney decline?
Conventional medicine focuses on using medications that block the RAS system such as Angiotensin Converting Enzyme (ACE) Inhibitors (i.e. enalapril, lisinopril) or Angiotensin II Receptor Blockers (ARBs) (i.e. losartan, irbesartan). There are a multitude of studies demonstrating their effectiveness in delaying the progression of various forms of kidney disease, including diabetic kidney disease, age-related kidney decline and others.
However, despite the delayed progression, patients continue to lose kidney function and end up requiring dialysis. Additionally, like all drugs, these medications come with side effects and risks*. Adverse effects include increased potassium retention, zinc depletion, and swelling of the throat, among other side effects. In fact, these medications have an established increased risk of acute kidney injury, particularly in elderly with kidney disease.
Arginine (or L-Arginine, we’ll use the terms interchangeably) is an amino acid that is essential for the body to produce NO2. It’s labeled as a conditionally essential amino acid because although the body can make it, production is dependent on factors including health and nutrition status. Levels tend to decline with age, therefore ensuring adequate intake through food sources or supplementation is important.
Arginine is a conditional essential amino acid because although the body can make it, production is dependent on factors including health and nutrition status. Levels tend to decline with age
Share on X
Arginine-rich foods include animal proteins, eggs fish, nuts (including walnuts, hazelnuts, pecans, peanuts, almonds, cashews, and Brazil nuts), seeds (especially pumpkin, also sesame and sunflower), oats, buckwheat, and cacao. Note, some of these foods are also high in phosphate which should be restricted in kidney patients even at early stages. (link-blog to write). With age and reduced gut function, absorption of nutrients from food may be diminished, so supplementation may be in order to prevent/slow age-related kidney decline. For kidney patients over 50-years-old, the recommendation* is to supplement 2 grams of L-Arginine three times a day to maintain good kidney circulation, stabilize function and prevent age-related kidney decline.
*Reminder: Always consult with your kidney doctor or primary care physician before taking any supplement, and never change or discontinue your medication without the instruction of your doctor.
References:
- Davies Df, Shock NW, Age Changes in glomerular filtration rate, effective plasma flow, and tubular excretory capacity in adult males. J Clin Ivest 1950; 29:496-507
- Llorens S, Fernandez AP, Nava E. Cardiovascular and renal alterations on the nitric oxide pathway in spontaneous hypertension and aging. Clin Hemorheol Microcirc 2007; 37:149-156
- Delp MD, Behnke BJ, Spier SA, Wu G, Muller-Delp JM. Aging diminishes endothelium-dependent vasodilation and tetrahydrobiopterin content in rat skeletal muscles arterioles. J Physiol 2008; 586:1161-1168
- Auelo JG. Normotensive ischemic acute renal failure. N Engl J Med 2007; 357:797-805
- Ahmed SB, Fisher ND, Hollenberg NK. Gender and the renal nitric oxide synthase system in healthy humans. Clin J Am Soc Nephron 2007;2:926-931






